A common pest insect in canola, the diamondback moth adults blow in from the southern United States each spring with their larval offspring – and subsequent generations – can cause varying amounts of feeding damage on green canola tissues throughout the growing season. There are relatively good monitoring options for this insect, from wind trajectory models, to pheromone traps in addition to in field scouting. In combination with economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More considerations, there are good tools for making management decisions.
Important tips for best management

- Adults that arrive in the spring mean there will be multi-generations and a higher risk of economic damage. Be aware of provincial agricultural and Prairie Pest Monitoring Network early warnings.
- Pheromone traps detect arrival of moths in the spring but are NOT to be used for economic thresholds.
- July and August is the main time to scout fields for potential damage and/or larvae. Increasing scouting if notable insect feeding is observed.
- When scouting: remove plants in an 0.1 square metre area (about 12 square inches), then vigorously shake the plants onto a clean surface and count the number of larvae dislodged from the plants.
- Use economic thresholds, which vary by the plant stage, as thresholds ensure only warranted insecticide applications are made. This minimizes the impact on beneficial insects. An insecticide application may be considered:
- if larvae exceed 100-150 per square metre (10-15 larvae per square foot) on immature to flowering plants.
- there are 200-300 larvae per square metre (20-30 larvae per square foot) at the late flowering or pod stage.
- at seedling stage when there has been 25-33 per cent defoliation and larvae are still present on the plant.
- Parasitoids and other biological controls often collapse infestations of diamondback moth prior to economic damage.
- Control brassicaceousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More weeds including volunteer canola (which are host plants for the insect)
- Check out canola production-related research on diamondback moths in the Canola Research Hub.
Diamondback moth overview

Diamondback moth (Plutella xylostella L.), was introduced into North America from Europe about 150 years ago. It now lives throughout North America wherever host brassicaceousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More plants (ex. canola, mustard and related vegetables and weeds) are grown. In most years the insect causes minor economic damage on canola or mustard crops, but in some years populations reach outbreak densities and substantial crop losses can occur.
Background on the diamondback moth
Although diamondback moths appear each year throughout the Canadian Prairies and north central United States, the severity of the infestation varies considerably from between years and by location.
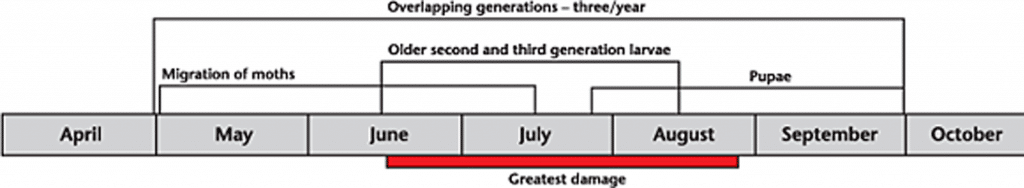
The insect may overwinter in the Prairies in some cases, but this doesn’t happen frequently or in large numbers. Instead, most spring populations in the Canadian prairies are due to new diamondback moths migrating northward from infested regions in the southern or western United States or northern Mexico on wind currents. The composition and timing of influxes has immense bearing on the damage caused by diamondback moth, since the species is multivoltineRefers to a species that has offspring more than once per year (more than one gerenation in one year). More. It is capable of producing as many as four generations per year in the Prairies, and early arrival times can therefore result in greater population build-up than later arrivals.
Diamondback moth is restricted in its host range to plants of the family Brassicaceae 1,2.
In addition to many other brassicaceousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More crops and weeds, host plants for the diamondback moth include:
- canola
- mustard
- cabbage
- cauliflower
- broccoli
- kale
On a worldwide scale, diamondback moth has been considered responsible for over one billion dollars (USD) in annual economic losses 3.
In western Canada, crop losses vary considerably from year to year. In some years the population densities and economic importance of diamondback moth can be negligible, but in other years the pest can cause many millions of dollars in lost revenue 4,5.
Life cycle of diamondback moth
In some years the moths can reach areas of the Canadian canola growing region even before many of the canola crops have emerged. In these situations brassicaceousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More (cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More) weeds and volunteer canola become important alternate hosts for laying eggs. Therefore controlling these weeds (including volunteer canola) will help reduce the chance of diamondback moths populations in canola fields.

