Bertha armyworm is a significant insect pests of canola in Canada and some years can cause severe infestations. Properly identifying the insect and potential natural enemies in a field is necessary before considering economic thresholds and making a final management decision.
Important tips for best management
- An early-maturing canola crop (achieved through seeding date, varietyA variety is a variant of a species that evolved in nature without the intervention of humans, e.g. Brassica oleracea variety (in short form, var.) botrytis (cauliflower), var. capitata (cabbage), var. italica (broccoli), etc. More selection, etc.) can help avoid a significant bertha armyworm infestation unless the adult moth flight is exceptionally early.
- Use provincial monitoring programs beginning in June to help identify regions at higher risk for potential larval outbreaks, based on elevated numbers of adult moths caught in pheromone traps.
- Begin scouting for larvae feeding on the crop after peak flowering or about two weeks after peak trap catches of adult moths. Monitor larval numbers in each canola field as densities can vary between fields.
- It is important to identify early defoliation in canola and distinguish the cause. Small bertha armyworm likely do not require control until they are past the full size of a diamondback moth larvae (which they may be confused with). Determine the average number of larvae per square metre, then consult the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More chart, which considers application costs and canola prices.
- Under drought stress, early leaf drop may increase the intensity of feeding on pods. Dividing the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More values in the chart by 1.48 may be more appropriate in these conditions.
- Apply the insecticide early in the morning or late evening when the larvae are actively feeding.
- Control larvae when they are at least 1.3 centimetres (0.5 of an inch) long. Newly hatched larvae are especially vulnerable to inclement weather and diseases, sometimes controlling them without insecticide. Also, it is not until the last two larval stages that the larvae eat about 80 to 90 per cent of the total plant material consumed during their life.
- Natural enemies often peak after a pest insect outbreak, and may help to reduce the longevity of outbreaks. Use economic thresholds to preserve natural enemy populations.
Overview
Bertha armyworm (Mamestra Configurata Wlk.) is one of the most significant insect pests of canola in Canada. It occurs throughout Manitoba, Saskatchewan, Alberta and the interior of British Columbia. Severe infestations can occur throughout most of this area but are usually limited to the parkland area of the prairies and the Peace River region of British Columbia and Alberta.
Background
Bertha armyworm is native to North America and belongs to a group of insects called “climbing cutworms.” The true armyworm and variegated cutworm are also in this group.
In most years, populations are kept low by unfavourable weather conditions such as cold winters and cool wet weather, and by parasites, predators and diseases. But when these natural regulators fail, populations can increase dramatically, creating the potential for widespread damage to a varietyA variety is a variant of a species that evolved in nature without the intervention of humans, e.g. Brassica oleracea variety (in short form, var.) botrytis (cauliflower), var. capitata (cabbage), var. italica (broccoli), etc. More of broadleaf crops. In extreme situations, infestations of more than 1,000 larvae per square metre (1,200 per square yard) have been reported while densities of 50 to 200 larvae per square metre (60 to 24 per square yard) may be common. Infestations may be localized or spread over millions of acres.
Bertha armyworm feed on a varietyA variety is a variant of a species that evolved in nature without the intervention of humans, e.g. Brassica oleracea variety (in short form, var.) botrytis (cauliflower), var. capitata (cabbage), var. italica (broccoli), etc. More of crops and weeds. Canola, rapeseed, mustard, alfalfa, lamb’s quarters and related plants are preferred host plants. Bertha will also feed on a range of secondary hosts including flax, peas, potato, quinoa and numerous weeds.
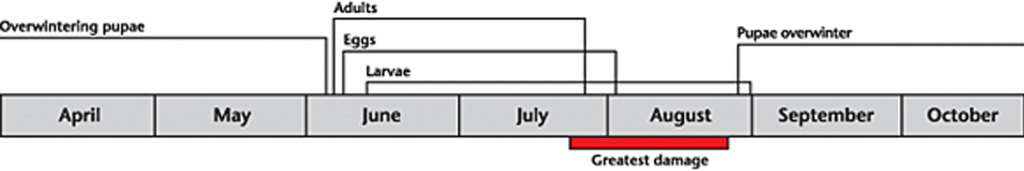
Life cycle
There is one generation of bertha armyworm per year. Adult moths emerge from overwintering pupae in mid-June and emergence continues until late July. The moth has a wingspan of about four centimetres (1.6 inches) and is active only at night. The forewing is predominantly gray, and flecked with patches of black, brown, olive and white scales. Near the middle of the forewing, toward the leading wing margin (front), there is a prominent, white, kidney-shaped marking defined with a ring of whitish scales. Near the tip of the forewing, there is a conspicuous white and olive coloured, irregular transverse marking that is characteristic of the species.
