Since this was only reported for the first time in 2014, there is still much to learn about the disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More, severity, impact and management strategies. Provincial monitoring and Canadian research is in progress on this soil-borne disease which has impacted other canola or rapeseed-growing nations around the world.
Important information on verticillium stripe
- Canola infected with the fungal species Verticillium longisporum was discovered in Manitoba in 2014. This prompted follow up soil surveys by the Canadian Food Inspection Agency in 2015, which then found V. longisporum in Alberta, British Columbia, Manitoba, Ontario, Quebec and Saskatchewan.
- Research projects on verticillium stripe were established in 2018 through the Canadian Agricultural Partnership program. These will be the first projects in Canada on verticillium stripe and look to help develop recommendations for management and understand crop impact.
- Since verticillium stripe is a soil-borne disease the management practices will be similar to that of clubroot (which is also a soil-borne disease) and will include strategies to minimizing soil movement, sanitize equipment, and extend crop rotations.
- Although differentiating this disease from others can take practice, scouting for symptoms is encouraged and is best done post-harvest.
- Learn more about verticillium stripe identification and management in this Canola Watch fundamentals article.
- Summaries of completed and ongoing verticillium stripe research and related blog can be found on the Canola Research Hub.
Overview
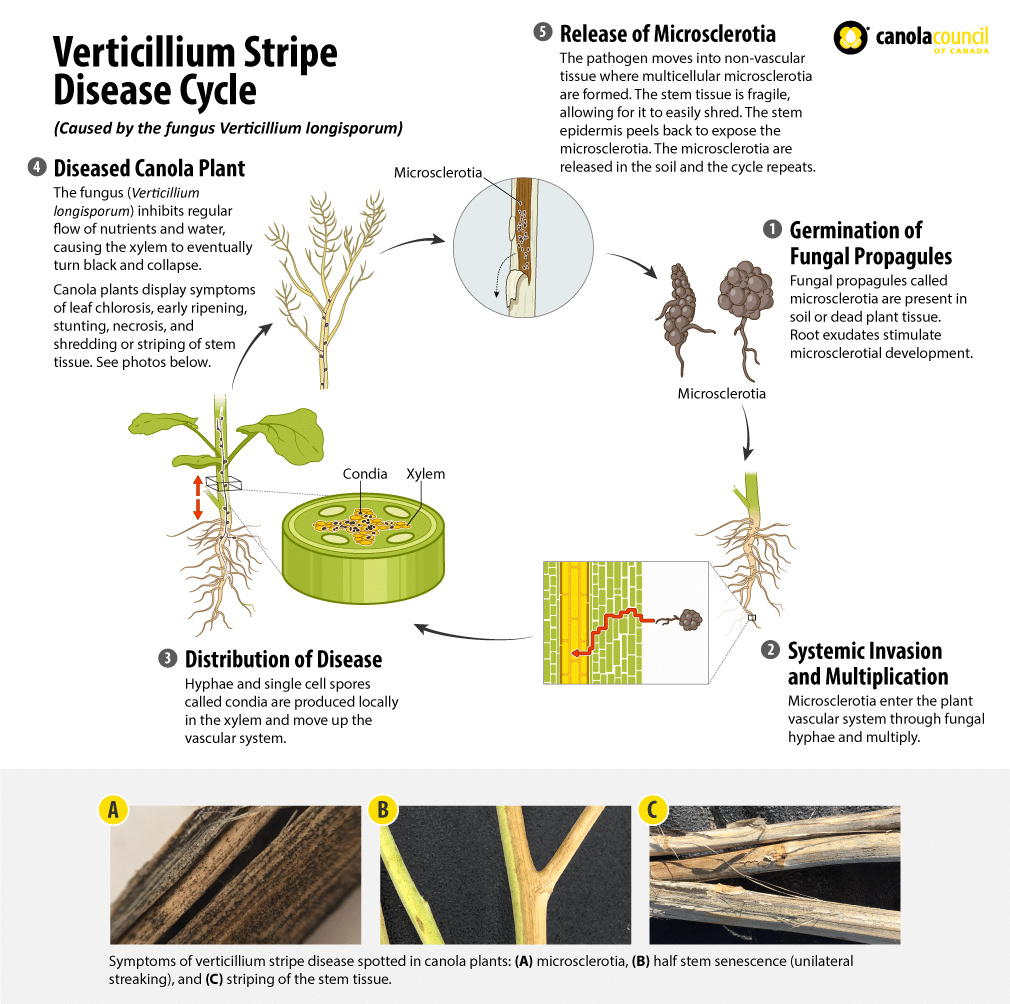