Normally, diamondback moths take about 32 days to develop from egg to adult. However, the time to complete a generation may vary from 21 to 51 days depending on weather and food conditions. There may be several generations per growing season and generations usually overlap and all four life stages (egg, larva, pupa and adult). Therefore many life stages may be present in the field at the same time.
Adult females lay an average of 160 eggs during their life span of about 16 days. Egg-laying occurs at night. The largest batch of eggs is laid the first night after adult moths emerge from their pupae and egg-laying continues for about 10 days.
Where does the diamondback moth lay its eggs?
Diamondback moth eggs are laid mainly on upper leaf surfaces 6 and hatch in four to eight days. Eggs are oval, yellowish-white and tiny. They are glued to the upper and lower leaf surfaces individually or in groups of two or three, usually along the veins or where the leaf surface is uneven.
First-instar larvae bore through the leaf epidermisThin outer layer of a tissue (such as the thin outer later of plant tissue which peels back from canola stems which are infected with verticillium stripe). More and feed internally on cells of the leaf mesophyll 7. The three subsequent larval instars are surface feeders, and consume leaves, buds, flowers, or pods. An average duration of 3.4 to 4.2 days are required for completion of the first to fourth larval instars, respectively, and 7.8 to 9.8 days were required for pupation under field conditions in Ontario 7. The larval stage lasts about 10 to 21 days, depending upon temperature and the availability of food.
Larvae pupate in delicate, white, open-mesh cocoons attached to the leaves, stems or pods of the host plant. Initially, the pupae are light green but as they mature they become brown and the adult moth becomes visible through the cocoon. The pupal stage lasts from five to 15 days depending on environmental conditions, and then adult moths of the next generation emerge.
Influence of environment
An infestation of diamondback moth cannot be predicted based on the previous year’s population because very few, if any, pupae survive the long, cold Canadian winters. Still, one central Alberta study using trap cages placed in a field during early spring, found diamondback moth was able to overwinter 8. However, the heavy, insulating snowfall early the previous fall was thought to help explain these observations. Other attempts to overwinter diamondback moth in the Canadian Prairies have been unsuccessful regardless of location (ex. Saskatoon, SK or Vegreville, AB), soil organic residue levels, or the insect life stage used 9.
Instead, the severity of the infestation in any given year primarily depends on two factors: populations overwintering in the United States and strong south spring winds to transport the moths north into Manitoba, central Saskatchewan and eastern Alberta. High densities of diamondback moth on the Prairies have been traced back to strong airflow from the southern United States 9,10, and in some cases, possibly California and the Pacific northwest of the United States 10.
The Prairie Pest Monitoring Network utilizes wind trajectory-modelling prior to and during the growing season to provide an early-warning system for agronomists and farmers. It integrates a network of sentinel sites with pheromone traps to deliver advanced notice for the potential arrival of diamondback moth populations into canola production areas of the prairies 11.
Environmental factors can also have a profound (negative) impact on diamondback moth populations (after they arrive in Canada). Cool, windy weather reduces adult activity and females often die before they lay all their eggs. Heavy rainfall can drown small larvae and reduce numbers by more than half. The humid conditions that occur within the crop following a rainfall can promote the spread of fatal fungal diseases throughout the diamondback moth population.
Identification
The adult moth is approximately eight to nine millimetres (0.3 to 0.4 inch) long with a wingspan of 12 to 15 millimetres (0.5 to 0.6 inch). At rest, the moth folds its wings over the abdomen in a tent-like manner. The folded wings flare upwards and outward at the tips. The wing tips are fringed with long hairs. In the male, the forewing margins have a series of creamy-coloured wavy markings. When the wings are folded while the moth is at rest, these markings come together to form three light coloured diamonds, hence the name diamondback.
The larvae (caterpillars) are a pale yellowish-green to green colour and are covered with fine, scattered, erect hairs. The posterior end of the caterpillar is forked. At maturity the larvae are cigar-shaped and about 12 millimetres (0.5 inch) long. The diamondback moth larva is easily identified by its peculiar reaction to being disturbed. It will wriggle backward violently and may drop from the plant, suspended by a silken thread. After several seconds, the larva will climb back onto the leaf and continue feeding.
