Sclerotinia stem rot is one of the most economically significant canola diseases in Canada. This disease, caused by the fungus Sclerotinia sclerotiorum, is heavily influenced by environmental conditions leading up to and during the flowering period of canola, which can make predicting outbreaks difficult. Control of this disease can be achieved with risk assessment tools, fungicides, and partial resistance available in some canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More.
Important tips for best management

- Sclerotinia stem rot is a major disease in canola most years.
- In addition to canola, other crops can be hosts for this disease, including mustard, soybeans, sunflowers, pulses (ex. dry beans, field peas, lentils), potatoes and other vegetable crops.
- Several broadleaf weeds are also hosts and can maintain the fungus in the soil even in non-host crop years (e.g. stinkweed, sow thistles, Canada thistle, wild mustard, redroot pigweed, knapweeds, dandelions and lambs quarters).
- Use the sclerotinia stem rot risk assessment tool or checklist to determine if there is risk of this disease developing and if a fungicide application is warranted.
- Risk forecasting services, like petal tests and weather models, are not yet comprehensive, but when used with checklist provide better risk predictability.
- Sclerotinia fungicides are the best form of control when there is a high risk of disease development. Be sure to rotate fungicide groups.
- Use moderately sclerotinia resistant or tolerant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More to reduce risk, remembering these are currently not as effective as fungicides.
- Crop rotation (with non-host crops like cereals and grasses) can reduce sclerotinia stem rot in subsequent years.
- Factors impacting a microclimate that is conducive for the development of sclerotinia stem rot can be further reduced by:
- Using canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with low lodging scores.
- Using row spacing that is wider (but still able to support optimal weed control) or the lower end of recommended plant densities (ex. five or six plants per square foot).
- Avoid using more than recommended fertility rates to prevent excessive canola biomass
- If irrigation is available, reduce water at critical sclerotinia infection timing.
About sclerotinia stem rot
Overview | What causes sclerotinia?
The fungus Sclerotinia sclerotiorum, which occurs in all the canola growing areas of Canada, causes sclerotinia stem rot of canola. Sclerotinia stem rot, or white mould as it is sometimes called, is the most destructive canola disease in Canada. The severity of sclerotinia stem rot is variable from year-to-year, region-to-region and even from field-to-field. Sclerotinia stem rot has become more serious as canola production has increased, likely due to a combination of more acres of canola in rotations and management practices that contribute to high yielding crop which produce dense canopies that have a more conducive microclimate for disease development. Wet weather has also favoured disease development in recent years 1.
In 2016, sclerotinia stem rot was widespread in canola across much of Western Canada, with over 90 per cent of fields surveyed showing symptoms of infection (disease prevalenceThe proportion of plants surveyed which are infected with the disease (referred to as a percentage of the crop which is infested). More). In the Prairie provinces, the average disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More of fields surveyed was between 14 per cent and 30 per cent 2, 3, 4. This incidence of disease (in 2016) would roughly equate to seven to 15 per cent yield loss. In 2019, the disease prevalenceThe proportion of plants surveyed which are infected with the disease (referred to as a percentage of the crop which is infested). More was lower ranging from 25 to 79 per cent, and the incidence ranged from five to 12 per cent.
Plants are infected with sclerotinia stem rot when the canola crop is flowering. Variation in the disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More and severity among fields not treated with fungicide is due to:
- rainfall/humidity/leaf wetness
- soil moisture
- crop density
- crop canopy/plant architecture
- crop height and vigourSeed properties that determine the potential for rapid uniform emergence and development of normal seedlings under a wide range of field conditions. More
- severity of lodging
- genetic tolerance/resistance of some varieties
- quantity of infectious spores (ascosporesMicroscopic asexual spores that typcially become airborne. More)
- timing of sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More germination
With the right combination of spore load, crop density and moisture conditions, severe infections can develop almost anywhere canola is grown in Canada. Even after plants are infected, the severity of sclerotinia stem rot symptoms and the resulting effect on yield will vary according to where plants are infected, crop density, lodging potential and especially the crop stage at the time of infection. Infections of the lower main stem or branch tissue initiated soon after early bloom will result in more severe symptoms.
Yield losses vary based on the reduction of seed filling per infected plant, the amount of pre-harvest shattering (causing seed loss), and the percentage of infected plants in a crop. In general, when conditions for the disease are favourable and infections are initiated during early flowering, yield reduction per infected plant can equal 50 per cent or more. Yield losses can also be attributed to fungal growth within stems and resulting lodging, which leads to some or all of the following:
- smaller and fewer seeds
- premature ripening
- shattered pods
- loss of smaller, shrunken seeds during combining
Sclerotinia sclerotiorum is virulentA form which causes infection (and can result in a disease) More on over 400 other plant species, including broadleaf crops such as canola, mustard, sunflowers, beans, lentils, peas and as well as most broadleaf weeds, such as chickweed, stinkweed, hemp-nettle, thistles, shepherd’s purse, narrow-leafed hawk’s-beard, false ragweed, wild mustard among others.
Of all the canola diseases in Canada, sclerotinia stem rot is perhaps the most dramatically affected by environmental conditions. Due to the abundance of host plants, the likelihood of S. sclerotiorum being present is high. Most canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More are highly susceptible to this disease. But the variability of environmental conditions (which makes it hard to predict when the disease will be an issue and control will be needed) may have the largest impact on the development of a severe infection in a field.
