Acidity, salinity and solonetzic soil characteristics can be major limiting factors in canola production. When severe, they can affect soil health and visibly impact crop growth. When less severe, effects may not be visible but, still reduce growth and yield. Identifying the extent of these and other soil characteristics, their effects on canola plants, and the steps to modify or reduce these limiting effects are important for maximizing canola production potential.
Important tips for best management
- Crop residues are essential for the maintenance of tilth on solonetzic soils. Surface mulch and below ground residue (roots) are better for solonetzic soils than incorporating straw. Use tillage only as required for manure incorporation, for example, or for post-harvest heavy harrowing in cereal stubble to distribute residue.
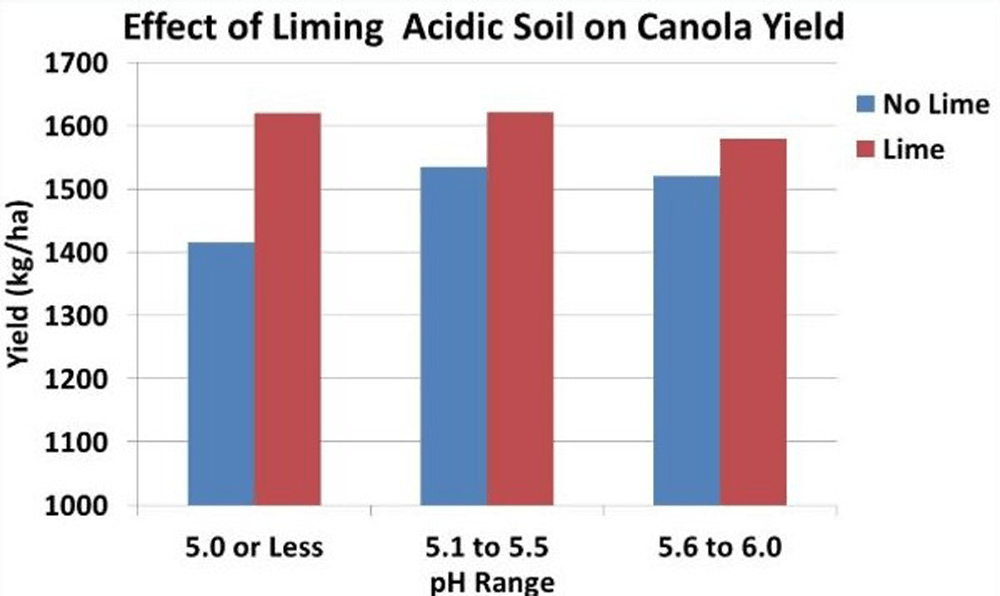
- Canola yields on acid soils (pH below 5.5) may be substantially increased by lime application.
- Higher organic matter will improve canola yield potential on most soils. To improve soil organic matter: limit soil erosion, retain crop residue, apply adequate nutrients, and add manure.
- Do not apply fertilizer or manure on frozen soil or snow, as spring runoff will remove many of those nutrients into rivers and lakes before they can enter the soil and be available to crops.
- Match nutrient rates to crop uptake. If using manure, test the nitrogen and phosphorus content of it and apply at appropriate rates. Applying more nitrogen than a crop can use could increase potential for surface water and groundwater contamination. Nitrogen fertilizer products that slow the rate of fertilizer conversion to nitrate may also help reduce leaching losses.
Soil texture
Soil texture refers to the mix of sand (course), silt (medium) and clay (fine) particles in the soil. Sand particles are much larger than silt and clay particles, so soils with a high percentage of sand in them (referred to as “light” soils) have lower water-holding capacities. Clay particles are smaller (as are the air spaces around them), so clay soils (referred to as “heavy” soils) are denser and have higher water-holding capacities.
Loam refers to a soil with a relatively equal mix of all three textures. Clay loam, for example, has all three, with more clay than sand or silt.
The best soil for canola production should have some water-holding capacity, lots of nitrogen mineralization potential and be in an area that receives adequate rainfall. Fine-textured soils (high in clay content) hold more water and will tend to produce better when rainfall is low, assuming they start out with ample water in the soil profile.
Soils can be classified by different orders, based on the properties determined by the main soil-forming processes. The major soil orders in Canada include Chernozemic, Gleysolic, Luvisolic, Regosolic, Solonetzic, Vertisolic, Brunisolic, Cryosolic, Podzolic and Organic soils. Each of these soil orders can be further divided into Great Groups and Subgroups1.