Diamondback moth scouting techniques
In the field, crop scouts may observe moths fluttering up when the crop canopy is disturbed. However, it is the larval feeding that will lead to yield losses, and environmental conditions will ultimately determine how many eggs are laid and whether the larvae emerge and survive, leading to economical feeding damage. Monitor plants for the characteristic “window paning” evidence (when only the green material leaf material is eaten and the clear, upper cuticle membrane is left behind, resembling a window pane), and any other insect feeding indications.
Consult with crop specialists and entomologists for the size and timing of the moth flight, based on trajectory modeling and pheromone trapping of adult. Also utilize the Prairie Pest Monitoring Network’s wind trajectory and pheromone trap network reports. These pheromone traps cannot predict the potential for crop damage, but trap counts can provide an early warning of a possible infestation. If these trap counts correlate well with the wind trajectory models it provides an even stronger indication of regions at greatest risk.
Check out more information and provincial real-time survey maps of diamondback moths from Alberta Agriculture and Forestry, Government of Saskatchewan and Manitoba Agriculture. Further information on Manitoba’s diamondback moth forecast can be found here.
Sweep net sampling can determine the presence and general abundance of the species in the field, but does not provide a good estimate of larval density because no correlative studies have been conducted to relate sweep net captures with density in the field (under local conditions). Therefore no thresholds exists for sweep net counts. Nevertheless, high population counts in sweep sampling can prompt producers to perform more accurate counts of densities per unit area.
The most accurate method of estimating diamondback moth population density in canola is to perform counts of diamondback moth larvae in several locations throughout the field and determine the average population per unit area.
Scout fields throughout season, especially in July and August. Monitor crops at least twice per week if insect numbers or feeding damage indicates the larvae are approaching economic thresholds. Sample at least five locations per field to get a representative sample. To collect a proper sample, remove plants in an 0.1 square metre (about 12 square inches) area, then vigorously shake the plants onto a clean surface and count the number of larvae dislodged from the plants.
See the Field Crop and Forage Pests guide and PPMN monitoring protocol for more information about scouting for diamondback moth.
If diamondback moths are identified, an insecticide application may be considered if the populations exceed economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More levels (which are different depending on the crop growth stage) 12. Note that these economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More recommendations are estimated or nominal, without basis in replicated experimental studies. Control may be required if:
- There is 25-33 per cent defoliation at the seedling stage and larvae are still present on the plant.
- Larvae populations exceed 100-150 per square metre (10-15 per square foot) on plants between the vegetative to the flowering stage.
- Larvae exceed 200-300 per square metre (20-30 per square foot) on plants between the flowering to podding stage. 13
Life cycle stage causing damage
Crop damage is caused by the larval stage. Diamondback moth larvae feed on any green tissue of host (ex. canola and mustard) plants but prefer leaves. The amount of damage varies greatly, depending on plant growth stage, larval density and larval size.
Type of crop damage
Damage as early as the first true-leaf stage can be caused by early diamondback moth infestations. In most instances, however, moth invasions of western Canadian canola and mustard crops occur later, when crops are in the rosette stage. Arriving in mid-May can allow the moths enough time to complete three or four generations by the time crops are in the pod development stages, and most vulnerable to attack 14.
When larvae are small, damage is evident as small irregular “shot” holes in the leaves. Due to the small size of the larvae, and its mouthparts, early feeding can cause the characteristic “window paning” effect, where only the green material leaf material is eaten and the clear, upper cuticle membrane is left behind (resembling a window pane).
If larvae are numerous, they may eat the entire leaf, leaving only the veins. Larval feeding damage to canola leaves is usually considered to have a minor effect on yield, but larval feeding on buds and flowers can be more damaging, especially when plants are under abiotic stress (such as drought) and cannot compensate by producing new buds and flowers. Feeding damage during the early flowering stage can be extensive, and if so, will delay plant maturity, causing the crop to develop unevenly and significantly reduce seed yields. When plants are fully podded and leaves begin to wilt, drop, die or were previously eaten in late July or early August, larvae will move up and remove the surface tissue from the stems and pods. The seeds within a damaged pod will not fill completely and pods may shatter, resulting in yield loss. Larvae may also chew into pods and eat the developing seeds. Yield loss can be extreme in prairie fields with high infestations.
