Potentially the most common insect pest of canola crops, flea beetles first attack plants in the seedling to early vegetative stages, when they are most vulnerable. The crucifer and striped flea beetle species have the combined potential to cause significant damage that will impact the crop for the rest of the growing season. Therefore, a good understanding of these insects and a strong consideration for management strategies to control them is imperative to a successful canola crop.
Important tips for best management
- Seed into a warm, moist seedbed to promote good stand establishment and rapid seedling growth, which will allow the crop to outgrow the susceptible stage (up to the four-leaf stage) sooner, resulting in a shorter time period for damage. Conditions that slow canola development allow flea beetles a longer time frame to feed on the susceptible stage of the plant. After the four-leaf stage there will be minimal impact on yield 1.
- Note that delaying seeding too much to achieve a warm, moist seedbed may decrease final seed yield.
- In Western Canada, insecticidal seed treatments are provided by every canola seed supplier and can provide good levels of protection. Additional seed treatment offerings may provide enhanced or additional insect control.
- Scout fields and assess plant damage regularly (daily if possible) throughout the susceptible stage, including the stems and the undersides of leaves. If leaf area loss is below thresholds, but flea beetles are present in high numbers and the crop is not advancing, stem feeding may be the reason.
- Continue to scout until the seedlings are past susceptibility (after the four-leaf stage), especially when temperatures exceed 14 degrees Celsius and new feeding continues. When scouting, note that time of day and weather can influence flea beetle activity.
- On rainy or cool days, flea beetles will usually take cover; these conditions may encourage damage to stems or under leaves, or even stop feeding temporarily. Rain can also help the crop more quickly recover (in moisture limiting situations).
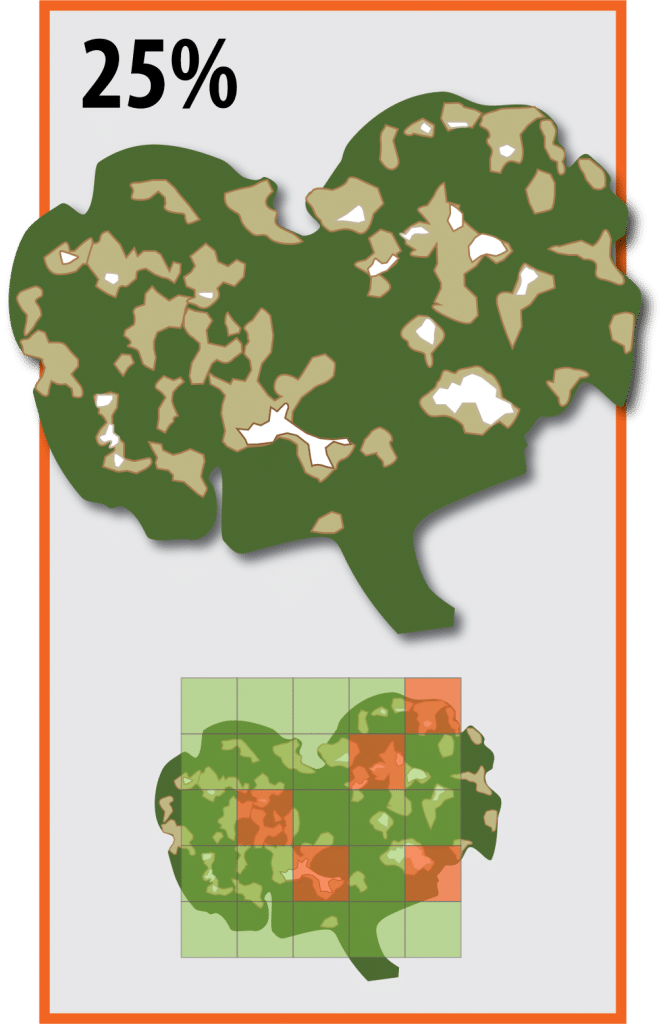
- Anticipate and follow the action threshold if considering applying foliar insecticide for flea beetles, which is typically at 25 per cent leaf area defoliation, if flea beetles are still present and actively feeding. For crops with very low plant populations this threshold may be lower.
- Recognize that predators, parasites and diseases can be important in regulating insect populations. However, since flea beetles emerge in large numbers during a short period of time, they tend to overwhelm the number of parasites and predators that attack them.
- Utilize cultural control methods to potentially reduce flea beetle damage, including:
- Creating a less favourable microclimate for flea beetles (ex. standing stubble or presence of residue) in direct seeded crops may reduce injury.
- Increasing seeding rates can help reduce the impact of flea beetle damage.
- For a given seeding rate, some research has demonstrated less flea beetle damage per plant for canola grown in 12 inch row spacing compared to narrower spacing 2.
- Note – When flea beetle populations are very high, no cultural controls will effectively reduce their attack.
- For more flea beetle management tips check out the Canola Watch fundamentals article: Flea beetles: Management tips.
- Flea beetle-related research can be found on the Canola Research Hub.
Flea beetle overview
Background

Flea beetles (Coleoptera: Chrysomelidae) feed on plants of the mustard family (Brassicaceae) grown across Canada (including the Peace River region) and throughout the Northern Great Plains of the United States. Eight flea beetle species are known to attack canola, mustard and rapeseed. Three main species that attack canola in western Canada are the:
- crucifer flea beetle – Phyllotreta cruciferae (Goeze)
- striped flea beetle – Phyllotreta striolata (F.)
- hop flea beetle – Psylliodes punctulata Melsh.
Where do flea beetles come from?
The crucifer flea beetle and the striped flea beetle were both introduced from Eurasia and are significant pests. Traditionally, striped flea beetles have been more common in the northern areas of Canada and crucifer flea beetles have been more common in the southern areas, as well as the most prevalent species across western Canada. However, the range and the population of the striped flea beetle has increased notably in the last few decades, becoming the most frequently encountered flea beetle species in central Alberta, central Saskatchewan and much of Manitoba 3,4.
The hop flea beetle is a native species that occurs in low numbers throughout the Prairies 5,6.
The economic impact of flea beetles on crop production varies with population densities. Yield losses of about 10 per cent are common where flea beetles are abundant (even when protected with insecticides) 7,8. A yield reduction of one per cent per acre results in a total crop loss of about 25 million to 35 million dollars. Annual crop losses in North America from flea beetles potentially exceed 300 million dollars 9.