Solonetzic soils
There are about six million hectares (15 million acres) of land with Solonetzic soil in western Canada. This includes about four to five million hectares (10 to 12 million acres) in Alberta, one and a half million hectares (four million acres) in Saskatchewan and 10,000 hectares (25,000 acres) in Manitoba2.
Solonetzic soils, often called burnout, blow-out or gumbo soils, are characterized by an impermeable, high sodium, clay hardpan from five to 30 centimetres (two to 12 inches) or more below the surface (top of the Bnt horizon). This hardpan layer severely restricts root and water penetration below the topsoil. Solonetzic soils are formed on material that is naturally high in sodium salts or from materials that have been enriched with sodium salts through the upward movement of ground water.
Aside from variations in depth of topsoil, these soils differ in the degree of hardpan formation. Some of these soils have a tough hardpan and are high in exchangeable sodium. Others have a hardpan that has been leached of sodium — which is not as tough and can be penetrated by some roots and moisture. The latter tend to be the more productive solonetzic soils. Most solonetzic soils in the Peace River region are of this type3.
Solonetzic soils usually occur in association with normal soils. A field of good soil may contain patches of Solonetzic soil. These patches may vary from one metre (three feet) to several hundred metres in diameter and may represent as little as 10 per cent or as much as 90 per cent of the soil in a particular field.
Within a given field, good growth is often contrasted by thin, stunted growth on patches of Solonetzic soil. Crops tend to develop an uneven or wavy appearance.
Moisture is generally the most limiting factor to crop growth on solonetzic soils. The hardpan restricts root development and water infiltration and thus moisture use is limited mainly to the topsoil above the hardpan. Rainfall that doesn’t infiltrate flows to lower areas causing flooding. The deeper the depth of topsoil, the greater the moisture storage available.
Solonetzic soils usually have a pH between 5.5 and 6.5 in the surface layers (A and upper B horizons), however, pH values may be 5.0 or lower4. Canola yields are reduced when the soil pH is 5.5 or lower.
Seedbed preparation is relatively difficult on Solonetzic soils (which have shallow topsoil) as they dry out rapidly with cultivation. These soils may be more suited to rangeland and pasture for this reason. Growers who farm Solonetzic soils are strongly encouraged to direct seed using minimum tillage openers5.
Solonetzic soils, if cultivated when wet, can form large, hard clods that are difficult to break down into a good seedbed. This condition inhibits moisture contact with the seed and results in poor germination. In addition, the surface soil structure tends to break down easily under rainfall, and dries to form a hard crust that restricts seedling emergence.
Crop residues are essential for the maintenance of tilth on Solonetzic soils. Surface mulch and below ground residue (roots) are better for solonetzic soils than incorporating straw. Use tillage only as required for manure incorporation, for example, or for post-harvest heavy harrowing in cereal stubble to distribute residue6.
Provide surface drainage whenever possible to prevent temporary accumulation of water (ponding) before and after seeding. This will allow timelier seeding of the entire field and reduce uneven germination. Seed as soon as the soil temperature and moisture conditions are favourable for germination.
Solonetzic soils, like most other soils, will respond to applications of commercial fertilizer. However, response to fertilizer may be limited by a lack of moisture. Therefore, the optimum fertilizer application rates tend to be lower for these soils compared to other types because of the lower yield potential3. Crops grown on Solonetzic soils often suffer from drought in the middle to late growing season. Therefore, it is especially important that crops get an early vigorous start in these soils.
Subsoiling (ripping) has increased production on some Solonetzic soils for up to five years, due to deeper root penetration that improves soil water extraction and to increased water use efficiency7. However, not all Solonetzic soils are suitable for subsoiling. Solonetzic layers that can be penetrated by roots and moisture may not benefit from subsoiling8. Soil sampling to determine the suitability of the soil for deep tillage and to identify the ideal depth of tillage is recommended3. Consult a qualified soils specialist before a soil is subsoiled.
Soil pH
Strongly acidic (pH below 5.5) or alkali (pH above 8.5) soils will lower canola yield potential9.
Most cultivated soils in western Canada are alkaline or neutral. However, large areas of soil with a pH of 6.0 or lower occur naturally in Saskatchewan, Alberta, northeast British Columbia, and Ontario. It has been estimated that medium to strongly acid soils occupy over three million hectares (7.4 million acres) in western Canada.