Moths appear to be strongly attracted to canola fields that are in bloom and secreting nectar.
Adult moths mate within five days of emergence and lay their eggs on the undersides of canola leaves soon after emergence. The eggs are laid in single-layer clusters of 50 to 500, in a honeycomb arrangement. Each female moth will lay about 2,150 eggs but as many as 3,500 eggs per female have been recorded. The eggs are sculptured, ridged and pinhead in size. When first laid, they are white but become darker as they develop. At average temperatures, the eggs hatch within a week.
Newly hatched larvae are about 0.3 centimetres (0.1 of an inch) long. They are pale green with a pale yellowish stripe along each side. While they look similar, it is useful to distinguish between small bertha armyworm larvae and diamondback moth larvae. Due to their size and colour, they are difficult to see on the undersides of leaves. When disturbed, small larvae may drop off the leaves by a fine silken thread. This behaviour makes it difficult to distinguish small bertha armyworm larvae from those of the diamondback moth, which display a similar behaviour. Large larvae drop off the plants and curl up when disturbed, a defensive behaviour typical of cutworms and armyworms.
Depending on the temperature, larvae take approximately six weeks to complete their development. During this period, they moult five times and pass through six growth stages. As they mature their colour becomes variable. Some remain green, but may become brown or velvety black. At maturity, the larvae are about four centimetres (1.6 inches) long, with a light brown head and a broad, yellowish-orange stripe along each side. The velvety black larvae have three narrow, broken white lines on their backs1.
Note that the diamondback larvae may complete their lifecycle in as little as three weeks, so the presence of adult diamondback moth and pupae should allow further identification.
Always watch for leaf defoliation from early larvae feeding and begin scouting to assess if larval numbers exceed the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More after peak flowering or about two weeks after peak trap catches of adult moths.
At maturity in late summer or early fall, larvae burrow into the ground and form pupae. Pupation usually begins in mid- to late August and continues through early to mid-September. If autumn is unusually warm, some pupae may continue their development and emerge as moths in late August or September, only to perish when winter arrives. Bertha armyworm overwinters as pupae in the ground at depths of 5 to 16 centimetres (two to 6.3 inches). The pupa is a reddish brown pod-like structure about 0.5 to 1.8 centimetres (0.2 to 0.7 of an inch) in size and tapered with flexible, terminal abdominal segments. The pupa protects bertha armyworm while it transforms from the larval stage to the adult moth. Bertha armyworm pupae are indistinguishable from other cutworm pupae.
Influence of Environment
Environmental conditions have a significant impact on bertha armyworm populations, especially on the overwintering pupae. During harsh winters in snow-free fields, most bertha armyworm pupae die. Bertha armyworm outbreaks appear to be favoured by snow accumulation, which protects pupae from prolonged exposure to temperatures below -10 degrees Celsius.
Newly hatched larvae are especially vulnerable to inclement weather and diseases.
The increase in reduced tillage and stubble conservation results in more snow accumulation on infested fields and could favour bertha armyworm survival, especially in years with early snowfall.
During hot, dry weather, the young larvae may be found feeding on the lower leaves or hiding under debris at the soil surface. The older larvae tend to feed both during the day and night. Larvae migrate from an infested field only when the food supply is short or when the crop is over-ripe.
Identification
Scouting techniques
The number of bertha armyworm larvae in a crop one year is not a reliable indicator of what to expect the following year. Bertha armyworm populations fluctuate widely between years.
Forecasting tools: Provincial monitoring programs starting in June raise awareness of potential outbreaks, based on number of adult moths caught in pheromone traps.
When: Check for larvae in each field regularly to minimize crop losses. Watch for leaf feeding and begin larval monitoring after peak flowering or about two weeks after peak trap catches. Continue scouting until either the mean number of larvae per square metre exceeds the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More (at which point the crop is sprayed) or until the time remaining until the crop is swathed no longer allows for application of a registered insecticide based on the allowed pre-harvest interval.