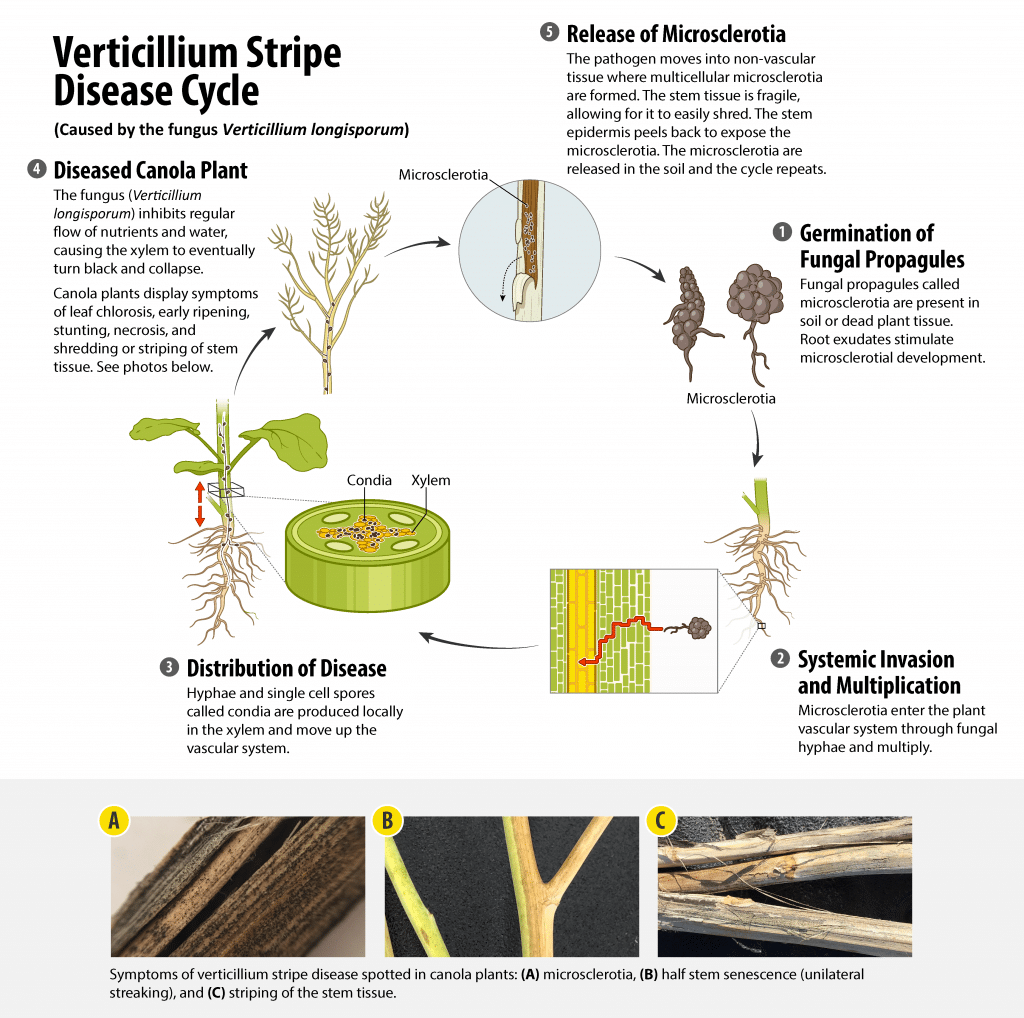
Verticillium stripe of canola is caused by the fungal species Verticillium longisporum. Verticillium stripe was first discovered in Canadian canola production areas in 2014. Disease symptoms in canola include leaf chlorosis, early ripening, stunting and, as the disease progresses, necrosisA symptom of plant cell or plant tissue death. More, shredding of the stem tissue. Once the plant is fully ripe, the stem peels to reveal tiny black microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More which resemble ground pepper in appearance. These microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More remain on the plant stem or fall to the soil. Those in stems are released in soil as the stems decay. They are hardy and can survive in the soil for many years. MicrosclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More can move with surface and ground water, through wind dispersal of infested soil or crop debris, planting and harvesting equipment contaminated with infested soil or crop debris, seed contaminated with infested soil or crop debris and people (for example, infested soil adhering to footwear) from one field to another((Dunker, S., Keunecke, H., Steinbach, P., & Tiedemann, A.V. 2008. Impact of Verticillium longisporum on yield and morphology of winter oilseed rape (Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More) in relation to systemic spread in the plant. Journal of Phytopathology, 156, 698-707.)),((Heale, J. B., & Karapapa, V.K. 1999. The Verticillium threat to Canada’s major oilseed crop: canola. Canadian Journal of Plant Pathology, 21, 1-7.)).