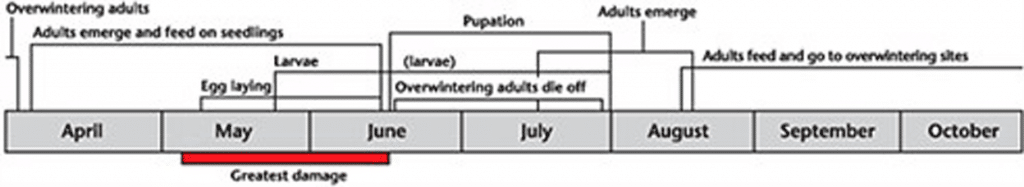
Flea beetle life cycle
Flea beetles have one generation per year in western Canada, although adults appear twice during the growing season. In the spring, overwintered adults emerge and feed on canola seedlings. In the fall, the adult offspring of those overwintering adults can be observed feeding on canola leaves, stems and seed pods.
Flea beetles overwinter (as adults) near the surface of the leaf litter, grass and debris beneath hedges, shelterbelts, poplar groves and in association with canola stubble and volunteer cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More plants. Within the leaf litter flea beetle densities may be as high as 140 to 250 beetles per square metre.
Flea beetles become active with the first extended period of warm weather in April and May, five to 11 days after leaf litter begins to thaw in the spring. Depending on temperature, it may take an additional three weeks before all the overwintering adults emerge.


Hop flea beetle species emerge first, followed by striped and then crucifer flea beetles. Hop flea beetle adults emerge about 10 days earlier than adult striped flea beetles, and the striped flea beetles emerge one to four weeks before the crucifer flea beetles 10. Peak emergence of the crucifer flea beetle occurs when ground temperatures reach 15 degrees Celsius 11.
Under cool conditions, flea beetles walk or hop into the adjacent cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More crops or weeds. They feed on volunteer canola and mustard, or on weeds such as wild mustard, flixweed, lamb’s quarters, stinkweed or peppergrass. Then they will move to newly emerged canola seedlings. When air temperatures exceed 14 degrees Celsius and winds are calm, they will fly, dispersing throughout the field and invading other fields, attacking seedlings as they emerge 12,13. After feeding (on host plants) has commenced, beetles mate repeatedly. Egg laying begins in late May and continues for about 30 days9,11. Some egg laying may continue until early August. Females deposit approximately 100 smooth, yellow, elongated, oval eggs 0.38 to 0.46 millimetres (0.01 to 0.02 of an inch) by 0.18 to 0.25 millimetres (0.006 to 0.009 of an inch) wide, either separately or in groups of three or four in the soil adjacent to the host plant’s roots 11. Eggs must be in contact with moist soil or they desiccate within a few hours. The eggs take about 12 days to hatch. Development from egg to adult takes about seven weeks. After laying eggs, the overwintering adults die off.
Flea beetle larvae are grub-like with off-white bodies and a brown head and anal plate. Larvae moult twice between days 25 to 34, completing three larval stages usually mid-June to late July. Full-grown larvae are about three to four millimetres (0.1 to 0.2 of an inch) long.

Larvae feed on the root hairs and taproots of seedlings. In a few cases, larvae have been observed burrowing into the plant near the juncture of the root and stem. When larval development is complete, larvae pupate in small earthen cells.
Flea beetle pupae are usually present in the field by early to mid-July. They are entirely white except for their eyes, which darken as the pupal stage progresses to completion. The body appendages are free and distinguishable. The pupal stage lasts for about seven to nine days.
Adult emergence begins after mid-July and continues until October 14. These late season flea beetles feed on the leaves, stems and pods of cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More plants. In late August and September, adults move into leaf litter and debris to overwinter.
Influence of environment
Sunny, warm, dry weather increases feeding activity. Cool, damp weather slows flea beetle activity (and may drive them down to the stems where they may feed, or the undersides of the leaves), but they tend to survive fairly well in spite of the reduced activity level. If the weather warms up, growers should continue to scout for damaging levels of flea beetles.
Flea beetle identification
Adult flea beetles which feed on canola, mustard and rapeseed are small, elliptical or oval-shaped and less than 2.5 millimetres (0.1 of an inch) long. When disturbed they use their powerful hind legs to jump away like a flea – hence, the name flea beetle.
Scouting techniques
Spring flea beetle populations and damage levels are unpredictable, but scouting and observing flea beetle densities in the fall can potentially provide some indication of populations the following spring.
Most injury occurs between the cotyledon to four-leaf stage. Heavy infestations can severely damage cotyledons, the first leaves, petioles and stems. It can also results in crop thinning and delayed crop development. Damage is most evident under poor growing conditions with limited crop growth, such as dry seedbed conditions and warm weather, which favours insect feeding. After the four-leaf stage, there will be minimal impact on yield 1, since plants are established and can generally outgrow the feeding damage. At this time, adult flea beetle numbers often begin to decline. The crop can usually compensate provided plant counts are adequate and large portions of the crop are not totally destroyed.
Feeding damage is most severe when flea beetles attack the growing point, limiting the plants’ ability to re-grow. Heavy attacks can destroy the entire crop, forcing growers to reseed or leave the field fallow. During windy, cold, inclement weather and periods of slow plant growth, flea beetles can feed on seedlings at or near the soil surface. This stem feeding reduces the plant’s ability to supply water to the leaves and particularly under hot, dry conditions, can result in seedling death. While stem feeding isn’t normally common, examining the canola cotyledon stems for damage when leaf feeding is occurring is an additional recommended scouting technique. No specific threshold exists to assist growers in evaluating the impact of stem feeding, but due to the function of the stem and its fragility when young, only low levels of feeding are required on new stems to cause plant fatality. However, seedlings that die from this are hard to find and seedling death could easily be confused with various other reasons, including cutworms, frost and seedling diseases.
Seedling loss from plant disease can also be mistaken for flea beetle damage. Dig damaged plants from the soil and examine carefully for evidence of disease on the shoots and roots. Consider marking individual plants while scouting to identify damage causes and severity. Delayed maturity from early flea beetle damage can expose the crop to adverse temperatures during flowering (ex. high temperatures) or to early fall frosts before the plants have matured. Uneven maturity at harvest reduces seed quality or yield. Delaying harvest to allow immature pods to ripen contributes to yield loss when over-ripe pods shatter during harvest. Harvesting too early can produce a crop with many immature seeds containing high chlorophyll levels, affecting seed quality and yield. Most of this damage can be prevented if canola is protected from flea beetle injury during the two to three weeks following emergence.