Where: For accurate larval estimates in a crop, sample at least three locations, a minimum of 50 metre (164 feet) apart. Do not sample headlands (20 metres or 66 feet wide) and areas within the crop that are not representative of the field.
How: At each location, mark out an area of one square metre and beat the plants growing within that area to dislodge the larvae. Push the plants aside, and pick up leaf litter to count the number of larvae in the square metre. It is important to wait for the disturbed larvae to begin moving again to allow for easier visibility. Carefully search the soil and leaf litter. The larvae are difficult to see and may be hidden underneath clumps of soil, in cracks in the soil or within curled leaves. Use the average number of larvae at the sites surveyed within each field to determine if the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More has been exceeded and an insecticide application is necessary.
Action threshold: Consult the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More chart, which considers application costs and canola prices.

Check out more information and provincial real-time survey maps of bertha armyworm from Alberta Agriculture and Forestry, Government of Saskatchewan and Manitoba Agriculture. The monitoring programs use accumulated degree-days to monitor the development of overwintering pupae into moths. Pheromone-baited traps that attract the male moths monitor the flight of bertha armyworm moths. The number of moths collected by these traps gives an indication of the risk of bertha armyworm larval infestations. Generally, higher moth numbers during the flight period (around mid-June through July) indicate greater risk of larval damage (in July and August). Risk assessment maps are available from crop specialists and on provincial websites. Manitoba Agriculture clearly describes the protocol for monitoring bertha armyworm with pheromone-baited traps.
Interpreting cumulative bertha armyworm moth counts
Cumulative moth counts are available on the provincial agricultural websites, and some sites show cumulative counts at each monitoring site. Each week moths caught in the traps are counted, and data for that week, as well as cumulative moth counts, are entered into the table for the appropriate agricultural region. The cumulative total number of moths will continue to rise until the end of the monitoring period. The following table relates the cumulative moth counts with the risk of larval infestation.
Monitor larval numbers in each field is critical. Adjacent fields may have very different larval densities, depending on how attractive the crop was when the moths were laying their eggs. Adjacent fields may also have different-sized larvae, depending on when the eggs were laid.
Damaging stage
The only development stage of bertha armyworm to cause crop damage is the larval stage.
The greatest amount of damage occurs as the larvae grow from about 1.3 centimetres to four centimetres (0.5 to two inches) in their last two larval instars. Larvae in the fifth and sixth instarA developmental stage within one life stage (ex. of an insect) More stage eat about 80 to 90 per cent of the plant material consumed during the life of the larvae2.
Type of crop damage
Larvae eat leaves, stems and pods. The degree of damage varies with the crop, the plant’s growth stage, the growth stage of the larvae and the number of larvae present. Depending on the season and crop location, significant crop damage usually occurs within a three-week period between late July and late August. A larval population of 200 per square metre (187 per square yard) can reduce yields by 50 per cent. The larvae are not highly mobile and migrate from an infested field only when the food supply is short, or the field is over-ripe.
Small larvae feed on the underside of leaves, chewing irregularly shaped holes in the leaves. They usually cause little damage at this stage, even when population levels are high.
If the plants drop their leaves before the larvae are mature, the developing larvae will feed directly on stems and pods. From a distance, infested canola fields look pale white because larvae eat the outer green layer of the stems and pods exposing underlying white tissue. Pods may be “debarked,” but more commonly, the larvae chew holes in the pods and eat the seeds. At high numbers, the entire pod may be consumed. Even if the pods are only stripped of their outer green layer and not eaten entirely, crop losses still may occur because of premature shattering. Swathing is not considered a control measure, as larvae can continue to feed for a few days until the crop dries in the swath. In some years, early-seeded canola can be swathed prior to damage.
Management
Natural enemies and biological control
In Canada and the United States, several insect parasitoids of bertha armyworm have been identified3:
- Tachinidae (Diptera) – 12 species
- Braconidae (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More) – three species
- Ichneumonidae (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More) – three species
- Hyperparasitic Ichneumonidae – four species
- Eulophidae (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More) – two species
- Trichogrammatidae (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More) – one species
In Canada, an ichneumonid wasp (Banchus flavescens) and a tachinid fly (Athrycia cinerea) are the only effective native parasites of bertha armyworm. B. flavescens lays its eggs in the first to third larval instars, and kills mature larvae after they enter the soil to pupate4.