Crop damage is usually first evident on plants growing on the ridges and knolls in fields. In severe cases, damage shows from a distance as abnormal whitening. However, Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More, B. rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More, and Sinapis alba can respond to larval feeding by increasing their root biomass, presumably as a strategy to enable them to compensate for foliage loss, through the uptake of greater quantities of soil water and nutrients 15. Although the magnitude of root mass increase is varied by varietyA variety is a variant of a species that evolved in nature without the intervention of humans, e.g. Brassica oleracea variety (in short form, var.) botrytis (cauliflower), var. capitata (cabbage), var. italica (broccoli), etc. More and species.
The greatest increase in B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More root biomass occurred to plants treated with intermediate levels of fertilizer (i.e. the rate recommended for canola production) rather than low or high fertilizer applications 16.
Management | How to manage diamondback moths
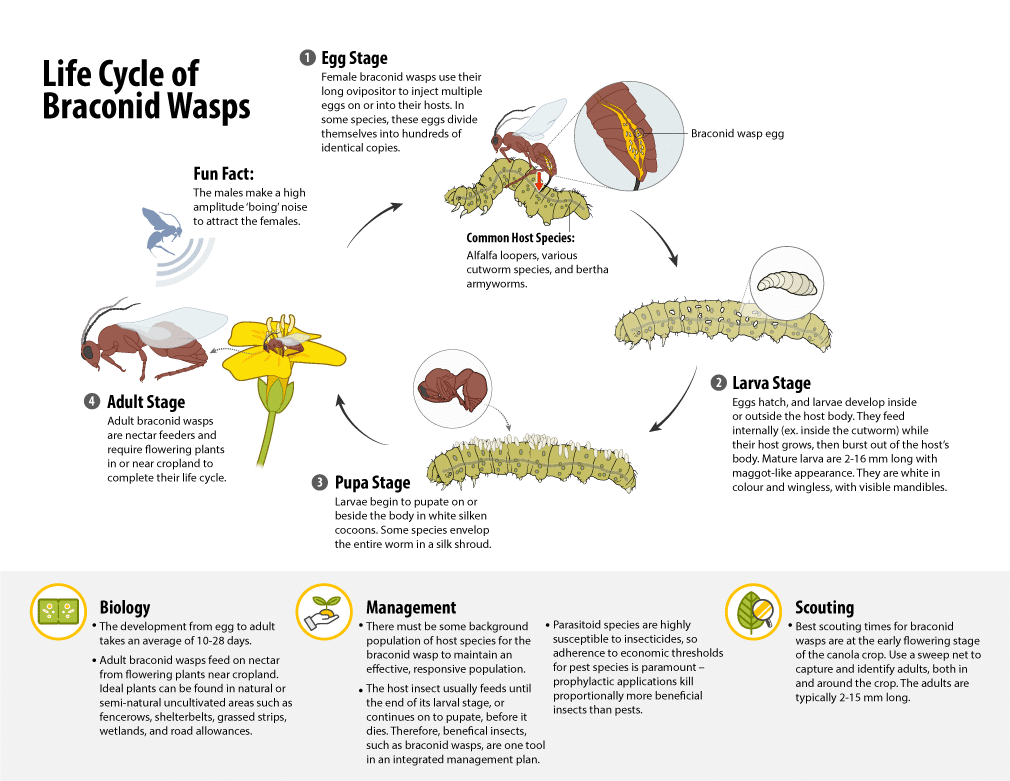
Natural enemies (including parasitoids, predators and fungi) of diamondback moth are important for biological control.
There are three main parasitoids of diamondback moths found in the western Canadian Prairies which belong to two wasp families (from the order HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More). The species Diadegma insulare (Cresson) and Diadromus subtilicornis (Gravenhorst) are members of the family Ichneumonidae, and the species Microplitis plutellae (Muesbeck) is from the family Braconidae 17,18. D. insulare and M. plutellae attack diamondback moth larvae and D. subtilicornis parasitizes diamondback moth pupae 19,18.
D. insulare is considered the most important for biological control. In Alberta this species parasitized 45 per cent of diamondback moth individuals in 1992 while M. plutellae and D. subtilicornis were each only responsible for approximately 15 per cent parasitism. Similarly, in Saskatchewan D. insulare accounted for 30 per cent of diamondback moth parasitism, while D. subtilicornis and M. plutellae accounted for about 15 per cent parasitism combined 20.