Flea beetles that emerge after mid-July can also affect yield. Heavy feeding during pod development and filling causes injury to pods, leading to premature pod drying, shriveled seeds, pod shattering, increased chlorophyll content, and encourages fungal growth within the pods during damp weather. Injury to the pods is usually concentrated on the youngest pods and on late seeded crops. Yield is not usually affected when the crop has matured to at least the stage when seeds in the lower pods have turned from translucent to green.
Flea beetle management
Action thresholds and economic injury
Leaf defoliation levels should be used to decide whether a foliar spray for flea beetle control will be economical. Since seed treatments are standard practice, the use of action thresholds (nominal economic thresholds) for flea beetle control in canola are only used when foliar sprays are being considered.
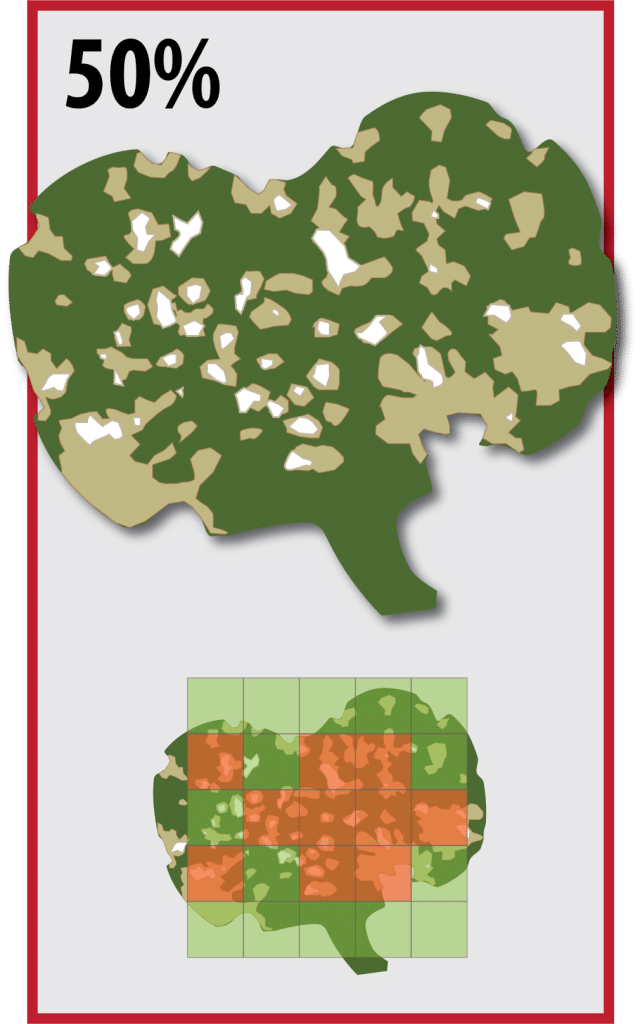
Canola seedlings may withstand 50 per cent leaf loss without significant yield penalty. However, the action threshold for flea beetle feeding on canola is actually when there is 25 per cent defoliation and flea beetles are continuing to feed. The reason for this discrepancy is that leaf area loss can escalate very quickly (sometimes even over the course of a single day) in a severe flea beetle infestation from 25 per cent (action threshold) to 50 per cent (level of damage anticipated to cause economic injury). With increased defoliation, plant tissue damage over and above simple feeding results in progressively less green leaf material for flea beetles to eat and further increases the rate of crop damage. Numerous provincial crop protection guides (e.g. from Manitoba, Saskatchewan, Alberta, etc.) refer to the 25 per cent action threshold as the nominal economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More in order to allow growers to proactively manage this aggressive insect pest. Therefore, anticipating 25 per cent defoliation and applying foliar insecticide controls will reduce the risk of flea beetles reaching a level where yield loss and plant development are substantially reduced. Research to re-evaluate these thresholds found that canola with up to 25 per cent leaf area loss will yield the same as canola with no leaf area loss at all.
Note that the action threshold of 25 per cent and the 50 per cent level of damage anticipated to cause economic injury are based on previous optimal target plant population recommendations of 75 to 150 plants per square metre (seven to 14 plants per square foot) and older seed treatments, so growers should take plant stand and growing conditions into consideration when making decisions.

Thinner plant stands will require a foliar insecticide application at a lower defoliation percentage than an optimal plant stand, because in this situation maintaining plant health is more critical to maximize production per plant in order to compensate for reduced plant numbers. Any plant mortality is also more likely to directly impact yield when plant density is already lower or less uniform than optimal.
When assessing whether a foliar insecticide spray is needed, growing conditions, soil moisture, overall crop health, yield potential and commodity prices all need to be considered along with flea beetle numbers and damage (both to leaves and stems). Control decisions should be based on current defoliation and the potential for crop growth to exceed feeding by flea beetles going forward. If soil moisture and growing conditions support rapid canola growth and seedlings are vigorous, the crop may be able to stand more feeding than slow growing drought stressed canola. However, when flea beetle feeding is combined with poor plant growth during hot, dry weather, canola can tolerate less feeding than if plants are growing under more ideal growing conditions.
Since it is the damage to the youngest tissue that is of immediate concern, the damage to the newest growth should be assessed to determine if feeding levels are decreasing or increasing.
When scouting fields for flea beetle damage, it is important to understand that flea beetles generally invade canola fields from the field edges. Flea beetle damage and the number of flea beetles may be higher at the field edge than farther into the field. An insecticide application to field perimeters may control invading beetles if they have not spread throughout the field. On hot and calm days, flea beetles are capable of moving longer distances and may populate the field more uniformly.
Flea beetle numbers can be very high in ripening canola in the fall. If crops are very late in maturity, extensive feeding on pods can lower yields and increase green seed content. However, seed yields are usually not affected when crop maturity is more advanced and the seeds in lower pods have turned from translucent to green 15.