Disease cycle
The rapid germination of fungal propagules (likely mainly microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More) in the soil (triggered by the root exudate) is necessary in order to successfully infect the roots at the most susceptible location of the fast growing root tip. The coordination of both the timing of these activities and the location of the root tip as the site of highest root exudates (stimulating the propagule germination) works to the advantage of the pathogenA disease-causing organism (such as a fungus or bacteria). More. Additionally, the timing of the commencement of flowering plant stage has been said to be important to the spread of V. longisporum in canola, when the plant has been reported to be most susceptible1.
The fungus can enter the vascular system by the root directly or through an opening (wound) in the root (which is preferred) via the fungal hyphae. After the hyphae multiply in the root1 , hyphae and single-cell spores called conidiaSingle-cell spores or fungal propagules (such as those Verticillium species form). More are produced locally in the xylem and moved through the vascular system of the plant to multiply. This prevents the regular flow and functionality of water and nutrient up to the plant tissues and eventually causing the xylem to plug, turn black, collapse and shrivel2.
As the plant begins to senesce, the pathogenA disease-causing organism (such as a fungus or bacteria). More moves from the xylem into the surrounding (non-vascular) tissues, where multi-cellular microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More are formed in the dying tissue1. Likewise, microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More production in the stem base and roots cause the tissues to turn dark grey, then black and can cause the lateral roots to eventually breakdown (making it easy to pull from the ground). While the plant begins to show progressively more intense symptoms of the disease, the microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More in the stems cause a peeling back of the stem epidermisThin outer layer of a tissue (such as the thin outer later of plant tissue which peels back from canola stems which are infected with verticillium stripe). More (thin outer layer). At this point the stem may take on a shredded appearance and the microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More will be released to the soil, where they will rest until stimulated to germinate again. The microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More remain viable in the soil for several years, especially if infected residue is incorporated into the soil after harvest ((Eastburn, D.M., & Paul, V.H. 2007. Compendium of Brassica Diseases. Verticillium Wilt.)).
Verticillium stripe is a monocyclic disease, meaning it only goes through one cycle of the disease each year2. However, if infected plants ripen prematurely, they can show significant yield reduction3.
Verticillium longisporum could cause significant yield damage in canola, but only when disease severity and disease infection are at high levels. Due to the late (in the growing season) onset of the disease, verticillium stripe is less damaging than other diseases such as blackleg or sclerotinia stem rot.
The longevity in the soil may differ between V. longisporum and V. dahliae due to differences in the microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More (due to the elongated versus compact shape, respectively). While the microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More may survive up to 10-15 years in the soil, even switching to a four to five-year rotation with non-Brassica crops may be considered useful. Unfortunately, V. longisporum has also been reported to be less sensitive to the canola plant’s defense mechanisms (natural fungicidal products that canola forms through the breakdown of glucosinolates) than V. dahlia. In fact, V. longisporum even suppressed the production of glucosinolates of infected hosts4.
Symptoms
The interference of water and nutrient uptake caused by verticillium stripe can cause the crop to show signs of stunting5 and premature senescence. When individual plants are closely observed, faint black (vertical) striping can be seen on the stems which can appear darker and more obvious when rubbed. By peeling back the epidermisThin outer layer of a tissue (such as the thin outer later of plant tissue which peels back from canola stems which are infected with verticillium stripe). More and outer cortex of the stem, blackening can be seen on the inside of the stem, and eventually microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More, later in the growing season. The striping will almost become more obvious deeper into the maturation process due to the tissues dying below the stem surface. At the end of ripening, the microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More will begin to germinate and produce conidiaSingle-cell spores or fungal propagules (such as those Verticillium species form). More spores externally, giving the outside of the stems a powdery look3.
Symptoms of this disease can be noticed on the leaves and pods, but primarily are noticed on the stem and roots, and will vary according to the plant age and environmental factors. The infection can be scattered throughout the field, in groups, or affect a whole field. It is best to start looking for symptoms after flowering, although symptoms are not typically observed until a little later in the growing season, when the plants are nearing maturation2.