D. insulare is native to the Neotropics 18,21, so it does not survive Prairie winters 22 and is believed to migrate northward in spring with its diamondback moth hosts. D. insulare can parasitize all four larval instars of diamondback moth and the parasitoid emerges as a final instarA developmental stage within one life stage (ex. of an insect) More when its hosts reach their pre-pupal stage. Adults require a nutrient source for survival, and as a result, they spend more time in habitats where flowering plants are abundant so they can feed 23. Flowering plants like alyssum, Lobularia maritime (L.) Desv. (Brassicaceae), can provide food sources for adults of D. insulare, and the parasitoid is attracted to plant tissue damaged by diamondback moth larvae 24. This appears to explain why field populations of D. insulare can be clustered, often in patches correlated with high populations of diamondback moth 25,26.
M. plutellae has a widespread distribution in North America 17,18, overwinters in western Canada, and can be present early in the season to parasitize hosts 27. However, M. plutellae is generally less abundant than D. insulare in canola fields.
D. subtilicornis is a solitary pupal endoparasitoid of the diamondback moth, but very little is known about its biology in western Canada. Females lay eggs in pre-pupae or in pupae, but much more frequently in newly-formed pupae rather than the older ones. Females of D. subtilicornis can use their mouthparts to enlarge wounds made by the ovipositor during egg-laying, and may then feed on fluid oozing from the wound 28.
The contribution of predators to biological control of diamondback moth in the Prairies is unknown. Canola and mustard crops harbour a diverse fauna of predators, comprised mainly of various species of:
- carabid beetles (Coleoptera: Carabidae)
- lady beetles (Coleoptera: Coccinellidae)
- syrphid flies (Diptera: Syrphidae)
- lacewings (Neuroptera: Chrysopidae)
- rove beetles (Coleoptera: Staphylinidae)
- several families of spiders (Araneae)
With the exception of the carabid beetles 29,30,31, little research on this fauna has been undertaken. For example, research documented 59 species of carabids in canola from a single site in central Alberta over three years of collections 32. However, no studies have focused specifically on predation of diamondback moth in the Prairies.
Due to the importance of predatory insects for reducing populations of diamondback moth in other cropping systems 33, further study is warranted to resolve this important gap in knowledge of mortality factors that can impact diamondback moth populations in prairie agro-ecosystems.
Fungi from the order Entomophthorales cause natural disease outbreaks in diamondback populations. These outbreaks usually occur late in the growing season when populations are high. The rate of infection of diamondback moth larvae can be high enough to limit the development of additional generations late in the season.
Despite the abundance of potential biological control agents, the only effective way of controlling a diamondback moth outbreak once the population exceeds the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More is to apply an insecticide.
Biological control of diamondback moth
Foliar applications with formulations of bioinsecticide containing the delta-endotoxin of the bacterium Bacillus thuringiensisA bacteria which can be used as a biological pesticide (insecticide) More (subspecies kurstaki) are proven to be effective and less damaging to non-target organisms than chemical insecticides 34,35. A Bacillus thuringiensisA bacteria which can be used as a biological pesticide (insecticide) More (subspecies aizawai) product with the tradename XenTari, which is available in the horticultural market, has been registered for use on canola in Canada, but is not yet used in canola.
Field management (cultural control)
Preseed weed control and tillage reduces the availability of cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More weeds and volunteer canola host plants, preventing the successful establishment of first generation larvae where moths arrive before canola emergence.
Research has found that, for egg-laying, females selected plants that received quantities of soil fertilizer recommended for canola production, compared to unfertilized plants or plants that received three and five times the recommended levels of fertilizer. Similarly, larval survival was greatest and larval development was fastest on plants grown at intermediate levels of soil fertility 16.
Sulfur appears to have the greatest fertility influence on diamondback moth, with females laying significantly more eggs on sulfur-fertilized plants than on plants grown in soil deficient in sulfur 36. II. Sensory regulation of oviposition of the adult female. Entomol. Exp. Appl. 3: 305–314.)),37,25.