Steps to ensure the proper flea beetle spray decision
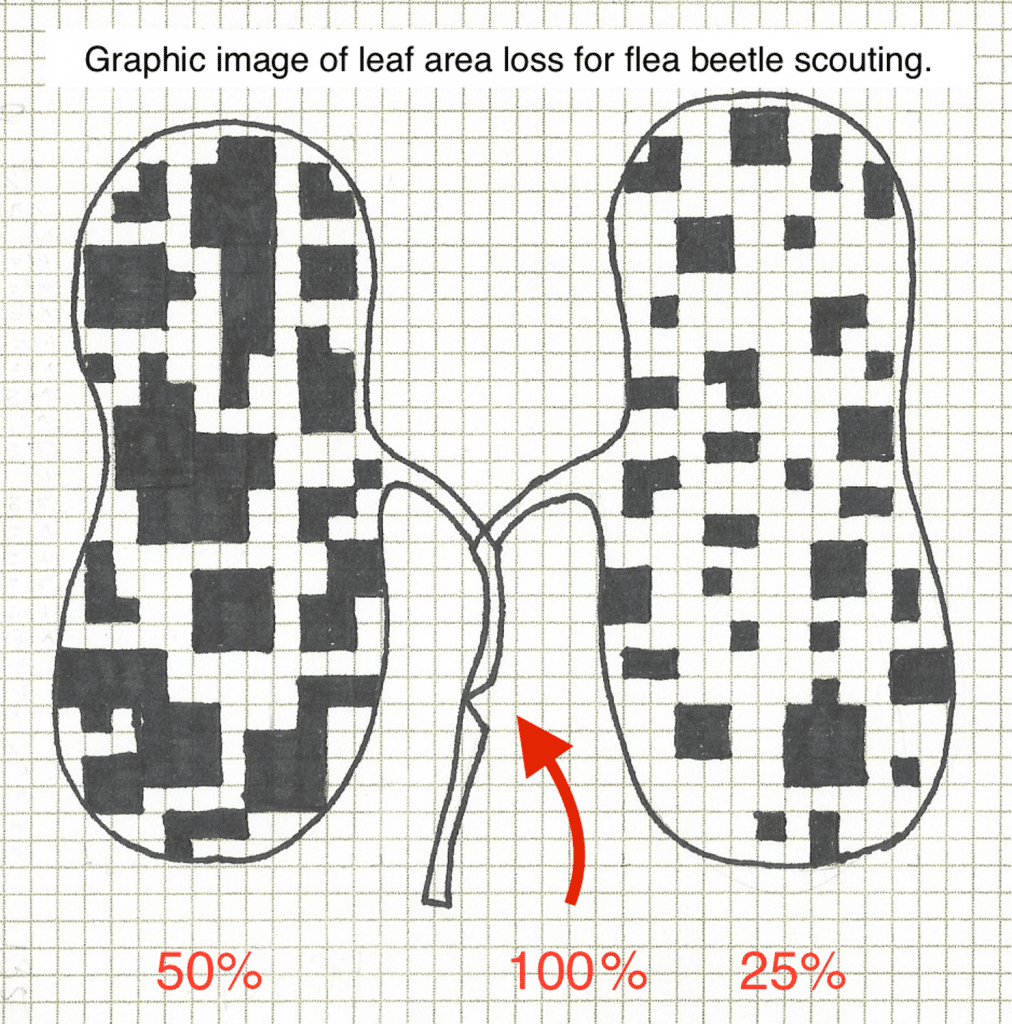
Step 1: Recall the spray threshold
The action threshold for flea beetles in canola crops in Canada is an average leaf area loss of 25 per cent or more. Yield loss starts to show after 25 per cent leaf area loss and becomes economic (yield loss is more than the total cost of the foliar spray operation) at around 50 per cent leaf area loss. The reason for the ‘action threshold’ for flea beetles is that leaf area loss can escalate quickly from 25 per cent to 50 per cent and beyond in a severe infestation.
Step 2: Assess leaf area loss
With thresholds in mind, check plants in a number of areas of the field to get an average damage level, scouting the newest leaves (as these tend to be preferred). Flea beetle populations and damage tend to be in concentrated areas. If cotyledons are chewed up but newest leaves show very little feeding, then plants may be outgrowing the threat or seed treatments may be having an effect (or both). However, if cotyledons are showing less than 50 per cent damage but first true leaves are not present, flea beetles may be nipping off fresh apical meristem – the growing point for first leaves. If the meristem is lost, these leaves could be considered 100 per cent lost. Mark those plants (could use the ‘washer’ test) and return in a day or two to see if true leaves are showing.
Cotyledons may show significant damage, but if the first leaves are in good shape, the flea beetle threat may have subsided.
Step 3: Assess stem damage

This is especially important in unfavourable weather (wind, cool) that drives flea beetles down to leaf undersides and stems. While feeding marks on the stem are likely much more consequential than feeding on a leaf, there isn’t currently a way to include stem feeding in the leaf-area loss thresholds. One shallow feeding mark (could be a few flea-beetle-sized bites) might not do any damage at all, but feeding that eats through the fragile new stem or causes it to break could be fatal. Plants that die due to stem feeding would have 100 per cent leaf area loss – but seedlings that die in this fashion are hard to find and could also be lost for various other reasons, including cutworms, frost and seedling diseases. They are often missed when it comes to damage assessment. The key is to look for stem damage when scouting, particularly if conditions are, or have recently been, cool, moist or windy. If leaf area loss is below thresholds, but flea beetles are present in high numbers and the crop is not advancing, stem feeding may be the reason. Growers and agronomists observing this situation may want to be more aggressive with their spray decision, especially if the stand is thin (when losing more plants is of greater concern).
Daily scouting needs to happen in areas approaching action threshold. Within two or three days, flea beetle feeding can escalate way beyond threshold, and actually wipe out whole areas of a field, especially in hot, dry and windy conditions.
Step 4: Assess flea beetle feeding activity
Consider if the insects still present in the field and continuing to feed. If it looks like populations are dwindling, an application may not be necessary. If flea beetles are slow and lethargic, it may mean they have ingested seed treatment insecticide and are no longer feeding. If they don’t jump away from your hand, they are likely near the end of their life. Check again to make sure before making a decision. Time of day and weather can influence flea beetle activity. On rainy days, for example, flea beetles will usually take cover, so rain will slow or even stop feeding for the time being. Rain can also help the crop more quickly recover.
Step 5: Consider the plant stand

This should influence your approach to the thresholds. With a thin stand of four plants per square foot, for example, growers can’t afford to lose any plants and may want to take action quickly as damage approaches 25 per cent. But with counts in the high end of the recommend range (of five to eight plants per square foot), growers can afford to lose a couple of plants without sacrificing yield.