A. cinerea lays eggs on the third to sixth instarA developmental stage within one life stage (ex. of an insect) More larvae and the emerging parasitoid larvae immediately burrow into the bertha armyworm larvae5.
A nuclear polyhedrosis virus is also known to attack bertha armyworm.
These natural enemies often do not destroy larvae until after considerable crop damage has occurred. They have their greatest impact on bertha armyworms produced a year or two after the peak of an outbreak. This is probably why severe infestations only last two or three years. Their presence in a crop does not, however, indicate that control measures are unwarranted.
Commercial formulations of Bacillus thuringiensisA bacteria which can be used as a biological pesticide (insecticide) More (Bt or BtkA bacteria which can be used as a biological pesticide (insecticide) More) varieties have not proven highly effective against bertha armyworm.
Field management (cultural control)
Canola losses from bertha armyworm populations can be minimized by:
- planting alternative (non-host) crops
- effective weed control
- early swathing
- fall cultivation
An early-maturing crop (achieved through planting date, varietyA variety is a variant of a species that evolved in nature without the intervention of humans, e.g. Brassica oleracea variety (in short form, var.) botrytis (cauliflower), var. capitata (cabbage), var. italica (broccoli), etc. More selection, etc.) can help avoid a significant bertha armyworm infestation unless the moth flight is exceptionally early. Canola harvested before August 15 is less likely to suffer yield loss from bertha armyworm. Mated female moths prefer to lay eggs on canola in the early flowering stage. Fields in this stage during the egg-laying period tend to be hardest hit.
Fall cultivation can kill some bertha armyworm pupae by mechanical damage. Tillage can also reduce the amount of snow trapped on a field by removing or flattening stubble and exposing pupae to sub-zero temperatures over the winter. This practice may be effective for individual fields but is not likely effective unless all growers in an area adopt the practice. Adult moths are strong flyers and can easily move to adjacent fields. Do not use fall cultivation on soils susceptible to erosion.
Effective control of weeds such as lamb’s-quarters and wild mustard can reduce bertha armyworm infestations in flax, peas, lentils and sugar beets. Egg-laying may occur on weeds attractive to the adult female in any crop and larvae will first feed upon these weeds and then move onto the crop after the weeds have been destroyed.
Resistance breeding

Feeding trials research found that B. junceaAlso referred to as brown mustard, it is a minor crop (from the Cruciferae or Brassicaceae plant family, commonly known as the mustard family) grown in Canada. More (brown mustard) and S. alba (white mustard) were the poorest host plants for bertha armyworm larvae, and that specific foliar glucosinolates, such as sinigrin, may provide crucifer crops with some protection from bertha armyworm feeding6. First instars of bertha armyworm did not survive on leaves of T. arvense (field pennycress). Although 97 per cent of larvae survived to the sixth instarA developmental stage within one life stage (ex. of an insect) More on M. sativa (alfalfa) complete mortality was observed on this host plant before pupation. Survival rates to sixth instarA developmental stage within one life stage (ex. of an insect) More and pupation were similar for B. rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More, B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More and B. junceaAlso referred to as brown mustard, it is a minor crop (from the Cruciferae or Brassicaceae plant family, commonly known as the mustard family) grown in Canada. More7.
White mustard is a preferred host for egg-laying despite being relatively resistant to larval feeding. B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More is preferred over B. junceaAlso referred to as brown mustard, it is a minor crop (from the Cruciferae or Brassicaceae plant family, commonly known as the mustard family) grown in Canada. More for egg-laying. Full flower stage of canola is preferred for egg-laying over plants in pre-flower or pod stages6.
Economic threshold
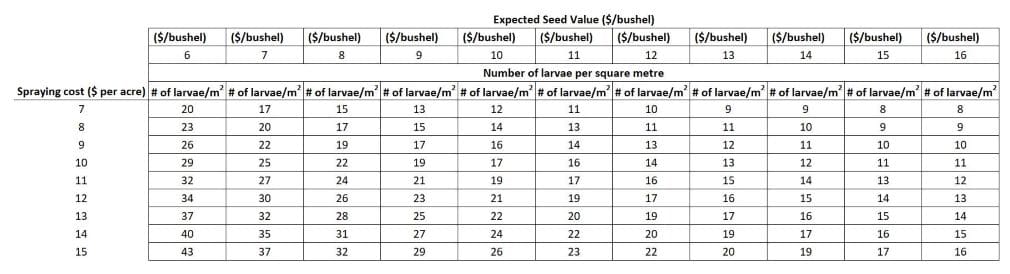
The economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More is the density of larvae where the economic value of the yield lost due to feeding equals the cost of control. If larval density exceeds this value then spraying is warranted to avoid economic losses. The economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More for bertha armyworm will vary with the cost of the insecticide, the method of application and the crop’s value. Using crop values and application costs, the following table indicates the larval density (larvae per square metre) at which an insecticide treatment in canola would be warranted.