Resistance breeding
Diamondback moths prefer some species of Brassicaceae hosts over others. Egg-laying females have been found to prefer S. alba and B. rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More, and to have most rapid larval and pupal development times on B. junceaAlso referred to as brown mustard, it is a minor crop (from the Cruciferae or Brassicaceae plant family, commonly known as the mustard family) grown in Canada. More and S. alba. 15.
Development of diamondback moth can also be influenced by varieties within species. Although survival of diamondback moth did not vary for individuals reared from egg to pupa on the B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More varieties Q2, Liberty, and Conquest, females laid significantly more eggs on Liberty than on Q2 or Conquest 15. Developmental time of females from larva to prepupa tended to be faster on Liberty and Conquest than on Q2, but female body weight was greater for individuals reared as larvae on Q2 than on Liberty 15.
Canola transgenic for expression of the cry1Ac gene had been developed and provided protection from diamondback moth infestation 38, but no such transgenic crops were commercialized in Canada with this trait.
Chemical control for diamondback moth
Early field monitoring and the application of insecticides can prevent damage, if larval numbers exceed the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More. After an infestation is controlled at the podding stage, a new infestation is not likely to become established because of the rapid advance of the crop toward maturity.
See the WCCP Guidelines for the control of insect pests in oilseed crops in Western Canada 39. For registered insecticides and current recommendations check the annual provincial publication and always refer to product labels.
- Alberta Crop Protection Guide (The Blue Book)
- Saskatchewan Guide to Crop Protection
- Manitoba Guide to Field Crop Protection
A single, well-timed application of an insecticide with either aerial or ground equipment is usually effective in controlling larval populations. Make insecticide applications when larval populations are high because the effectiveness is reduced against adults or pupae. Always apply insecticides with enough water to ensure adequate coverage. Use high water volumes and label rates when the crop canopy is dense. If the leaves are beginning to turn yellow and dry up, damage will become more serious as larvae move to feed on pods. If this is the case, consider control at the lower end of the economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More range. For most effective control, apply at dusk or at night when diamondback moths are most active.
Injury to honeybees and other pollinating insects can be minimized by not spraying flowering crops. When it is necessary to apply an insecticide to a flowering crop, contact local beekeepers prior to application, use the safest product available and apply it during the evening.
Table 1. Insecticides registered for diamondback moth larvae control in canola
Note: LD50 values represent the relative toxicity of a pesticide. They represent the dose (in mg/kg body weight) that will kill 50 per cent of the test animals. Thus the lower the number the greater the toxicity. Values given are for oral LD50.Do not make more than 3 applications per season. Do not apply more than once every 5 days.
*Do not make more than 3 applications per season. Do not apply more than once every 5 days.
**Talk to your grain buyer prior to application.
† Do not apply deltamethrin at temperatures above 25°C.
‡ Do not apply malathion at air temperatures below 20°C.
Note: Always check your provincial crop protection guide for current listings of registered control products, and always read and follow current pesticide label directions.
Footnotes
- Sarfraz, M., Dosdall, L.M. and Keddie, B.A. 2010. Performance of the specialist herbivore Plutella xylostella (Lepidoptera: Plutellidae) on Brassicaceae and non-Brassicaceae species. Can. Entomol. 142:24-35.[↩]