Soil pH affects the structure, chemical properties and biological properties of soils and therefore can impact crop yields. Canola yields are often reduced substantially when grown on strongly acid soils10. Canola plant growth on acid soils can be limited by one or more factors, including:
- toxicity of hydrogen ions, aluminum, iron, or manganese.
- deficiency of calcium, magnesium, potassium, phosphorus, boron, nitrogen, or molybdenum.
- reduced organic matter breakdown and nutrient cycling by microflora.
- reduced uptake by plant roots and inhibition of root growth11.
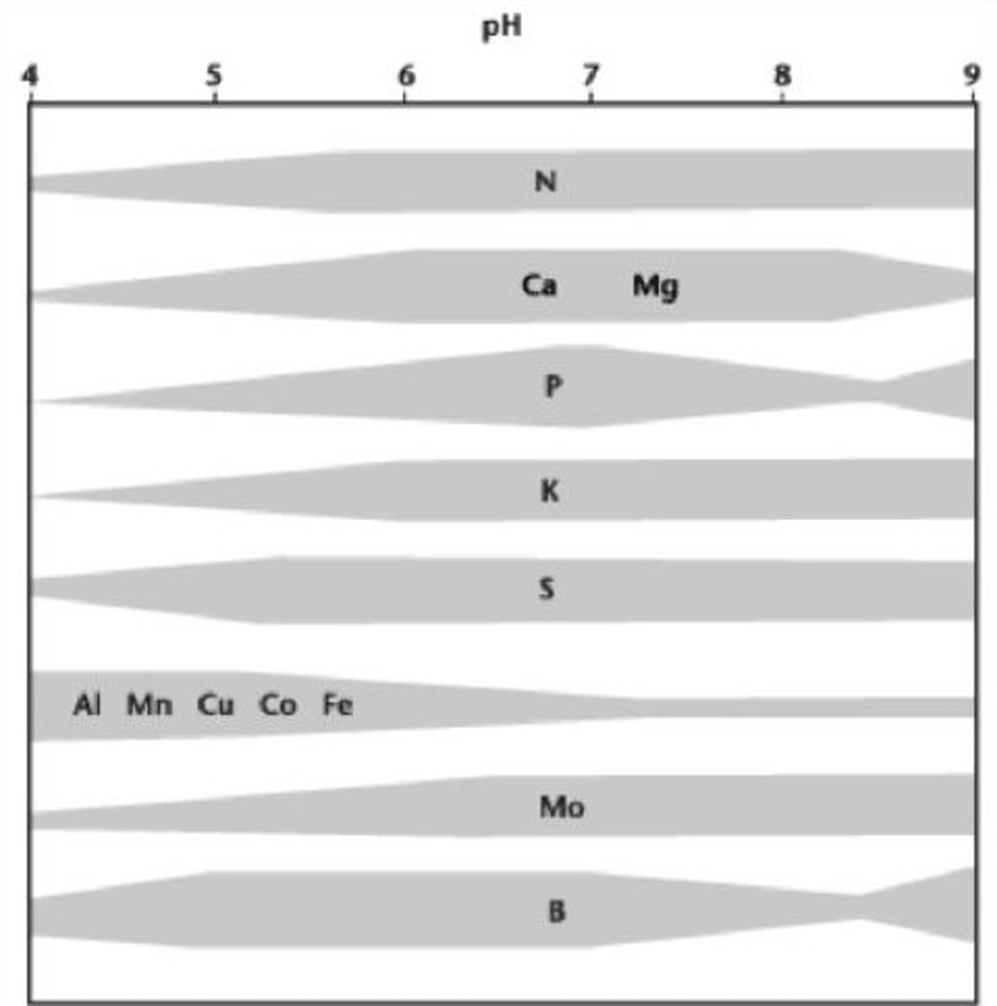
Acid soils often contain soluble forms of aluminum and manganese. As soil acidity drops below pH 5.5, soluble aluminum and manganese increase to toxic levels. Aluminum toxicity restricts root growth, phosphorus uptake and translocation within the plant. While the primary effect of aluminum toxicity is at the root level, the more visible foliar injury is due to nutrient deficiency, drought (due to poor root penetration) or pathogens, which are more pronounced on soils with aluminum toxicity. Manganese toxicity causes chlorosis on leaf margins and cupping of leaves in canola. Aluminum and manganese often reduce the yields of crops grown on acid soils12. The graph below shows the general relationship between pH of mineral soil and the availability of various elements.