In a lab setting, when cotyledons are inoculated, chlorosis of the cotyledons has been observed. Similarly, chlorosis followed by dead plant tissue was the reaction to infected leaves which first showed symptoms in the lower leaves and progressively moved upwards, just like typical ripening6.
Since some of the symptoms of this disease overlap with those of regular senescence, and variability in disease severity based on the plant health, environmental conditions, symptoms may be hard to notice (or diagnose) in less severe cases6.
The disease can easily be mistaken for other diseases such as sclerotinia stem rot and blackleg at first, but careful diagnosis and can be used to accurately identify this disease. While there may be shredding of the stem similar to sclerotinia stem rot, the large sclerotiaHard, black resting bodies made of compact masses of hyphae; the plural term for sclerotium More and hollowing inside the stem of sclerotinia is different than the tiny microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More of verticillium stripe. While the discoloured stem and premature ripening could also be symptomatic of blackleg, cutting the stem at ground level and observing the cross-section of the stem will clarify. Blackening inside the stem will identify the disease as blackleg, as opposed to stem with no blackening in the stem cross-section. Hail or physical damage can also cause lesions similar to verticillium stripe, but this can be distinguished by the lack of microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More beneath the epidermisThin outer layer of a tissue (such as the thin outer later of plant tissue which peels back from canola stems which are infected with verticillium stripe). More7.
Influence of the environment
Verticillium stripe is most effective in hot, dry conditions, as soil temperatures of 15 to 19 degrees Celsius and air temperatures of 23 degrees Celsius are favourable temperatures for it, and root and xylem function is reduced due to stressful conditions (while excess moisture makes the disease less of an issue)2. Plants with damaged roots also make it easier for the disease to enter the vascular system ((Heale, J. B., & Karapapa, V.K. 1999. The Verticillium threat to Canada’s major oilseed crop: canola, Canadian Journal of Plant Pathology 21: 1-7.)).
Verticillium stripe can use weedy host species such as wild mustard and others outside of the Brassicaceae family as host species2.For example, disease symptoms were visible on scentless mayweed (chamomile) in a Swedish study5.
While this is the first report of this particular pathogenA disease-causing organism (such as a fungus or bacteria). More in Canada, V. longisporum can affect a number of annual and perennial plant species in both temperate and subtropical zones,2 and is present in oilseed rape in Sweden, Germany, Poland, Ukraine, Russia, France, the Czech Republic, Belgium, the Netherlands, the U.K. and Japan. First reported in 1960 in Sweden, the disease is now fairly widespread in Europe5. Closer to Canada, V. longisporum has also been found in cauliflower in California and horseradish in Illinois. The pathogenA disease-causing organism (such as a fungus or bacteria). More can infect many crops (broccoli, cabbage, cauliflower, horseradish, radish, and canola) and wild mustard in the mustard family.
While the disease is capable of significant yield loss if infection is established early, with yield losses in Europe reportedly being 10-50 per cent,8 severe yield losses have been confined to isolated fields and have not been reported as significant in regions or large areas. One of the most important factors affecting yield loss is reduction in seed size9 and lipid content10.
Management


Root cross-section of plants prior to harvest. Verticillium stripe (left), blackleg (middle), and healthy plant (right).
There are no foliar or seed treatment fungicides currently registered for control of verticillium stripe in canola in Canada. At this time there is no characterized host resistance in canola varieties to V. longisporum. Control measures for the closely related V. dahliae causing verticillium stripe or Early Dying of Potato include increasing rotation length of non-host crops, weed management, increasing soil fertility, use of non-host trap crops, green manures, fumigation (vapam and chloropicrin), and some limited genetic crop resistance.
While there are no commercial varieties considered to be resistant to verticillium stripe, differences in susceptibility between varieties has been reported2. The low susceptibility (showing almost no resulting yield loss) of other Brassica species (B. carinata and B. oleraceae) could suggest the potential for breeding solutions to counter this disease, if determined to be a significant disease in Canadian canola3,((Eynck, C., Koopmann, B., & Tiedemann, A. V. 2009. Identification of Brassica accessions with enhanced resistance to Verticillium longisporum under controlled and field conditions. Journal of Plant Diseases and Protection, 116, 63–72.)).
The potential for true seed-borne transmission (colonization of seed) of V. longisporum in canola has not been documented. However other Verticillium species have been found on seed due to contamination with infected pod or pedicel debris3.