Disease cycle for sclerotinia stem rot
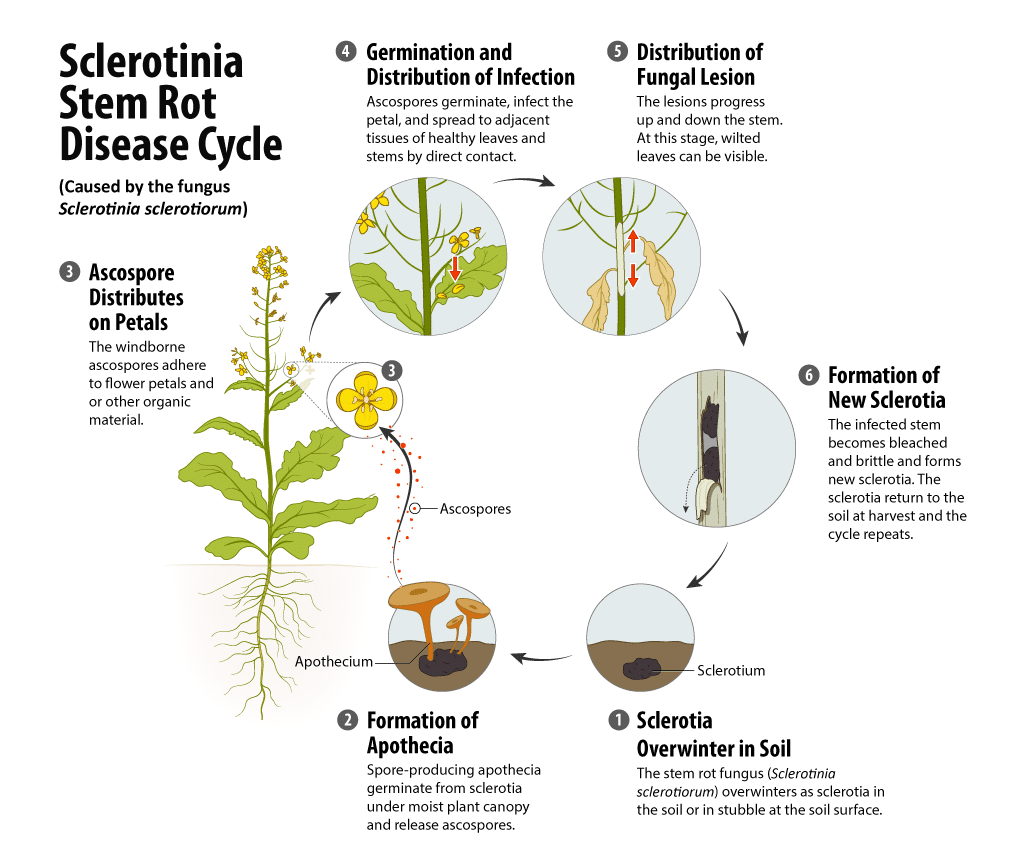
The stem rot fungus overwinters as sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More in the soil, in stubble at the soil surface and mixed with seed. SclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More can remain viable in the field for five years or more. The amount of sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More in the soil will largely reflect (be impacted by) the crop rotations used. The amount of sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More will increase under short canola rotations, when susceptible pulse or forage crops are grown in rotation or where significant broadleaf weed populations occur (especially in wetter regions of the Prairies). Areas with low rainfall are likely not conducive for the production of sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More and the development of the disease. Each year some sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More will germinate when conditions are suitable but others will remain dormant. Germination produces either myceliumMicroscopic fungal filaments More, which may infect plants, especially root tissues in direct contact with sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More, or spore-producing apotheciaSmall golf-tee shaped structures which produce ascospores. More.
Nearly all infections in canola result from microscopic airborne spores (called ascosporesMicroscopic asexual spores that typcially become airborne. More) produced by apotheciaSmall golf-tee shaped structures which produce ascospores. More at the soil surface. However, for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More to germinate and produce apotheciaSmall golf-tee shaped structures which produce ascospores. More, they require prolonged moist soil conditions (at least 10 days above the wilting point) and moderate temperatures of 15 to 25 degrees Celsius. Normally such conditions do not occur until the crop canopy closes (typically at the late rosette stage) and permanently shades the soil surface. If the crop canopy closure coincides with a 10-day period of prolonged moist soil conditions, (which is required for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More to germinate and produce apotheciaSmall golf-tee shaped structures which produce ascospores. More) apotheciaSmall golf-tee shaped structures which produce ascospores. More may appear as the crop starts flowering. Only sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More in the top few centimetres of soil will produce functional apotheciaSmall golf-tee shaped structures which produce ascospores. More, since the apothecial stalks are rarely longer than five centimetres (two inches). Deeply buried sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More will not produce apotheciaSmall golf-tee shaped structures which produce ascospores. More but can remain dormant. If brought near the surface by cultivation, they may germinate.
A single sclerotium can produce up to 15 apotheciaSmall golf-tee shaped structures which produce ascospores. More, either at one time or over a period of weeks. ApotheciaSmall golf-tee shaped structures which produce ascospores. More only grow from sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More and not from any plant tissue or residue in the soil. Occasionally apotheciaSmall golf-tee shaped structures which produce ascospores. More can emerge from sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More inside plant tissue trapped within undecomposed stubble.
In Western Canada, apotheciaSmall golf-tee shaped structures which produce ascospores. More normally begin to appear in June, but most develop during flowering. ApotheciaSmall golf-tee shaped structures which produce ascospores. More can continue to develop until late September but the critical period for them to cause damaging infections to crops is from early to full bloom. Plant infections rarely develop before the mid-flowering stage and apotheciaSmall golf-tee shaped structures which produce ascospores. More rarely appear in fields before plants are at the bud stage.
ApotheciaSmall golf-tee shaped structures which produce ascospores. More are typically five to 15 millimetres in diameter. The upper end of this range is about the size of a small fingernail or slightly smaller than a dime. ApotheciaSmall golf-tee shaped structures which produce ascospores. More produce millions of ascosporesMicroscopic asexual spores that typcially become airborne. More that are released into moving air currents. Even the lightest breeze easily carries the ascosporesMicroscopic asexual spores that typcially become airborne. More across a field or into adjacent fields, possibly as far as several kilometres away. Honeybees can also carry ascosporesMicroscopic asexual spores that typcially become airborne. More.
AscosporesMicroscopic asexual spores that typcially become airborne. More normally cannot infect the leaves and stems directly, they must first grow in dead petals or other organic material adhering to leaves and stems. The petals provide the food source necessary for the spores to germinate and produce the hyphae that grow and eventually penetrate the plant. Moist conditions from rainfall or heavy dew, which may keep leaves and stems wet for two to three days, are also necessary for infection. Radiation, relative humidity and temperature influence ascospore survival.