Step 6: Check the crop stage
After the four-leaf stage, the threat is likely over because the crop usually has enough plant material to feed flea beetles without compromising growth, and the plants can compensate for feeding by this stage. If the crop is uneven (some plants are at the four-leaf stage and some are earlier), keep scouting until most of the crop has at least three or four true leaves.
Step 7: Check canola fields frequently
Seed treatment insecticide starts working when the seed imbibes water and it can remain active for about three to four weeks after that point. If you have enough moisture for seed to imbibe and germinate, you have enough to solubilize the active ingredient and allow it to move into the plant. Flea beetles need to consume some plant tissue to get a dose of the insecticide. High levels of flea beetles can overwhelm seed treatments. In a slow developing crop, seed treatments can lose their protective capacities before the crop reaches the four-leaf stage. Scout often during these first three weeks, and if flea beetle levels are building and are close to thresholds, daily checking for a few days may be required.
Step 8: If spraying is required, only use pesticidesPesticides (herbicides, insecticides or fungicides) used to protect against or reduce the amount of damage caused by weeds, pest insects or plant diseases. More registered for flea beetles in canola
Refer to product labels for proper use instructions. No pre-harvest interval concerns exist at this stage, but this could be a deciding factor on which product to use later in the season: Specific products are limited to one application per season and a grower may want to keep these in reserve. See the insecticide control options section for further detail.
Insecticide control options
Canola, mustard and rapeseed crops can be protected from flea beetle attack through insecticide application as a seed treatment or post-emergence foliar sprays. Granular, in-furrow insecticides were once popular, but have been withdrawn from the market due to environmental concerns.
Since flea beetles emerge early in the spring and can migrate into seedling canola fields rapidly, insecticide-coated seed treatments are the most effective means of flea beetle control 6,16,17.
Since 2001, all seed treatments registered for control of flea beetles in Canada contain a neonicotinoid insecticide. This insecticide class has systemic activity in plants and inhibits nervous conduction in insects by blocking the nicotinoid acetylcholine receptor 18.
Coating seeds with both insecticide and fungicide seed treatment formulations prior to planting is a widespread practice. Essentially all of the canola grown in western Canada is treated with an insecticide seed treatment 19. Seed treatments protect canola seedlings from flea beetles as soon as the plants emerge. Currently available neonicotinoid-based seed treatments may also have the option to include an additional Group 4C, 4D or 28 insecticide as part of the seed treatment package. Depending upon the rate selected, these particular enhanced seed treatment offerings can extend the period of protection from flea beetles, with some improvement in striped flea beetle control, and the addition of cutworm control with the Group 28 insecticide especially under conditions that lead to slow crop emergence and development (e.g. early and/or direct seedingA seeding method where the crop is sown into the field without any previous spring tillage operations. More into cool soil temperatures).

In a greenhouse trial when crucifer flea beetles were exposed to seedlings of canola grown from neonicotinoid-treated seed, they fed less and had higher mortality than striped flea beetles 20. The differential toxicity of seed treatments to the two species may result in a shift in their geographic distribution and economic importance. Also, for growers in regions where striped flea beetles are naturally more prevalent, the importance of frequent crop scouting to assess the need for foliar insecticide rescue treatments may be increased.
If high flea beetle populations occur and the seed treatments are not protecting the seedlings adequately, a foliar spray should be considered. Application of foliar insecticide may be required when feeding damage is anticipated to encompass 25 per cent of the leaf surface 21,22. If damage around field edges is high but flea beetles have not dispersed throughout the field, limiting insecticide application to the field perimeter may be sufficient to control the insect. Always check provincial crop protection guides for a list of insecticides registered for flea beetle control and follow product labels when applying products.
- Alberta Crop Protection Guide (The Blue Book)
- Saskatchewan Guide to Crop Protection
- Manitoba Guide to Field Crop Protection
Growers who routinely mix a foliar insecticide with their herbicides should check that flea beetle damage is actually at the threshold. Spraying before damage reaches the level of damage anticipated to cause economic injury can increase the risk of flea beetles developing resistance to insecticide, and has a negative impact on beneficial insects.
Regular field scouting for flea beetles is important, particularly on hot, calm days. If plants are missing, determine if the cause is flea beetles, cutworms, seedling blights or other causes. A post-emergent insecticide application may be required to protect seedlings that are exposed to severe or prolonged periods of intense attack. If heavy flea beetle damage nears threshold levels and high numbers of flea beetles are noticed in the field, apply foliar sprays as soon as possible, since flea beetles can cause substantial damage quickly.
Where damage starts at the field margins, it is possible that only a small portion of the field may require treatment. Apply sprays when it is sunny and warm, and the beetles are active and exposed on plants and soil. Additional foliar sprays may sometimes be needed since flea beetles may continue to move into fields at the susceptible stage after residual from the first foliar spray has become ineffective. Scout fields daily to determine if the insects have moved into the untreated areas and are damaging plants.
Table 1. Insecticide products registered for control of flea beetles in canola
*LD50 (mammalian toxicity) values represent the relative toxicity of a pesticide. They represent the dose (in mg/kg body weight) that will kill 50% of the test animals. Thus the lower the number the greater the toxicity. Values given are for oral LD50.
Note – Always check your provincial crop protection guide for current listings of registered control products, and always read and follow current pesticide label directions.
Source: Alberta Crop Protection Guide 2025 (The Blue Book), the 2025 Manitoba Guide to Field Crop Protection and applicable product safety data sheets.
Preventative strategies
Agronomic practices that promote good stand establishment and rapid seedling growth will reduce the impact of flea beetles on canola yield. For example, targeting a plant stand of five to eight plants per square foot should ensure that the crop can withstand some feeding prior to reaching a defoliation threshold. Seed treatments, tillage choices, seeding date, seeding rate and row spacing can also impact flea beetle effects on canola fields.
Seed treatment
All canola seed marketed in western Canada currently includes a Group 4A neonicotinoid insecticide. Additional seed treatments, which offer increased flea beetle and/or cutworm control, include a Group 4C insecticide (sulfoxaflor), a Group 4D insecticide (flupyradifurone) and a Group 28 insecticide (cyantraniliprole). Striped flea beetle has shown to be more tolerant to the effects of neonicotinoid seed treatment insecticides than crucifer flea beetles 23. Feeding still occurs when seed treatments are used, as insecticide ingestion is required to control flea beetles. Prior to considering the addition of supplementary insecticide or biological seed treatment products, action thresholds (or nominal economic thresholds), expected plant stands and anticipated damage levels should all be considered (as well as cost). When feeding damage exceeds the action threshold, a foliar insecticide application should be considered to limit economic loss. See the insecticide control options section for more information.