In northern Europe, where this disease has been an important issue for more than 30 years, it is recommended that growers leave three years between canola crops. This allows the pathogenA disease-causing organism (such as a fungus or bacteria). More populations to naturally decline in the soil, but, due to the long-lived microsclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More, rotation alone is not enough to manage this problem effectively. As V. longisporum is a soil-borne pathogenA disease-causing organism (such as a fungus or bacteria). More, biosecurity practices can help mitigate the spread of this disease on- and off-farm. Biosecurity practices include equipment and tool sanitation, controlling off-farm traffic, monitoring seed, feed and fertilizer sources, and developing an on-farm biosecurity plan.
Testing
A reliable way to confirm if verticillium stripe is present in your field is by submitting samples to commercial laboratories. Labs in Canada that provide this service include:
Alberta
20/20 Seed Labs
507 11th Ave
Nisku, AB T9E 7N5
(780) 955-3435
Saskatchewan
Discovery Seed Labs
450 Melville Street
Saskatoon, SK S7J 4M2
(306) 249-4484
Manitoba
Pest Surveillance Initiative (PSI)
5A 1325 Markham Road
Winnipeg, MB R3T 4J6
(204) 813-2171
Genetic lineage
Molecular evidence suggests that V. longisporum is a plant pathogenA disease-causing organism (such as a fungus or bacteria). More that is a diploid hybrid (allodiploid), and that three species, including V. dahliae, contributed to the genetics of V. longisporum11. V. dahliae is present in Canada as a pathogenA disease-causing organism (such as a fungus or bacteria). More of potato, tomato, sunflower, strawberry, cabbage and maple. In order to determine the origination of V. longisporum, researchers used phylogenetic analyses of seven nuclear loci and a dataset of 203 isolates of V. longisporum, V. dahliae and related species. Based upon genetic analysis, the researchers showed that V. longisporum had originated independently at three different times, involving four different lineages and three different parental species. Each V. longisporum lineage was genetically homogenous and contained one set of alleles that were identical across lineages. This has potential impact on screening for resistance genes in B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More and other related Brassica species, implications for disease management and enforcement of quarantine.
- Zhou, L., Hu, Q., Johansson, A., & Dixelius, C. 2006. Verticillium longisporum and V. dahliae: infection and disease in Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More. Plant Pathology, 55, 137-144. [↩] [↩] [↩]
- Eastburn, D.M., & Paul, V.H. 2007. Compendium of Brassica Diseases. Verticillium Wilt. [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Heale, J. B., & Karapapa, V.K. 1999. The Verticillium threat to Canada’s major oilseed crop: canola, Canadian Journal of Plant Pathology, 21, 1-7. [↩] [↩] [↩] [↩]
- Heale, J. B., & Karapapa, V.K. 1999. The Verticillium threat to Canada’s major oilseed crop: canola, Canadian Journal of Plant Pathology 21: 1-7. [↩]
- Johansson, A., Goud, J.C., & Dixelius, C. 2006. Plant Host Range of Verticillium longisporum and MicrosclerotiaTiny, hardy spores or fungal propagules (such as those found in verticillium stripe-infected canola plants). More Density in Swedish Soils. Eur J Plant Pathol., 114, 139–149. https://doi.org/10.1007/s10658-005-2333-2 [↩] [↩] [↩]
- Heale, J. B., & Karapapa, V.K. 1999. The Verticillium threat to Canada’s major oilseed crop: canola. Canadian Journal of Plant Pathology, 21, 1-7. [↩] [↩]
- Saskatchewan Agriculture and Alberta Agriculture joint presentation in Saskatoon at the Canola Council of Canada canoLAB on February 10 and 11, 2015. [↩]
- Dunker, S., Keunecke, H., Steinbach, P., & Tiedemann, A.V. 2008. Impact of Verticillium longisporum on yield and morphology of winter oilseed rape (Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More) in relation to systemic spread in the plant. Journal of Phytopathology, 156, 698-707. [↩]
- Zhou, L., Hu, Q. Johansson, A., & Dixelius, C. 2006. Verticillium longisporum and V. dahliae: infection and disease in Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More. Plant Pathology, 55, 137-144. [↩]
- Johansson, A. 2006. Verticillium longisporum, infection, host range, prevalence and plant defense responses. Licentiate thesis Swedish University of Agricultural Sciences, Uppsala. [↩]
- Inderbitzin, P., Davis, R.M., Bostock, R.M., & Subbarao, K.V. 2011. The ascomycete Verticillium longisporum is a hybrid and a plant pathogenA disease-causing organism (such as a fungus or bacteria). More with an expanded host range. PLoS ONE 6: e18260. [↩]