AscosporesMicroscopic asexual spores that typcially become airborne. More are fairly short-lived and only remain viable for up to four to five days. Research has found that the average ascospore survival was up to 50 per cent after two days and up to one per cent after three days of field exposure. Other factors such as exposure to radiation, higher temperatures (greater than 25 degrees Celsius) and high relative humidity (greater than 35 per cent) can result in more rapid loss of ascospore viability after one to two days on canola petals. The combination of ascosporesMicroscopic asexual spores that typcially become airborne. More being released and deposited on petals along with the crop coming into full bloom creates the highest risk for infection, depending on weather and environmental factors 5, (Turkington, T.K., Morrall, R.A.A. and Rude, S.V. 1991. Use of Petal Infestation to Forecast Sclerotinia Stem Rot of Canola: The impact of diurnal and weather-related inoculum fluctuations. Canadian Journal of Plant Pathology. 13(4): 347-355.)), 6, 7.
Sites of initial infection become lesions on the plant, which can grow to become quite large. When lesions develop on leaves and leaf axils or bases, these lesions can spread down into the stem, invading the healthy stem tissue. A dense canopy provides higher humidity and more favourable conditions for disease development.
In some cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, heavy stands can lodge, and sclerotinia stem rot will spread from plant-to-plant by direct contact, especially if wet weather delays swathing or straight combining. Spread will also occur in wet newly-cut swaths. The fungus eventually forms new sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More in diseased plants that are returned to the soil at harvest, completing the disease cycle.
Influence of environment
Rainfall and soil moisture are necessary for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More germination, spore production, and spore germination and growth. Frequently water from heavy dews dripping off the plant is enough moisture for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More germination. Ideal canola growing weather is also ideal for sclerotinia stem rot.
Lesion development is favoured by humid conditions and temperatures between 20 and 25 degrees Celsius. Dry, warm conditions will impede or stop infection and lesion development, but established lesions may resume growth when favourable conditions return.
Identification
Symptoms of sclerotinia stem rot
Sclerotinia stem rot develops late in the season, with the first visual symptoms appearing by the end of flowering. Two to three weeks after infection, soft watery lesions or areas of very light brown discolouration become obvious on the leaves, main stems and branches. Lesions expand, become greyish white, and may have faint concentric markings. Plants with girdled stems wilt, ripen prematurely and become conspicuously straw-coloured in a crop that is otherwise still green.
The stems of infected plants eventually bleach, taking on a whitish appearance and infected tissues tend to shred and shatter easily. Infected plants may produce fewer pods per plant, fewer seeds per pod or small, shrivelled seeds that blow out the back of the combine. The extent of damage depends on whether the main stems or branches are infected and at what stage during flowering infection occurs. Severely infected crops frequently lodge, are difficult to swath and shatter during swathing.
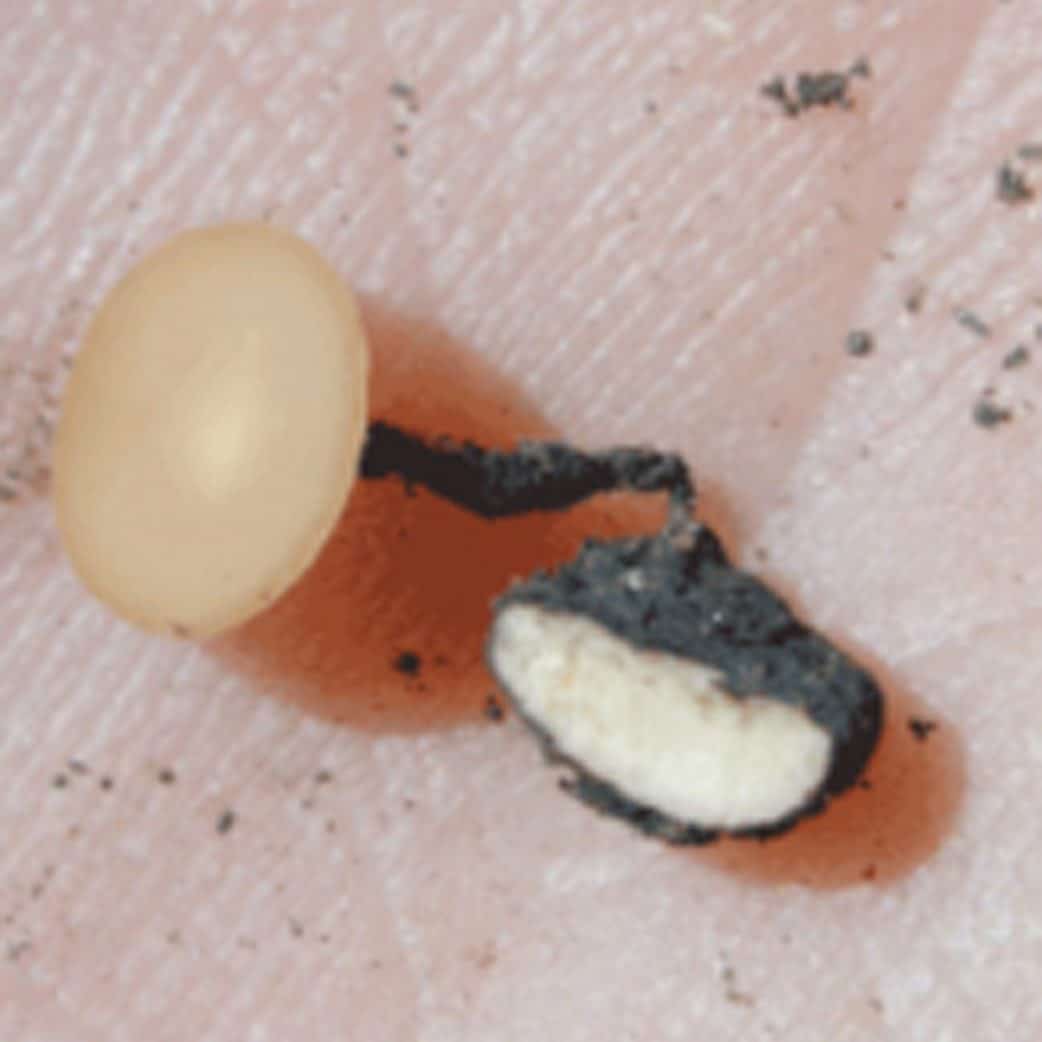
When the bleached stems of diseased plants are split open, a white mouldy growth and sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More are visible. SclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More vary in size and shape. They may be small and round like a canola seed or up to two centimetres (0.8 inch) long and cylindrical, ovoid or irregular in shape. Under moist conditions, sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More and the white mouldy growth may also occur on the surface of infected areas of the plant. At harvest the sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More are either threshed out with the seed or left in the field.