Manitoba soil pH levels are outlined in the pH status of Manitoba Soils map. These numbers may be less than soil test numbers (because they are done with the saturated paste method). Most commercial soil test labs use a 1:1 or 2:1 soil: water ratio method which produces a pH reading about 0.5 points greater than the saturated paste.
Liming
Research has shown that canola yields on strongly acid soils (pH below 5.5) can be substantially increased by lime application. On slightly acid (pH 6.1 to 6.5) and moderately acid (pH 5.6 to 6.0) soils, liming will have a minor effect on canola yields13,14,15,16.
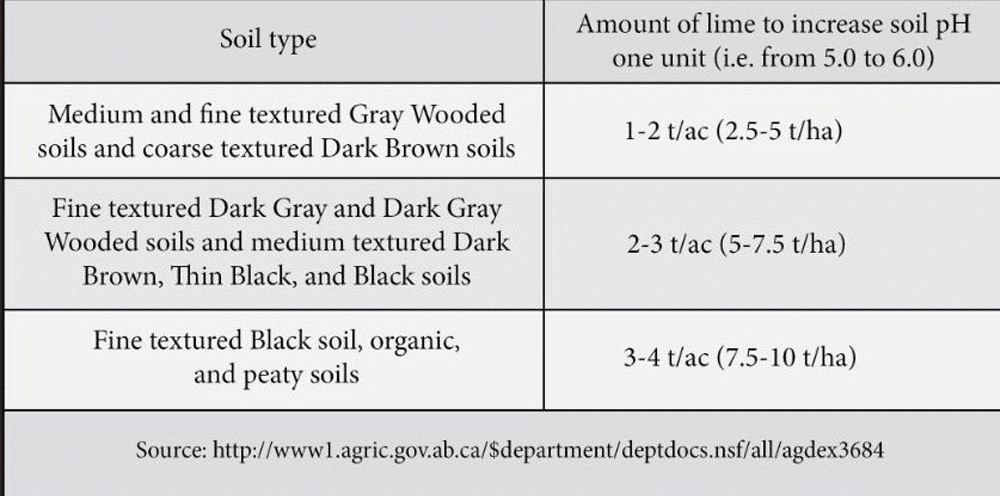
A soil test is the only reliable way of determining whether soil is acid or not, and a lime requirement test should be used to determine the rate of lime to apply. Rates could be as high at 10 tonnes per hectare, depending on the soil type. The pH increase is limited to the top soil layer when the lime was incorporated17.
Soil fertility benefit:
The increase in soil pH resulting from lime application provides a more favourable environment for soil microbiological activity that increases the rate of release of plant nutrients, particularly nitrogen. Reduced soil acidity following liming also increases the availability of several other plant nutrients, notably phosphorus. A crop takes up only about 20 per cent of fertilizer phosphorus in the application year.
The remainder is fixed in the soil in various degrees of availability for succeeding crops. In a soil with less than pH 6.0, the fixed phosphorus is retained in less available forms than in slightly acid and neutral soils (pH 6.1 to 7.5). Phosphorus availability is reduced because of the formation of relatively insoluble compounds through reactions of phosphorus with iron and aluminum18.
Therefore, one of the benefits of liming acid soils is the increased utilization of residual fertilizer phosphorus by crops. However, in soils with above pH 7.0, fertilizer phosphorus availability is reduced due to reactions with calcium and magnesium, so liming will not be beneficial.
Soil texture benefit:
Liming may improve the physical properties of some medium and fine-textured soils (particularly Grey and Dark Grey Wooded soils). Research at the Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More research centre in Beaverlodge, Alberta16 showed that liming improved the stability of soil aggregates and therefore, soil structure of Grey Wooded soils. Soils with a stable soil structure are well-aerated, less prone to crusting after intense precipitation, have a high rate of water infiltration and allow for good germination and establishment of small-seeded crops like canola.
Aluminum and manganese benefit:
On strongly acid (pH 5.1 to 5.5) and very strongly acid (pH <5.1) soils, liming reduces soluble aluminum and manganese to non-toxic levels and increases yield. In a three-year study at Beaverlodge, liming a soil raised the pH from 5.0 to 6.5, increasing plant dry matter production and increasing canola yields by 37 per cent in tilled and 17 per cent in no-tillA type of tillage management technique where soil is undisturbed from harvest through planting, seeding with narrow knives/hoes or discs. More soils16. The benefit of no-tillA type of tillage management technique where soil is undisturbed from harvest through planting, seeding with narrow knives/hoes or discs. More systems was lower because these systems have been observed to accelerate soil acidification19,20.