- Sarfraz, M., Dosdall, L.M., Keddie, B.A. and Myers, J. 2011. Larval survival, host plant preferences and developmental responses of the diamondback moth Plutella xylostella (Lepidoptera: Plutellidae) on wild brassicaceousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More species. Entomol. Sci. 14: 20-30.[↩]
- Talekar, N. S., and Shelton, A.M. 1993. Biology, ecology, and management of the diamond back moth. Ann. Rev. Entomol. 38:275-301.[↩]
- Western Committee on Crop Pests. 1995. Minutes of the 34th Annual Meeting, October 20-21, 1995. Victoria, BC, Canada.[↩]
- Dosdall, L.M., Weiss, R.M., Olfert, O., Mason, P.G. and Soroka, J.J. 2008. Diamondback moth, Plutella xylostella (L.), as a pest of canola in Canada: Its historical impact on the crop and predicted effects of climate change on its pest status. Proceedings of the Fifth International Workshop on the Management of Diamondback Moth and Other Crucifer Pests, Beijing, China, October 24-27, 2006. 112-121[↩]
- Justus, K.A., Dosdall, L.M. and Mitchell, B.K. 2000. Oviposition by Plutella xylostella (Lepidoptera: Plutellidae) and effect of phylloplane waxiness. J. Econ. Ent. 93: 1152-1159.[↩]
- Harcourt, D.G. 1957. Biology of the diamondback moth, Plutella maculipennis (Curt.) (Lepidoptera: Plutellidae), in Eastern Ontario. II. Life-history, behaviour, and host relationship. Can. Entomol. 12: 554-564.[↩][↩]
- Dosdall, L.M. 1994. Evidence for successful overwintering of diamondback moth, Plutella xylostella (L.) (Lepidoptera: Plutellidae), in Alberta. Can. Entomol. 126: 183-185.[↩]
- Dosdall, L.M., Mason, P.G. Olfert, O., Kaminski, L. and Keddie, B.A. 2004. The origins of infestations of diamondback moth, Plutella xylostella (L.), in canola in western Canada. Proceedings of the Fourth International Workshop on the Management of Diamondback Moth and Other Crucifer Pests, Melbourne, Australia, November 26-29, 2001. 95-100.[↩][↩]
- Hopkinson, R.F., and Soroka, J.J. 2010. Air trajectory model applied to an in-depth diagnosis of potential diamondback moth infestations on the Canadian prairies. Agric. Forest Meteorol. 150: 1-11.[↩][↩]
- Prairie Pest Monitoring Network Blog. 2019. Wind Trajectory Reports. Retrieved from: https://prairiepest.ca/weekly-update/[↩]
- WCCP Guidelines for the control of insect pests. 2020.[↩]
- Manitoba Agriculture and Resource Development. 2020. Diamondback Moth. Retrieved from www.gov.mb.ca/agriculture/crops/insects/diamondback-moth.html[↩]
- Philip, H., and Mengersen, E. 1989. Insect Pests of the Prairies. University of Alberta Press, Edmonton, Alberta. 122.[↩]
- Sarfraz, M., Dosdall, L.M. and Keddie, B.A. 2007. Resistance of some cultivated Brassicaceae to infestations by Plutella xylostella (Lepidoptera: Plutellidae). J. Econ. Entomol. 100: 215-224.[↩][↩][↩][↩]
- Sarfraz, M., Dosdall, L.M. and Keddie, B.A. 2009. Bottom-up effects of host plant nutritional quality on Plutella xylostella (Lepidoptera: Plutellidae) and top-down effects of herbivore attack on plant compensatory ability. Eur. J. Entomol. 106: 583-594.[↩][↩]
- Braun, L., Olfert, O., Soroka, J., Mason, P. and Dosdall, L.M. 2004. Diamondback moth biocontrol activities in Canada. Proceedings of the International Symposium, October, 21-24, 2002, Montpellier, France. 144-146[↩][↩]
- Sarfraz, M., Keddie, B.A. and Dosdall, L.M. 2005. Biological control of the diamondback moth, Plutella xylostella (L.): A review. Biocontr. Sci. Technol. 15: 763-789.[↩][↩][↩][↩]
- Statistics Canada. 2007. Field Crop Reporting Series, Catalogue number 22-002-XIE.[↩]
- Braun, L., Olfert, O., Soroka, J., Mason, P. and Dosdall, L.M. 2004. Diamondback moth biocontrol activities in Canada. Proceedings of the International Symposium, October 21-24, 2002, Montpellier, France. 144-146[↩]
- Wagener, B., Reineke, A. Löhr, B., Zebitz, C.P.W. 2006. Phylogenetic study of Diadegma species (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Ichneumonidae) inferred from analysis of mitochondrial and nuclear DNA sequences. Biol. Control 37:131-140.[↩]