Sclerotinia management | How do you manage sclerotinia?
Fungicides
Fungicides are the most effective management tool to control sclerotinia stem rot in canola when the risk of infection is high. Where required, the use of fungicides not only protects (remaining) yield but also reduces dockage caused by sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More contamination and small, shrivelled seed. Since the cost of spraying a fungicide is high and sclerotinia disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More varies greatly among years, regions, and fields, systematic spraying is not profitable. By estimating the risk of infection, severe yield losses and unnecessary fungicide applications can be avoided 1.
There are several fungicides registered for sclerotinia stem rot control in canola (provincial crop protection guide should always be consulted for an up-to-date list of registered products). Most fungicides recommend timing application to cover the maximum number of canola petals with fungicide at the early stage of the flowering period. Some of these products are registered for a split application, which has the advantage of providing longer protection if the bloom period is extended due to cool, wet conditions or uneven maturity due to staggered emergence or excessive branching from low plant populations. There are also biological products available, which may require differing methods and timings of application.
Table 1. Sclerotinia stem rot fungicides for canola in Canada
Source: Guide to Field Crop Protection 2024 (Manitoba Edition)
Fungicide timing
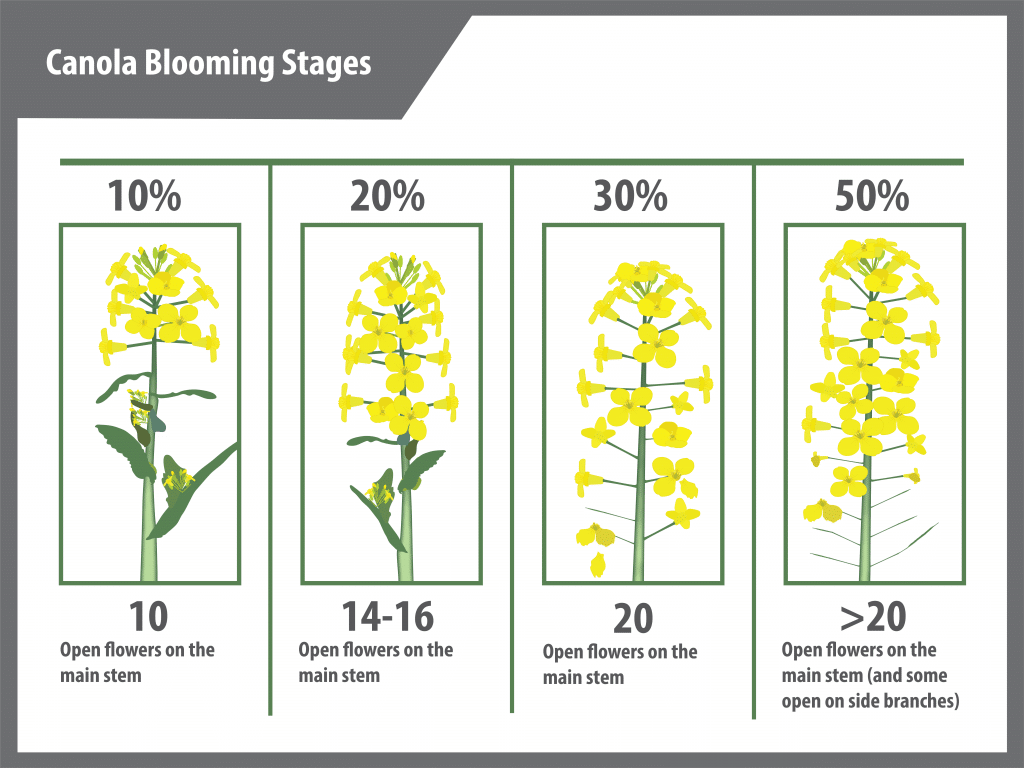
If planning to use a fungicide for sclerotinia control, the optimal time to spray must be decided. For most fungicides, the recommended window for application falls between 20 and 50 per cent bloom, with optimum timing typically around 30 per cent bloom. To assess the bloom stage, sample several plants over the field and count the number of open flowers. One method is to identify the main stem, remove the secondary branches, and count only the open flowers on the main stem. Generally, it takes a crop two to four days to move from first flower to 10 per cent bloom.
The objective of the fungicide application is to cover as many petals as possible while ensuring that some chemical also penetrates into the canopy to help protect potential infection sites (such as leaf axils and bases). However, the fungicide is only active on petals present at the time of spraying and will not protect petals that emerge after spraying. Most fungicides are contact based with limited systemic function. Additionally, the curative abilities of most fungicides are limited on established sclerotinia stem rot lesions, especially those that are large or have penetrated stem tissue. Therefore it is important to apply the fungicide prior to significant petal drop, when environmental conditions are conducive to sclerotinia infection.
Registered fungicides are antagonistic and lethal to S. sclerotiorum ascosporesMicroscopic asexual spores that typcially become airborne. More and to newly-germinated hyphae, preventing the fungus from initiating the infection process. Canola flower petals which are not covered by fungicide may still initiate infection in crops that were sprayed. Since coverage of flower petals is a key consideration, the use of high water volumes is often recommended.