A practice that has been of interest to some growers and researchers from the standpoint of reducing cost and insecticide load on the environment is blending a proportion of seed without insecticide together with treated seed for planting. Under moderate levels of flea beetle damage, research has demonstrated that as much as one-third of planted seed could be untreated with insecticide without subsequent seed yield reduction 19. However, this practice is currently not recommended because of the highly variable and unpredictable nature of flea beetle damage, and the potential for the use of farm saved seed for the untreated proportion (which has other negative agronomic and legal risks). This strategy could be further explored in the future in conjunction with other integrated management strategies or with improved forecasting.
Tillage
Flea beetles prefer bright relatively warm conditions. Direct seedingA seeding method where the crop is sown into the field without any previous spring tillage operations. More provides a microclimate that is less ideal for flea beetles than tillage before seeding. Planting into stubble may provide a microclimate that is cooler, moister and less favourable to flea beetles. Injury may be reduced due to the cooler microenvironment created by stubble shading on surface soil. Cooler temperatures at the soil surface slow flea beetle activity reducing damage. However, crop development can also be slowed by cooler temperatures, so frequent crop scouting is still essential to ensure damage thresholds are not exceeded.
In Alberta, zero till canola production has been found to reduce flea beetle damage to levels equivalent to those found with the use of insecticidal seed treatments 2.
Weed control in nearby fields can contribute to additional insect pressure or damage within a canola crop. Nearby fields may contribute additional flea beetles if weed control or tillage operations leave flea beetles looking for food sources before the crop passes the four-leaf stage. Leaving a trap strip of volunteer canola near overwintering sites can be an effective control strategy if the trap strip is sprayed before beetles move into the canola crop, although this strategy can be difficult to implement successfully.
Seeding date
Producers are urged to seed canola as early as practical to maximize seed yield. Early seeding will maximize plant size before flea beetle emergence and the larger plants tolerate more injury 24. One study completed in southern Alberta, found canola planted in April had fewer flea beetles and suffered less damage than canola planted in May, while the opposite was true in central and northern Alberta 25.
Seeding rate
Increasing seeding rates can help reduce the impact of flea beetle damage, by spreading the feeding damage over more plants per unit area, resulting in less feeding damage per plant and allowing seedling to recover more readily from flea beetle injury.
Other research found damage to individual plants to be lower with a 10 kilogram per hectare (8.9 pounds per acre) seeding rate than with a five or 7.5 kilogram per hectare (4.5 or 6.7 pounds per acre) rate 26. Higher seeding rates and plant densities are believed to dilute and reduce damage to individual plants. However, the benefits of higher seeding rates for flea beetle control need to be tempered by the costs, which include increased production costs and pesticide load to the environment and the possibility of increased lodging and susceptibility to diseases. Hybrid specific recommendations for lower planting densities, typically for reducing lodging concerns and to optimize performance of specific cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, should also be taken into consideration.
Seed costs can represent one of the single highest investments in the canola production year. Growers are well advised to prioritize all of their investments in trash management, seeding tools, genetics, additional traits and seed treatment costs on a field by field basis. In some cases, it will be difficult to determine if challenging seedbed conditions or anticipated insect pressure can be best managed by a different choice of crop, additional canola seed or enhanced seed treatment protection. Practices that encourage higher plant establishment rates such as slower seeding speeds, optimal planting depth, only safe rates of seed placed fertilizer can reduce the risk flea beetle damage poses by encouraging good crop establishment.
Row spacing
At a given seeding rate, row spacing of 20 to 30 centimetres (eight to 12 inches) have been shown to result in less flea beetle damage per plant than 10-centimetre (four inch)- row spacing 27. Although the reasons are not yet clear, it appears that flea beetles are more attracted to the reduced visual contrast between vegetation and soil that occurs at narrow row spacing.
Crop rotation
Crop rotation is not an effective means of controlling flea beetles. Adults overwinter inside and outside of the cropped areas and are capable of long range migration. While intercropping with non-host plants has reduced flea beetle damage in BrassicaceousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More vegetables, the benefits of field-scale intercropping of canola and wheat 28 or canola and field peas 29 is currently are not sufficient to warrant their use as flea beetle management options.
Natural enemies and biological control
Predators, parasites and diseases can be important in regulating insect populations. To date, the effect of biological control agents seems to be limited, but several insects have been observed attacking adult flea beetles.
The flower beetle, Collops vittatus (Say) 30, big-eyed bug, Geocorus bullatus (Say) 31, damsel bugs (Nabis spp.) 32, field crickets (Gryllus pennsylvanicus Berm.) 33 and lacewings (Chrysopa spp.) play a minor role in controlling flea beetles.
The native braconid wasp (Microctonus vittatae Muesebeck) parasitizes flea beetle adults and is widespread across the Prairies 34. However, parasitism by the wasp usually averages less than five per cent of the flea beetle population 35,36.
A braconid wasp, Townesilitis bicolor (Wesmael), that inhabits central Europe, was found to parasitize up to 50 per cent of the crucifer-feeding flea beetles there 37. This wasp was imported and released in Manitoba in multiple introductions from 1978 to 1983, but did not become established for unknown reasons.
Isolates of entomopathogenic nematodes that are promising candidates for the biological control of P. striolata under field conditions have been recently found in China 38. Unfortunately, flea beetles emerge in large numbers during a relatively short period of time and tend to overwhelm the parasites and predators.
Resistance breeding
While no canola varietyA variety is a variant of a species that evolved in nature without the intervention of humans, e.g. Brassica oleracea variety (in short form, var.) botrytis (cauliflower), var. capitata (cabbage), var. italica (broccoli), etc. More has resistance to flea beetle, research aimed at developing resistance has been done, since several studies have shown that flea beetles prefer certain crucifer hosts 39,40,41.