The increased growth resulting from liming was likely due to a decrease in brown girdling root rot, reduced weed populations, increased pH-related changes in soil fertility, and potentially other factors. Aluminum stress predisposes plants to infection by brown girdling root rot (Rhizoctonia solani Kuhn) due to reduced root vigourSeed properties that determine the potential for rapid uniform emergence and development of normal seedlings under a wide range of field conditions. More. In the Beaverlodge study from 1993-95, brown girdling root rot was observed in all plots of each treatment. Liming significantly reduced the severity of this disease in the tilled soil, but not in the no-tillA type of tillage management technique where soil is undisturbed from harvest through planting, seeding with narrow knives/hoes or discs. More system16.
Research shows that the benefits of a single application of lime can last for up to 30 years21. Liming is a substantial investment, although by-product liming materials from municipal and industrial facilities may provide an alternative low-cost liming material for farmers. It is important to identify the extent and severity of an acid soil problem. The most reliable method of identifying an acid soil problem is through soil tests. With careful sampling of fields, soil tests can determine the extent and severity of soil acidity, the rate of lime required and provide an estimate of crop response to lime.
Wood ash versus lime:
Wood ash, the calcium carbonate-rich residue remaining from the combustion of bark, sawdust, and yard waste for the energy generation of forestry product operations, is an effective liming material on acid agricultural soils17. Many of these forestry operations are in the northern Prairies, where a large percentage of highly acidic soils are located. Given the high cost of transporting agricultural lime to these regions, wood ash should be an appropriate and economic substitute for lime17. A Peace River region study compared wood ash, lime and untreated plots, where treatments were applied on an equivalent calcium carbonate basis. Canola yield (and barley and pea yields) were higher in treated plots than in untreated plots, and in most cases, canola yield was higher in plots treated with wood ash than those treated with lime. This may be due to the greater solubility of wood ash, and the greater amounts of nitrogen and phosphorus in wood ash compared to lime17. Wood ash also contains various other nutrients, including potassium, calcium, magnesium, manganese, copper, zinc, boron and molybdenum.
Crops vary greatly in their tolerance to acidity:
The numbers in the table represent the lowest pH these crops will tolerate.
| Table 1. Acidity tolerance of various crops |
Organic matter
Organic matter is anything in the soil composition that is living or was once alive. It includes plant and animal remains, soil organisms, and substances produced by soil microbes. Organic matter is the material besides clay, silt and sand that gives rich soil its dark colour, sponginess and earthy smell.
Organic matter is an essential component of soil because it:
- holds soil particles together and stabilizes the soil, thus reducing the risk of erosion.
- aids crop growth by improving the soil’s ability to store and transmit air and water.
- stores and supplies many nutrients needed for the growth of plants and soil organisms.
- prevents compaction, making for a better seedbed
- retains carbon from the atmosphere.
- reduces the negative environmental effects of pesticidesPesticides (herbicides, insecticides or fungicides) used to protect against or reduce the amount of damage caused by weeds, pest insects or plant diseases. More, heavy metals, and many other pollutants22.
To get an estimate of organic matter content in the Prairies, see the Organic carbon status of Manitoba soils map and the Organic matter content of Alberta soils map.
Over hundreds and thousands of years, soil biological and chemical components along with inorganic components contributed to the degradation and synthesis of complex soil organic matter and humic substances. Soil organic matter and humic substances contain at least 10 chemical classes of compounds, including proteins, lipids, carbohydrates and lignins23.
Soil tests measure only the organic carbon component of soil organic matter, and then multiply by a factor of 1.72 to determine the organic matter reading provided with soil test results24. With this estimate, growers can then estimate, roughly, the pounds per acre of nitrogen, phosphorus and sulphur that organic matter will provide to a crop in a given year.
Nitrogen: Mineralization of soil organic matter can generate six to 20 pounds of available nitrogen per acre for each percentage point of organic matter. For example, soil with four per cent organic matter will supply 24 to 80 pounds of nitrogen through the growing season. Soil with eight per cent organic matter will supply 48 to 160 pounds of nitrogen per season. Warm conditions, ample moisture and high microbial activity all increase the rate of mineralization. Mineralization can also increase with the number of years utilizing minimum tillage or direct seedingA seeding method where the crop is sown into the field without any previous spring tillage operations. More.