Research at Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More Saskatoon compared five methods of fungicide application over three years to determine the effect of nozzle type and pressure on sclerotinia stem rot infection of canola and crop yield. Variation in product performance among years indicated that environmental conditions had a major effect on crop development, sclerotinia stem rot infection, the effect of fungicides and subsequent yield. Overall, conventional flat fan nozzles (TeeJet XR), low-drift venturi nozzles (CFFC TurboDrop) and hollow cone nozzles (TeeJet TXVS-8) were all effective at reducing disease symptoms. Both 275 and 550 kilopascals (kPa), or 40 and 80 pounds per square inch (PSI) provided similar performance although increasing the venturi nozzle pressure to 550 kPa (80 PSI) improved disease control slightly. These results indicate that venturi nozzle technology is appropriate for use with foliar fungicides for sclerotinia control in canola provided pressures are adjusted to optimize nozzle performance 8
Economics of fungicide application
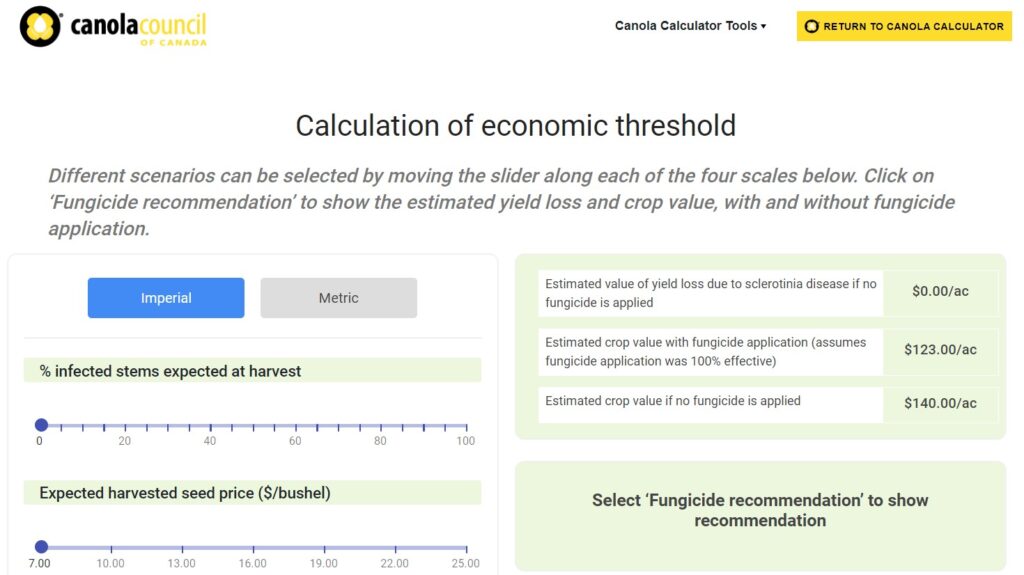
The decision to apply a fungicide to prevent sclerotinia stem rot depends on the risk of the disease developing. Forecasting and risk analysis is discussed in detail in the section below. This decision may become clearer by answering these four questions:
- Have environmental conditions prior to flowering been wet enough for apotheciaSmall golf-tee shaped structures which produce ascospores. More development and survival?
- Is the canola crop canopy dense and is yield potential high?
- Does the weather forecast predict precipitation and/or humidity during the flowering period?
- Is the pathogenA disease-causing organism (such as a fungus or bacteria). More present in sufficient quantities?
If the answer is “yes” to all four of these questions, then spraying is generally recommended. If the answer to some of the questions is “no”, then the decision is more difficult. Utilize the sclerotinia stem rot checklist to aid the decision, but also consider canola price and overall profit potential of the crop before investing more in it. Canola canopies with yield potential of less than 30 bushels per acre are less likely to suffer a sclerotinia stem rot infection big enough to make spraying economical, unless conditions for infection are ideal. Crops with an open canopy could be at lower risk, particularly if warm and windy weather allows them to dry out in the afternoons. However, if frequent rains and high humidity keep the plants wet, then there is still a threat. Rainfall at flowering and thick canola canopies increase the sclerotinia risk, especially if the field is located in an area where sclerotinia sprays are often warranted.
Canola Council of Canada research shows that it may be economically justifiable to apply fungicide when scouting indicates that disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More levels will reach 15 per cent in B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More and over 30 per cent in B. rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More by crop maturity (no data on B. junceaAlso referred to as brown mustard, it is a minor crop (from the Cruciferae or Brassicaceae plant family, commonly known as the mustard family) grown in Canada. More is available). This is based on an $8 return per bushel ($353 per tonne) and an application cost of the fungicide at around $22 per acre ($55 per hectare).
As a rule of thumb, the potential yield loss in a field can be determined by:
Per cent potential yield loss = Per cent disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More x 0.5
(where disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More = per cent of plants infected)
For example, a canola field with a 50 per cent of the plants infected with sclerotinia stem rot would have an approximate yield loss of 25 per cent (50 per cent disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More x 0.50 = 25 per cent). The actual yield losses depend on the environmental conditions, canopy structure, cultivar, and time of infection.
To calculate loss in revenue per acre, use the estimated per cent yield loss with the estimated yield of the field and the estimated market price for canola. If this lost revenue value is greater than the cost of a fungicide application per acre, then a fungicide application is recommended.
Revenue loss/acre = Per cent potential yield loss x Estimated yield x cost per bushel
For example, consider a canola field with an estimated yield of 40 bushels per acre which is expected to sell for 10 dollars per acre. If the yield loss due to sclerotinia infection is 15 per cent, then the loss in revenue will be 60 dollars per acre. If the cost of fungicide application was 30 dollars per acre, then spraying a fungicide would have been economical (in this example).
Forecasting and risk assessment
There are a few different forecasting systems have been developed for predicting the risk of sclerotinia stem rot outbreaks in canola. While no forecast system is completely accurate, they do provide practical direction on making a decision as to whether a fungicide application is warranted. These tools may help a canola grower decide whether or not to spray fungicide, as long as the system used accounts for all the factors that will impact disease development.
Factors involved in forecasting
Many factors influence a forecasting system and its ability to predict the actual incidence of disease. Most predictive models evaluate several environmental and crop variables (in order of importance):
- rainfall
- soil moisture
- weather forecast
- canopy density and yield potential
- cultivar resistance
- ascospore presence
- field disease history
Other variables that may affect the incidence of the disease include:
- field cropping history
- adjacent field disease history
- changing ascospore levels during flowering
- heat units
- daily and weather related ascospore fluctuations
- light penetration
- leaf area index
- crop height and lodging susceptibility
- leaf wetness
Environmental conditions, in particular, the amount of available moisture leading up to and during flowering, has the largest impact on the development of sclerotinia stem rot in a field. The presence of the pathogenA disease-causing organism (such as a fungus or bacteria). More is normally expected to be present in most fields in Western Canada now that this fungus is so widely established and has so many hosts (including substantial canola acreage each year).