- Okine, J.S., Mitchell, E.R. and Hu, G.Y. 1996. Low temperature effect on viability of Diadegma insulare (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Ichneumonidae) and effect of this parasitoid on feeding rate of diamondback moth larvae (Lepidoptera: Plutellidae). Fla. Entomol. 7: 503-509.[↩]
- Idris, A.B., and Grafius, E. 2001. Evidence of Diadegma insulare (Cresson), a parasitoid of diamondback moth, Plutella xylostella (L.), present in various habitats. Pakistan J. Biol. Sci. 1:742-743.[↩]
- Mitchell, E.R, Hu, G.Y., Okine, J.S. and Carpenter, J.E. 1999. Parasitism of diamondback moth (Lepidoptera: Plutellidae) larvae by Cotesia plutellae (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Braconidae) and Diadegma insulare (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Ichneumonidae) in cabbage fields after inundative releases of C. plutellae. J. Entomol. Sci. 34: 101-112.[↩]
- Sarfraz, R.M., Dosdall, L.M., Blake, A.J. and Keddie, B.A. 2010. Leaf nutrient levels and the spatio-temporal distributions of Plutella xylostella and its larval parasitoids Diadegma insulare and Microplitus plutellae in canola. BioControl 55: 229-244.[↩][↩]
- Ulmer, B.J., Dosdall, L.M. and Gibson, G.A.P. 2005. Spatio-temporal distribution patterns of the diamondback moth and its principal parasitoid, Diadegma insulare, in canola crops and the occurrence of hyperparasitism. Brassica 6:63-69.[↩]
- Putnam, L.G. 1978. Diapause and cold hardiness in Microplitis plutellae, a parasite of the larvae of the diamondback moth. Can. J. Plant Sci. 58: 911-913.[↩]
- Tran, T., and Takasu, K. 2000. Host age selection by the host-feeding pupal parasitoid Diadromus subtilicornis (Gravenhorst) (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Ichneumonidae). Appl. Entomol. Zool. 35: 549-556.[↩]
- Cárcamo, H.A., and Spence, J.R. 1994. Crop type effects on the activity and distribution of ground beetles (Coleoptera: Carabidae). Environ. Entomol. 23: 684-692.[↩]
- Butts, R.A., Floate, K.D., David, M., Blackshaw, R.E. and Burnett, P.A. 2003. Influence of intercropping canola or pea with barley on assemblages of ground beetles (Coleoptera: Carabidae). Environ. Entomol. 32:535-541.[↩]
- Bourassa, S., Cárcamo, H.A., Larney, F.J. and Spence, J.R. 2008. Carabid assemblages (Coleoptera: Carabidae) in a rotation of three different crops in southern Alberta, Canada: A comparison of sustainable and conventional farming. Environ. Entomol. 37:1214-1223.[↩]
- Broatch, J.S. 2008. Root Maggot and Beneficial Insect Dynamics in Response to Weed Populations in Canola. Ph.D. Thesis, University of Alberta. 255.[↩]
- Furlong, M.J., Ju, K.H., Su, P.W., Chol, J.K., II, R.C. and Zalucki, M.P. 2008. Integration of endemic natural enemies and Bacillus thuringiensisA bacteria which can be used as a biological pesticide (insecticide) More to manage insect pests of Brassica crops in North Korea. Agric., Ecosys. and Environ. 125:223–238.[↩]
- Talekar, N. S., and Shelton, A.M. 1993. Biology, ecology, and management of the diamond back moth. Ann. Rev. Entomol. 38: 275-301.[↩]
- Kumar, K.P., and Gujar, G.T. 2005. Baseline susceptibility of the diamondback moth, Plutella xylostella (Linnaeus) to Bacillus thuringiensisA bacteria which can be used as a biological pesticide (insecticide) More Cry1A toxins in India. Crop Prot. 24: 207-212.[↩]
- Gupta, P.D., and Thorsteinson, A.J. 1960. Food plant relationship of diamondback moth (Plutella maculipennis (Curt.[↩]
- Marazzi, C., Patrian, B. and Städler, E. 2004. Secondary metabolites of the leaf surface affected by sulfur fertilization and perceived by the diamondback moth. Chemoecol. 14: 81–86.[↩]
- Ramachandran, S., Buntin, G. D., All, J.N., Raymer, P.L. and Stewart, C.N. 1998. Greenhouse and field evaluations of transgenic canola against diamondback moth, Plutella xylostella, and corn earworm, Helicoverpa zea. Agron. J. 92: 368-374.[↩]
- Western Committee on Crop Pests. 2021. Guide to Integrated Control of Insect Pests of Crops: Insect Management in Oilseed Crops in Western Canada. 2021. Retrieved from: https://westernforum.org/Documents/WCCP/WCCP_documents/WCCP_Guidelines/WCCP_20/Oilseeds-WCCP-2021-Final.pdf[↩]