Phosphorus: Each percentage point of organic matter provides up to five pounds per acre of phosphorus. For example, soil with four per cent organic matter will provide up to 20 pounds of phosphorus through the season, and soil with eight per cent organic matter will provide up to 40 pounds.
Sulphur: Each percentage point of organic matter provides two to three pounds per acre of available sulphur. For example, soil with four per cent organic matter will provide eight to 12 pounds of sulphur throughout the season, and soil with eight per cent organic matter will provide 16-24 pounds.
Micronutrients: Soil organic matter complexes can also supply zinc, chromium, iron, and other inorganic elements to provide crops with many of the micronutrients required.
Given the many benefits soil organic matter provides for a crop, growers do benefit from maintaining and increasing soil organic matter levels. Soil organic matter decreased dramatically across the Prairies in the decades after the plains were first broken for agriculture. Heavy tillage and summerfallow accelerated this decline, but in recent decades continuous cropping, conservation tillage and higher yielding crops have stopped and in many cases reversed this trend25.
Soil organic matter can be compared to a bank account. To increase its size you need to ensure that additions to the account exceed the rate of spending. Practices to build soil organic matter include:
- limiting erosion through minimum tillage and continuous cropping.
- retaining crop residue, especially roots, in the soil and residue on the soil surface.
- increasing yields by applying adequate nutrients. Higher yielding crops tend to have larger root systems and above ground biomass to build up organic matter.
- Adequate nutrient rates also reduce or stop the mining of organic matter reserves, which will reduce organic matter over time.
- Adding animal manure, green manure, sewage sludge, wood chips, or peat. Soils amended with manure accumulated more organic matter than soils amended only with inorganic fertilizer26.
In a long-term wheat yield study on the Prairies, the highest average grain yield was maintained in plots that lost the least amount of soil. When topsoil is lost, it takes organic matter with it, which impacts the soil’s ability to produce top yields26. This long-term study (which took place at sites on Brown Chernozemic and Grey Luvisol soils) also reported the highest average grain yield in fertilized continuous wheat plots in the Brown Chernozemic soil. Interestingly, those soils also received the lowest average annual precipitation (359 millimetres) compared to other sites in the study. This suggests that measures to conserve soil organic matter can provide a buffer against lower rainfall by improving moisture use efficiency.
Not many studies have looked at the connection between organic matter levels and canola yields. In general, canola grown in Western Canada tends to yield best on high organic matter soils, especially when conditions permit a long growing season and with not too many high temperature days27.
Soil salinity
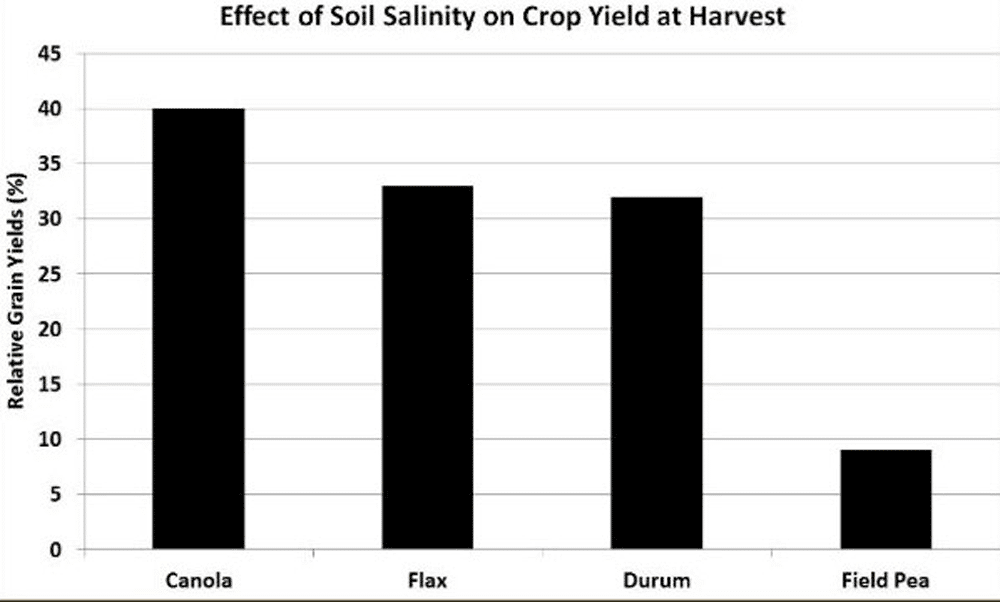
All soils contain soluble salts. When the levels are sufficient to harm plants, the soils are referred to as saline. Crops will vary in their tolerance to salinity. Canola is considered moderately tolerant to salt and sodium, and can tolerate salinity up to levels of five to six deciSiemens per metre (dS/m).