Sclerotinia stem rot checklist
Sclerotinia stem rot infection can vary greatly among fields and years, making scheduled routine spraying of fungicides unprofitable. However, when sclerotinia risk is high, preventative fungicide applications can effectively lower disease severity and improve yield. Assessment of disease risk within each field is essential to improve the odds that fungicides are only applied when it is economical to do so.
Determining whether or not the risk of sclerotinia warrants a fungicide application is a difficult decision. This decision depends on many variables including:
- potential yield and price of the crop
- cost of the fungicide
- current and predicted rainfall
- presence and level of ascosporesMicroscopic asexual spores that typcially become airborne. More
- canola susceptibility
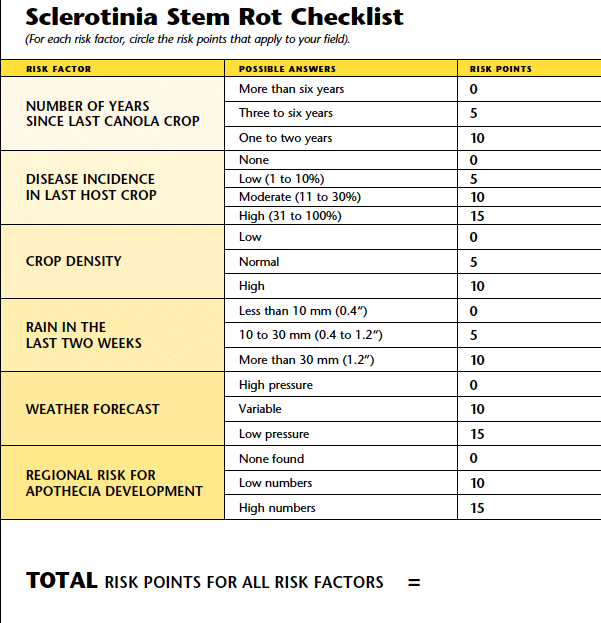
The Sclerotinia stem rot checklist is a comprehensive tool to assess the risk of sclerotinia stem rot developing in a given field. The checklist was first developed in Sweden, and has been modified for Canadian conditions. Growers should assess the crop and fill out the checklist for each field shortly after first flower (when 75 per cent of the canola plants have at least three open flowers). Usually this occurs during the last week of June or the first week of July. Read each question and add the point value assigned to the answer that best describes the conditions for that field. The total of the risk points from all questions represents the overall risk for sclerotinia. It is important to assess the risk early in the flowering period so that arrangements for chemical and application can be made if required. Continual monitoring up to full bloom is advisable as risk can change dramatically from early flower to full bloom.
The greater the score (total risk) for a field, the higher the probability of a positive economic return to a foliar fungicide application. Swedish researchers who developed this model (through evaluation of 800 fields over a 10-year period) found that fields with a total of less than 40 points, had a low risk of heavy infection (disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More exceeding 25 per cent). A threshold value of 40 risk points accurately identified 75 per cent of the fields that needed spraying for sclerotinia but also 16 per cent of the fields that did not need a fungicide. A threshold of 35 risk points increased the accuracy to almost 90 per cent of the fields that needed spraying but also included more than 20 per cent of the fields that did not need spraying. Therefore, they concluded that under their conditions if the risk points were 40 or higher, it was likely worth spraying. If the risk points were less than 40, it was not likely worth spraying. This provides a good guideline for use of the checklist in Canada, but growers should keep in mind that it may vary a bit under our field conditions, and also depending on fungicide cost and commodity price.
Measuring the pathogen
There are a few different methods to measure whether the pathogenA disease-causing organism (such as a fungus or bacteria). More S. sclerotiorum is likely present in a field. Petal tests, spore traps, and scouting for apotheciaSmall golf-tee shaped structures which produce ascospores. More will all help to determine this factor for infection.
There are two types of petal tests currently available to determine if S. sclerotiorum is present on canola petals. One type of test uses DNA marker technology. Canola petals sampled from early flowers are sent to labs offering this service. The results are available in one to two days after the petals are received. The second type of petal test involves placing sampled petals on specifically produced selective agar-plates to observe if S. sclerotiorum myceliumMicroscopic fungal filaments More grows from these petals. This test can be done by canola grower or agronomists, but it takes about a week before results are available.

Spore trapping samplers can be installed in a canola field, which will capture the wind-blown S. sclerotiorum ascosporesMicroscopic asexual spores that typcially become airborne. More on a media or cassette. These spore-trapped samples are sent to a testing lab, which will provide results in one or two days after the samples are received.
Scouting for apotheciaSmall golf-tee shaped structures which produce ascospores. More is an indirect method to determine if ascosporesMicroscopic asexual spores that typcially become airborne. More may be present and infecting canola flower petals. ApotheciaSmall golf-tee shaped structures which produce ascospores. More are tan or honey-coloured, five to 15 millimetres across, and have tops which are cupped like a golf tee. They will be growing from sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More in the soil or decaying canola. There are other fungal structures or mushrooms produced by saprophytic fungi, meaning that they survive on decaying crop residue that can occur in similar conditions but are not apotheciaSmall golf-tee shaped structures which produce ascospores. More. The spores produced from these fungi do not contribute to increased sclerotinia risk. Above the soil surface the stalks of apotheciaSmall golf-tee shaped structures which produce ascospores. More tend to be short, so mushrooms with stalks that extend above the soil surface (e.g. 25 millimetres or more) are unlikely to be apotheciaSmall golf-tee shaped structures which produce ascospores. More of sclerotinia. Mushrooms with rounded tops or bright colours are not likely apotheciaSmall golf-tee shaped structures which produce ascospores. More of the sclerotinia fungus. ApotheciaSmall golf-tee shaped structures which produce ascospores. More may be found in a canola field, but they are often likely observed in adjacent fields that had canola or some other susceptible crop (such as sunflowers, soybeans, lentils, etc.) in the previous year.