For example, spraying would not be economical if larval counts were less than 16 per square metre given a crop price of $485.10 per tonne ($11.00 per bushel) and a spray cost of $24.70 per hectare ($10.00 per acre).
Table. 1 The bertha armyworm larval density at which an insecticide treatment in canola would be warranted (depending on expected seed value and spraying costs)
Source: Manitoba Agriculture
Under drought conditions, where bertha armyworm feeding is concentrated on canola pods by early leaf drop, economic thresholds may be lower than indicated. Under drought stress, dividing the economic thresholds above by 1.48 may give more appropriate values (e.g. above the threshold would convert to 11 larvae per square metre rather than 16).
Chemical control options
An insecticide is the grower’s last line of defense against bertha armyworm. With severe infestations widespread crop losses can be minimized with insecticides if the infestation is detected early. However, failure to detect infestations early may result in insufficient time to apply insecticides before severe damage is done. Also, there may be temporary insecticide shortages if suppliers are not aware of the potential outbreak. Insecticides cannot be applied to canola if it will be cut by either swathing or combining prior to the allowed pre-harvest interval (PHI) for the product. Products with short pre-harvest intervals may require a higher rate and additional cost to control large bertha armyworm larvae.
For best results, apply an insecticide as soon as economic thresholds are reached. A single well-timed application of any registered insecticide is usually effective.
- Ensure the larvae are at least 1.3 centimetres (0.5 of an inch) long (before the fifth instarA developmental stage within one life stage (ex. of an insect) More larvae appear).
- Apply the insecticide early in the morning or late evening when the larvae are actively feeding. Do not apply during warm afternoons. A single, well-timed application of any registered insecticide applied with aerial or high clearance ground equipment is usually effective.
- Use enough water to ensure adequate coverage.
- Use high water volumes in crops with dense canopies.
- Use the higher label rates of application when a range is indicated.
To protect foraging honeybees, delay insecticide applications until after the crop has finished blooming. If this is not possible, select the safest insecticide to control the bertha armyworm larvae and apply during the evening.
Check your provincial crop protection guide for up to date listings of control products and registrations.
Table 2. Insecticides registered for bertha armyworm in canola
1 LD50 values represent the relative toxicity of a pesticide. They represent the dose (in mg/kg body weight) that will kill 50% of the test animals. Thus the lower the number the greater the toxicity. Values given are for oral female rat LD50.
*Talk to your grain buyer prior to application
Note – Always check your provincial crop protection guide for current listings of registered control products, and always read and follow current pesticide label directions.
Source: Alberta Crop Protection Guide 2023 (The Blue Book)
- Philip, H., Mori, B.A., & Floate, K.D. 2018. Field crop and forage pests and their natural enemies in Western Canada: Identification and management field guide. Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More, Saskatoon, SK. [↩]
- Philip, H., & E. Mengersen. 1989. Insect Pests of the Prairies. University of Alberta Press, Edmonton, AB. 122. [↩]
- North American Dipterists Society [↩]
- Arthur, A.P., & Mason, P.G. 1985. The Life History and Immature Stages of Banchus flavescens. Can. Entomol., 117, 1249-1255. [↩]
- Wylie, H.G. 1977. Preventing and terminating pupal diapause in athrycia cinerea (DIPTERA:TACHINIDAE). Can. Entomol., 109, 1083-1090. [↩]
- Ulmer, B., Gillott, C., & Erlandson, M. 2002. Oviposition Preferences of Bertha Armyworm Mamestra configurata Wlaker (Lepidoptera: Noctuidae) on Different Crucifer CultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and Growth Stages. Environmental entomology, 31, 1135-1141. [↩] [↩]
- Dosdall, L.M., & Ulmer, B.J. 2004. Feeding, development, and oviposition of bertha armyworm (Lefpidoptera: Noctuidae) on different host plant species. Environmental entomology, 33, 746-764. [↩]