The lower levels of flea beetle feeding observed on yellow mustard seedlings compared to canola seedlings is likely due to an aversion to the levels of glucosinalbin in mustard seedlings and to seedling tolerance to feeding injury 42,43 Trichomes (leaf hairs) also impart resistance to flea beetles in certain crucifers 44,45. Researchers have found canola germplasm with increased numbers of hairs on young true leaves displayed resistance to flea beetles in field trials 46,47, in some cases surpassing the protection level provided by neonicotinoid seed treatments. These hairs may have discouraged flea beetles from walking up hairy stems. Past research has explored transgenic options for this trait, but current research continues on non-transgenic canola lines with further elevated numbers, lengths, and complexities of plant leaf hairs that may achieve levels of resistance to flea beetles at present seen only with insecticide use.
Footnotes
- Lamb, R.J. 1984. Effects of flea beetles, Phyllotreta spp. (Coleoptera: Chrysomelidae), on the survival, growth, seed yield and quality of canola, rape and yellow mustard. Can. Entomol., 116, 269-280.[↩][↩]
- Dosdall, L.M., Dolinski, M.G., Cowle, N.T., & Conway, P.M. 1999. The effect of tillage, row spacing, and seeding rate on feeding injury by flea beetles, Phyllotreta spp. (Coleoptera: Chrysomelidae) in canola in central Alberta, Canada. Crop Prot., 18, 217-224.[↩][↩]
- Soroka, J.J. 2012. Potential flea beetles species composition shift in Prairie canola. CARP project 2009-6.[↩]
- Costamagna, A. 2017. Management of Flea Beetles on the Canadian Prairies. University of Manitoba. Presentation. Retrieved from: http://umanitoba.ca/faculties/afs/agronomists_conf/media/5_PM_DEC_13_COSTAMAGNA_MAC_fleaBeetles_Costamagna_2017v2.pdf.[↩]
- Chittendon, F.H. 1923. Notes on the distribution and habits of North American Phyllotreta (Coleop.) Proc. Entomol. Soc. Wash., 25, 131-139.[↩]
- Wylie, H.G., & Loan, C. 1984. Five Nearctic and one introduced euphorine species (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Braconidae) that parasitize adults of crucifer-infesting flea beetles (Coleoptera: Chrysomelidae). Can. Entomol., 116, 235-246.[↩][↩]
- Lamb, R. J., & Turnock, W.J. 1982. Economics of insecticidal control of flea beetles (Coleoptera: Chrysomelidae) attacking rape in Canada. Can. Entomol., 114, 827-840.[↩]
- Madder, D.J., & Stemeroff, M. 1988. The economics of insect control on wheat, corn, and canola, 1980-1985. Bull. Entomol. Soc. Can. (Suppl.), 20, 1-22.[↩]
- Knodel, J.J., & Olson, D.L. 2002. Crucifer flea beetle: biology and integrated pest management in canola. North Dakota State Univ. Coop. Ext. Serv. Publ. E1234. North Daklota State University, Fargo, ND.[↩][↩]
- Westdal, P.H., & Romanow, W. 1972. Observations on the biology of the flea beetle Phyllotreta cruciferae (Coleoptera: Chrysomelidae) Manitoba Entomol., 6, 34-45.[↩]
- Ulmer, B.J., & Dosdall, L.M. 2006. Emergence of overwintered and new generation adults of the crucifer flea beetle, Phyllotreta cruciferae (Goeze) (Coleoptera: Chrysomelidae). Crop Prot., 25, 23-30.[↩][↩][↩]
- Burgess, L. 1977. Flea beetles (Coleoptera: Chrysomelidae) attacking rape crops in the Canadian rairie provinces. Can. Entomol., 109, 21-32.[↩]
- Lamb, R.J. 1983. Phenology of flea beetle (Coleoptera: Chrysomelidae) flight in relation to their invasion of canola fields in Manitoba. Can. Entomol., 115, 1493-1502.[↩]
- Wylie, H.G. 1979. Observations on distribution, seasonal life history, and abundance of flea beetles (Coleoptera: Chrysomelidae) that infest rape crops in Manitoba. Can. Entomol., 113, 665-671.[↩]
- Soroka, J.J. 2009. Effects of late season flea beetle feeding on canola yields. Final Report February 2009. Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More. Pest Management Centre. Pesticide Risk Reduction Strategies Initiative PRR06-110. Ottawa, ON. 37.[↩]
- Beran, F., Mewis, I., Ramasamy, S., Svoboda, J., Vial, C., Mosimann, H., Boland, W., Büttner, C., Ulrichs, C., Hansson, B.S., & Reinecke, A. 2010. Male Phyllotreta striolata(F.) produce an aggregation pheromone: Identification of male-specific compounds and interaction with host plant volatiles. J. Chem. Ecol., 37, 85-97. https://doi.org/10.1007/s10886-010-9899-7[↩]
- Weiss, M.J., McLeod, P. Schatz, B.G. & Hanson, B.K. 1991. Potential for insecticidal management of flea beetle (Coleoptera: Chrysomelidae) on canola. J. Econ. Entomol., 84, 597-1603.[↩]
- Tomizawa, M., & Casida, J. E. 2005. Neonicotinoid insecticide toxicology: Mechanisms of selective action. Ann. Rev. Pharmacol. Toxicol., 45, 247-268.[↩]
- Soroka, J.J., Grenkow, L.F., & Irvine, R.B. 2008. Impact of decreasing ratios of insecticide-treated seed on flea beetle (Coleoptera: Chrysomelidae, Phyllotreta spp.) feeding levels and canola seed yields. J. Econ. Entomol., 101, 1811-1820.[↩][↩]
- Tansey, J.A., Dosdall, L.M. Keddie, B.A., & Sarfraz, R.M. 2008. Differences in Phyllotreta cruciferae and Phyllotreta striolata (Coleoptera: Chrysomelidae) responses to neonicotinoid seed treatments. J. Econ. Entomol., 101, 159-167.[↩]
- Cárcamo, H.A., & Blackshaw, R.E. 2007. Insect pest incidence and injury to herbicide-tolerant canola in western Canada. Agron. J., 97, 1570-1578.[↩]
- Knodel, J.J., Olson, D. L., Hanson, B.K., & Henson, R.A. 2008. Impact of planting dates and insecticide strategies for managing crucifer flea beetles (Coleoptera: Chrysomelidae) in spring-planted canola. J. Econ. Entomol., 101, 810-821.[↩]