All of these pathogenA disease-causing organism (such as a fungus or bacteria). More measurement tools only help determine one factor in forecasting this disease: ascospore presence. Environmental conditions still need to be considered before one decides to apply a fungicide. Using pathogenA disease-causing organism (such as a fungus or bacteria). More measurement results along with the Sclerotinia stem rot checklist is recommended.
Modelling the weather
Rainfall and soil moisture are necessary for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More germination, spore production, and spore germination and growth. Ideal canola growing weather is also ideal for sclerotinia. Soil moisture indicates sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More germination and, therefore, the potential for ascospore production rather than infection and disease development. Frequently water from heavy dews dripping off the plant is enough moisture for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More germination.
Weather forecasts can improve the reliability of the disease forecast because sudden weather changes can cause infestations to occur unexpectedly or high risk fields may show limited disease development. Hot, dry weather can greatly reduce the risk of sclerotinia infection.
Commercial weather measuring services offer sclerotinia forecasts based on local environmental conditions recorded at the field level. These highly specific measurements provide a comprehensive data set to help make a fungicide application decision. However none of the weather models have been validated for their accuracy in Western Canada, so far. Canola growers should be aware that these may not be providing the right forecast at this point in time. Weather prediction models do not provide a measurement of pathogenA disease-causing organism (such as a fungus or bacteria). More presence, so pathogenA disease-causing organism (such as a fungus or bacteria). More sampling may still be required. Combining weather prediction with the Sclerotinia stem rot checklist is recommended.
Field history
Field and nearby field cropping and disease history are an indirect average of measuring the potential for presence of ascosporesMicroscopic asexual spores that typcially become airborne. More. While apotheciaSmall golf-tee shaped structures which produce ascospores. More produced from sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More within the field are considered the main source of spores, spores produced in nearby fields and blown into the crop are important in disease development. Research has shown that most ascosporesMicroscopic asexual spores that typcially become airborne. More are deposited within 100 meters of the source, and therefore adjacent fields can be an important source of inoculum. Fields with sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More at distances of greater than 500 metres to two kilometres would likely represent a minimal source of inoculum, as most ascosporesMicroscopic asexual spores that typcially become airborne. More would have been deposited within 100 to 400 metres of the apotheciaSmall golf-tee shaped structures which produce ascospores. More from which they were produced 9, 10, 11.
Crop canopy density
Crop canopy density can influence the risk of sclerotinia, given favourable environmental conditions 12. The crop canopy can be modified by seeding rate, row spacing and fertility, which is the main factor impacting crop canopy density. Generally a high population of rapidly growing plants leads to faster canopy closure and thick, dense canopies. While these dense canopies often have the greatest yield potential, they tend to maintain high soil moisture levels beneath the canopy, increasing the germination of sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More. Moist soil conditions for approximately 10 days or more induces sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More to form apotheciaSmall golf-tee shaped structures which produce ascospores. More and subsequent spore release. Moist conditions in the lower canopy also increase the chances of infection being initiated when petals carrying spores land on lower leaves or leaf axils. In short season growing areas, B. rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More varieties tend to have lighter canopies and generally lower infection levels.
Cultivar selection
There are currently canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More on the market in Canada with sclerotinia resistance or tolerance. These cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More have demonstrated reduced levels of sclerotinia stem rot infection in field and laboratory trials. The mechanisms for this increased level of tolerance or resistance are not well described, but they do appear to reduce lesion development in stem and other tissues. This reduced severity of sclerotinia stem rot is effective but may not match the same level of control as a fungicide application. Therefore under high risk of disease development a fungicide application to these cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More may still be necessary.
To determine the amount of resistance or tolerance a cultivar has to sclerotinia stem rot, the canola industry has agreed to use the protocol developed by Dr. Lone Buchwaldt (from Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More) and the Pathology sub-committee of the Western Canada Canola/Rapeseed Recommending Committee. This protocol outlines the steps necessary in order to correctly test for sclerotinia resistance and to correctly label the amount of resistance for cultivar descriptions and marketing purposes. Additionally some seed developers may use their own sclerotinia stem rot rating system to describe the reduction in sclerotinia stem rot infection, which may incorporate architectural and canopy structural components which may reduce infection.
Sclerotinia stem rot has been observed to be severe in heavily lodged fields or areas within a field. Therefore choosing cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More that resist lodging may reduce sclerotinia severity.
Biological controls
Currently there are two biological products available for sclerotinia stem rot control in Canada: Contans and Serenade OPTI. Contans is a biological control product registered for control in canola as well as soybeans, dry edible beans, sunflower and safflower. The active ingredient is Coniothyrium minitans, a fungus that colonizes and slowly degrades the sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More when it comes in contact. The product is a pre-emergent bio-fungicide that requires several months for the fungus to destroy the viability of the sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More.
Serenade OPTI is a bacterial formulation of Bacillus subtillis which is applied in a foliar fashion at the 20 to 30 per cent bloom stage of canola, similar to how fungicides are applied. B. subtillis is antagonistic to fragile ascosporesMicroscopic asexual spores that typcially become airborne. More and newly developing hyphae.
Cultural controls
There are a number of agronomic or cultural considerations to examine with sclerotinia stem rot.
Crop rotation
Crop rotation is not always effective for managing sclerotinia stem rot because S. sclerotiorum can be host on many plants (including many popular crops in Western Canada) and because the fungus has the ability to survive for years in the soil as sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More. Also, air-borne ascosporesMicroscopic asexual spores that typcially become airborne. More can blow in from nearby fields, where they are released by apotheciaSmall golf-tee shaped structures which produce ascospores. More germinating from sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More left from previous broadleaf crops. Regardless, the opportunity for high sclerotinia stem rot disease levels is maximized if broadleaf crops are grown consecutively due to increased potential for spore production within the field.
Cereals and grasses are not susceptible, and can help reduce viable sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More in the soil through decay and germination in the absence of susceptible hosts. Therefore, it is better to avoid seeding canola adjacent to a field that had a heavily infected crop the previous year. Control of susceptible weeds and volunteer plants in cereal crops also helps to avoid replenishing viable sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More levels.