Saline soils are highly fertile and often very high in organic matter. This may be caused by the reduced crop yields in these areas, where nutrients have built up over years of blanket nutrient application. Zone-mapping a field based on electrical conductivity (ex. using Veris or EM38) to develop a variable rate prescription could improve this situation by automatically shutting off fertilizer applications when passing over saline areas. This would result in reduced nutrient buildup, increased fertilizer use efficiency, and fewer greenhouse gas emissions.
Salinity is not just a salt issue. Salinity is the result of excess groundwater moving downward and laterally through the soil, dissolving and transporting soluble salts28. Large areas in the Canadian Prairies contain soil that is relatively high in soluble salts. The redistribution of these soluble salts by groundwater movement causes some areas to become excessively saline. When drainage is effective, the salts are washed down through the soil and out of the root zone. When drainage is unsuccessful (ex. in low, flat or areas with depressions, or in areas where roads or other construction interferes with normal drainage), the water table rises. When the water table rises to within one metre (three feet) of the surface, water and salts can rise to the surface by capillary action. The water evaporates and the salts accumulate at the surface.
Salinity also occurs in saline seeps on hill slopes, often part way up the slope on very long and high hills. Rainfall on the upslope part of the hill (recharge area) moves down through the soil, picking up salts on the way. The water not used by crops moves down until it reaches an impermeable layer, which impedes its progress. The water then flows laterally (seeps) until it reaches a position lower down the slope where the water table is closer to the surface. There, seep water causes the water table to rise allowing the salt-laden water to move up to the soil surface by capillary action. The seep phenomenon can take place over relatively short distances within the same field or over distances of several miles. Salinity levels vary widely across a saline seep. Salinity also varies from spring to fall. Salinity usually appears on the soil surface just after spring thaw28.
The dominant salts in Prairie saline seeps are calcium (Ca), magnesium (Mg), and sodium (Na) cations and sulphate (SO4) anions. If sodium levels are high or not balanced with the calcium and magnesium , soil tilth can also be affected. The positively charged sodium cations attach to the negatively-charged clay particles in the soil, causing the soil to be sticky when wet and hard and impermeable when dry.
Saline soils can be recognized by spotty growth of crops or by white crusts of salt, which accumulate on the soil surface, usually in low-lying areas. Streaks of salt may be present in the soil even though white crusts may not appear on the surface. In many saline soils, it is not possible to see the salts and a laboratory soil analysis must be used to confirm their presence.
Plants growing in saline areas may develop a blue-green tinge. Salt-tolerant weeds such as Russian thistle, kochia, wild barley and goosefoot species are commonly found in areas of high salt concentration28.
The process of salt buildup is reduced when plants intercept the upward flow of saline groundwater and reduce the amount that reaches the soil surface. When plants are not present, almost all of the water loss takes place by evaporation at the soil surface. Summerfallow is a major contributor to salinity as it encourages a buildup of a water table. Water not used by crops accumulates in the subsoil, eventually coming to the surface laden with salts. These salts interfere with seed germination and crop establishment.
All irrigation water contains some salt. Over an extended period of irrigation where soil drainage is inadequate, salts accumulate in the soil and a salinity problem may develop. Water used in the major irrigation areas of Alberta and Saskatchewan is of high quality with little risk of salt buildup. Seepage or high water tables resulting from over irrigation cause most salinity under irrigation.
Effects of salinity on canola growth and yield
Some plant species tolerate high levels of salinity while others can tolerate little or no salinity. Therefore salt tolerance refers to the relative growth of plants in the presence of salinity. Salt-tolerant plants avoid toxicity by the sequestration of the sodium (Na+) and chloride (Cl-) ions into either vacuoles or roots by a salt-exclusion mechanism29. Salt tolerances are usually given in terms of stage of plant growth over a range of electrical conductivity levels. The degree of salinity or total soluble salt concentration in a soil is routinely measured by soil testing laboratories with a conductivity test. Electrical conductivity (EC) is the ability of a solution to transmit an electrical current. The units of conductivity are usually given in deciSiemens per metre (dS/m). The following Table categorizes salinity into general ranges from non-saline to very severely saline. These values are used for plant selection for saline soils.