- Tansey, J., Dosdall, L.M., Keddie, B.A., Sarfraz, R.M. 2008. Differences in Phyllotreta cruciferae and Phyllotreta striolata (Coleoptera: Chrysomelidae) responses to neonicotinoid seed treatments. Journal of Economic Entomology (Insecticide resistance and resistance management), 101(1), 159-167.[↩]
- Dosdall, L.M., & Stevenson, F.C. 2005. Managing flea beetles (Phyllotreta spp.) (Coleoptera: Chrysomelidae) in canola with seeding date, plant density, and seed treatment. Agron. J., 97, 1570-1578.[↩]
- Cárcamo, H.A., Otani, J.K., Dosdall, L.M., Blackshaw, R.E., Clayton, G.W., Harker, K.N., O’Donovan, J. T., & Entz, T. 2008. Effects of seeding date and canola species on seedling damage by flea beetles in three ecoregions. J. Appl. Entomol., 132, 623-631.[↩]
- Elliott, R.H., Franke, C., & Rakow, G.F.W. 2008. Effects of seed size and seed weight on seedling establishment, vigourSeed properties that determine the potential for rapid uniform emergence and development of normal seedlings under a wide range of field conditions. More and tolerance of Argentine canola (Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More) to flea beetles, Phyllotreta spp. Can. J. Plant Sci., 88, 207-217.[↩]
- Dosdall, L.M., Dolinski, M.G., Cowle, N.T., & Conway, P.M. 1999. The effect of tillage, row spacing, and seeding rate on feeding injury by flea beetles,Phyllotreta spp. (Coleoptera: Chrysomelidae) in canola in central Alberta, Canada. Crop Prot., 18, 217-224.[↩]
- Hummel, J.D., Dosdall, L.M., Clayton, G.W., Turkington, T.K., Lupwayi, N.Z., Harker, K.N., & O’Donovan, J.T. 2009. Canola-wheat intercropping for improved agronomic performance and integrated pest management. Agron. J., 101, 1190-1197.[↩]
- Weiss, M.J., Schatz, B.G., Gardner, J.C., & Nead, B.A. 1994. Flea beetle (Coleoptera: Chrysomelidae) populations and crop yield in field pea and oilseed rape intercrops. Environ. Entomol., 23, 654-658.[↩]
- Gerber, G., & Osgood, C. 1975. Collops vittatus (Coleoptera: Melyridae): a predator of flea beetle adults in rapeseed (Phyllotreta cruciferae). Manitoba Entomol., 9, 61.[↩]
- Burgess, L. 1977. Geocoris bullatus, an occasional predator on flea beetles (Hemiptera: Lygaeidae). Can. Entomol., 109, 1519-1520.[↩]
- Burgess, L. 1982. Predation on adults of the flea beetle Phyllotreta cruciferae by the western damsel bug, Nabis alternatus(Hemiptera: Nabidae). Can. Entomol., 114, 763-764.[↩]
- Burgess, L., & Hinks, C.F. 1987. Predation on adults of the crucifer flea beetle, Phyllotreta cruciferae (Goeze), by the northern field cricket, Gryllus pennsylvanicus Bermeister (Orthoptera: Gryllidae). Can. Entomol., 119, 495-496.[↩]
- Soroka unpublished.[↩]
- Wylie, H.G. 1981. Effects of collection method on estimates of parasitism and sex ratio of flea beetles (Coleoptera: Chrysomelidae) that infest rape crops in Manitoba. Can. Entomol., 113, 665-671.[↩]
- Wylie, H.G. 1982. An effect of parasitism by Microctonus vitattae (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Braconidae) on emergence of Phyllotreta cruciferae and Phyllotreta striolata Coleoptera: Chrysomelidae) from overwintering sites. Can. Entomol., 114, 727-732.[↩]
- Wylie, H.G. 1988. Release in Manitoba of Townesilitus bicolor (Hym: Braconidae), an European parasite of Phyllotretaspp. (Coleoptera: Chrysomelidae). Entomophaga, 33, 25-32.[↩]
- Xu, C., De Clercq, P. Miens, M. Chen, S., & Han, R. 2010. Efficacy of entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) against the striped flea beetle, Phyllotreta striolata. BioControl, 55, 789-797.[↩]
- Lamb, R.J., & Palaniswamy, P. 1990. Host discrimination by a crucifer-feeding flea beetle, Phyllotreta striolata (F.) (Coleoptera: Chrysomelidae). Can. Entomol., 122, 817-824.[↩]
- Palaniswamy, P., Lamb, R.J., & McVetty, P.B.E. 1992. Screening for antixenosis resistance to flea beetles, Phyllotreta cruciferae (Goeze) (Coleoptera: Chrysomelidae), in rapeseed and related crucifers. Can. Entomol., 124, 895-906.[↩]
- Anderson, M.D., Peng, C., & Weiss, M.J. 1992. Crambe, Crambe abyssinica Hochst., as a flea beetle resistant crop. J. Econ. Entomol., 85, 594-600.[↩]
- Bodnaryk, R.P. 1991. Developmental profile of sinalbin (p-hydroxybenzylglucosinolate) in mustard seedlings, Sinapis alba L., and its relationship to insect resistance. J. Chem. Ecol., 17, 1543-1556.[↩]
- Bodnaryk, R.P., & Lamb, R.J. 1991. Mechanisms of resistance to the flea beetle, Phyllotreta cruciferae (Goeze), in mustard seedlings, Sinapis alba L. Can. J. Plant Sci., 71, 13-20.[↩]
- Lamb, R.J. 1980. Hairs protect pods of mustard from flea beetle damage. Can. J. Plant Sci., 60, 1439-1440.[↩]
- Palaniswamy, P., & Bodnaryk, R. P. 1994. A wild Brassica from Italy provides trichome resistance against flea beetles. Can. Entomol., 126, 1119-1120.[↩]
- Gruber, M.Y., Wang, S., Ethier, S., Holowachuk, J., Bonham-Smith, P.C., Soroka, J.J., & Lloyd, A. 2006. “HAIRY CANOLA” – Arabidopsis GL3 induces a dense coating of trichomes on Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More seedlings. Plant Mol. Biol., 60, 679-698.[↩]
- Soroka, J.J., Holowachuk, J.M., Gruber, M.Y., & Grenkow, L.F. 2011. Feeding by flea beetles (Phyllotreta spp.) (Coleoptera: Chrysomelidae) is decreased on canola (Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More) seedlings with increased trichome density. J. Econ. Entomol., 104, 125-136.[↩]