Seeding
Use of clean pedigreed seed, free of sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More is recommended. Since crop density is an important factor, avoid exceeding the recommended seeding rate. Research at the University of Manitoba found that increasing seeding rates two or three times above normal seeding rates can lead to lodging, which can increase sclerotinia infection 13. Lighter seeding rates or wider row spacing may increase ventilation within the crop canopy and reduce the moist soil microclimate required for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More to germinate, helping to reduce sclerotinia levels. However, seeding rates need to be adequate to produce a minimum of five to eight viable plants per square foot or yields may be compromised regardless of sclerotinia levels. Also, frequent rainfall and/or persistently high humidity may create a suitable environment for infection in spite of an open canopy.
Harvest and grade
Growers can apply fungicide for sclerotinia stem rot control and achieve good to excellent results in the standing crop. Unfortunately, this disease may progress rapidly in the swath in wet years particularly in B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Do not swath canola crops with significant incidence of infection if rain is forecast, particularly if the crop is immature (green) when cut. In wet compacted swaths, particularly on the turns, sclerotinia rot can progress rapidly. The disease can be detected by a rotten egg-like smell coming from the swaths. This problem is obviously more prevalent in the wetter regions of Western Canada. The heavier and more compact the swath, the greater the likelihood for sclerotinia to rot the swath before combining.
Steps to follow to control sclerotinia in the swath:
- Do not swath immature stands (target greater than 60 per cent seed colour change among the healthy plants).
- Do not swath if rain is in the immediate forecast.
- Avoid compacting swaths.
- Use a high cut to allow for better drying.
- Avoid heavy swaths on the turns.
- Consider straight combining the crop, but recognize that shattering losses from prematurely ripened infected plants will be significant if a pod shatter cultivar is not grown.
Up to one-third of a canola crop may be lost in the swath due to sclerotinia stem rot. Additionally, this increases the number of sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More which may be left in a field after harvest.
It may also result in the downgrading of canola due to sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More in the seed sample. Tolerances for sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More are very low. Seeds Regulations, which applies to mustard and canola, has a separate standard for the number sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More bodies allowed for each grade. When performing a purity analysis on the crop, sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More bodies are reported per 50 grams (Table VII with SCHEDULE I of the Seeds Regulation).
Table 2. Canola grade quality factor
Footnotes
- Kutcher, H.R, Dokken-Bouchard, F., Turkington, T.K., Fernando, W.G.D. Boyetchko, S. Buchwaldt, L. Hegedus D. and Parkin, I. 2011. Managing sclerotinia stem rot in canola. Canola Research Summit in Winnipeg, Manitoba, April 12-13.[↩][↩]
- Harding, M.W., Hill, T.B., Daniels, G.C., Rennie, DC., Zuzak, K., Feng, J. and McDonald, J. 2017. A survey for blackleg and sclerotinia stem rot on canola in Alberta in 2016. Canadian Plant Disease Survey.[↩]
- Ziesman, B., Bruce, D., Chant, S., Cubbon, D., Epp, I., Friesen, S., Frederick, J., Gray, K., Hertz, S., Hicks, L., Hladun, M., Ippolito, J., Jacob, C., Jurke, C., Kindrachuk, K., Kowalski, J., Borhan, H. Larkan, N., Loverin, J., Zemanek, J. 2017. Survey of canola diseases in Saskatchewan 2016. Canadian Plant Disease Survey.[↩]
- McLaren, D.L., Kubinec, A., Graham, J., Bargen, E., Brackenreed, A., Brar, D., Buss, T., Chomokovski, D., Farooq, A., Henderson, T.M., Kaskiw, L., Kristjanson, I., Lange, D., McCracken, M., Cornelsen, J., Picard, R., Pradhan, M. and Valentino, M. 2017. Survey of canola diseases in Manitoba in 2016. Canadian Plant Disease Survey.[↩]
- Turkington, T.K. 1991. Factors influencing a petal-based forecasting system for sclerotinia stem rot of canola. PhD Thesis. University of Saskatchewan, Saskatoon.[↩]
- Turkington, T.K. and Morrall, R.A.A. 1993. Use of Petal Infestation to Forecast Sclerotinia Stem Rot of Canola: The Influence of Inoculum Variation over the Flowering Period and Canopy Density. Phytopathology. 83: 682-689.[↩]
- Clarkson, J. P., Staveley, J., Phelps, K., Young, C.S. and Whipps, J.M. 2003. Ascospore release and survival in Sclerotinia sclerotiorum. Mycol. Res. 107(2): 213-222.[↩]
- Kutcher, H.R. and Wolf, T.M. 2006. Low-drift fungicide application technology for sclerotinia stem rot control in canola. Crop Protection. 25(7): 640-646.[↩]
- Qandaha, I.S., and del Río, L. E. 2006. Dispersal of Sclerotinia sclerotiorum ascosporesMicroscopic asexual spores that typcially become airborne. More in canola fields from area source of inoculum. Department of Plant Pathology North Dakota State University, Fargo[↩]
- Ben-Yephet, Y. and Bitton. S. 1985. Use of a selective medium to study the dispersal of ascosporesMicroscopic asexual spores that typcially become airborne. More of Sclerotinia sclerotiorum. Phytoparasitica 13:1.[↩]
- de Jong, M.D., Bourdôt, G.W., Hurrell, G.A. Saville, D.J., Erbrink, H.J., Zadoks, J.C. 2002. Risk analysis for biological weed control – simulating dispersal of Sclerotinia sclerotiorum (Lib.) de Bary ascosporesMicroscopic asexual spores that typcially become airborne. More from a pasture after biological control of Cirsium arvense (L.) Scop. Aerobiologia 18: 211–222. https://doi.org/10.1023/A:1021339202533[↩]
- Kutcher, R., Turkington, K., Rashid, K., Strelkov, S., Lange, R. and Brandt, S. 2006. Management of Oilseed Diseases. SSCA Conference.[↩]
- Jurke, C.J. and Fernando, W.G.D. 2006. Effects of seeding rate and plant density on sclerotinia stem rot incidence in canola. Archives of Phytopathology and Plant Protection. 41: 142-155. https://doi.org/10.1080/03235400600679743.[↩]