Table 2. Salinity rating and electrical conductivity value
Table 2. Salinity rating and electrical conductivity value
dS/m = deciSiemens per metre
Excess soluble salts cause osmotic stress and ion toxicity in plant cells. Plants need both water and the nutrients dissolved in water for proper growth. The sap in plant roots contains salt that attracts water into the plant via osmotic pressure. Dissolved salts in the soil increase the osmotic pressure of the soil solution. This decreases the rate at which water from the soil will enter the roots. If the soil solution becomes too concentrated, plants will slowly dehydrate, lose turgor and starve, even though the supply of water and nutrients in the soil may be quite high. If the salt content of the soil water becomes too saturated, water may actually be withdrawn from the roots. Osmotic stress also results from desiccation and, therefore, is a common component of both drought and salt stress. High concentration of certain salts in the soil may also be toxic because some plants may absorb an excess amount of the salt, reducing growth or causing death.
Canola is considered moderately tolerant to salt and sodium, and can tolerate moderate salinity up to levels of five to six deciSiemens per metre. Canola is more sensitive to salinity during germination and emergence than later growth stages30.
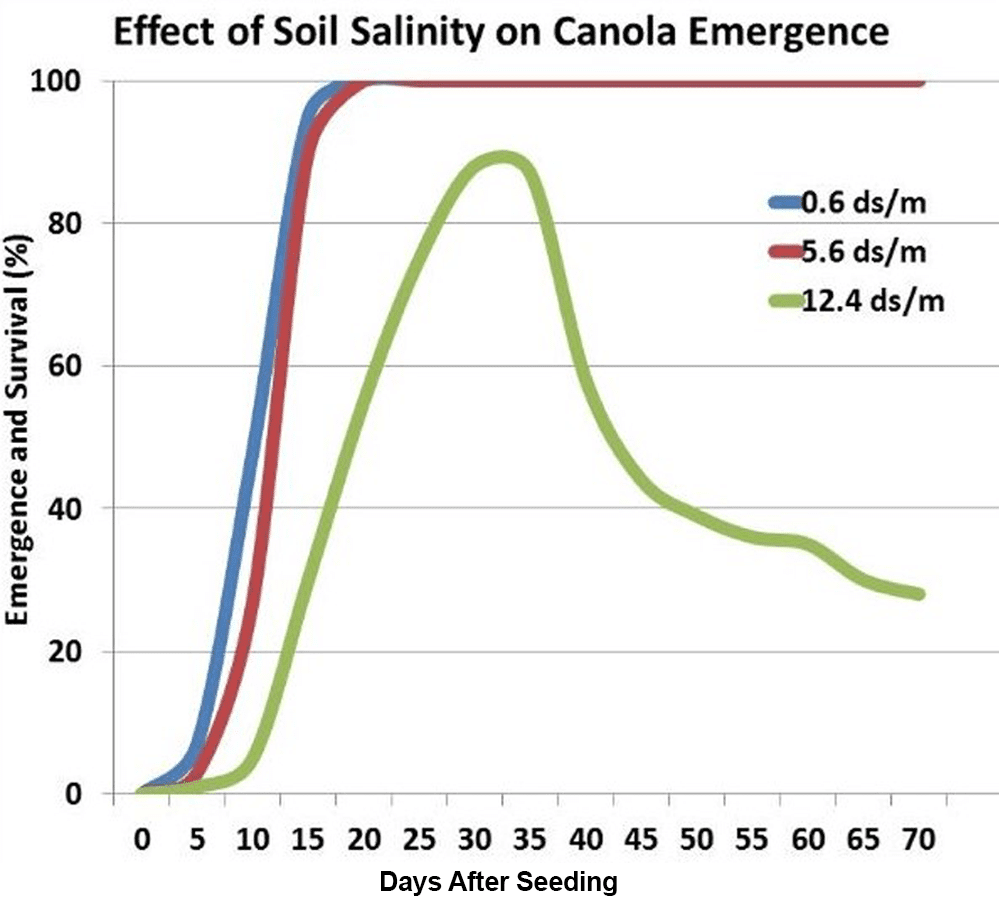
In a salinity study by the Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More in Swift Current, Saskatchewan, increasing levels of soil salinity reduced germination, emergence and emergence rate31. This greenhouse study showed that the per cent emergence and plant survival was not significantly affected until salinity exceeded 5.6 deciSiemens per metre. Above this value, the number of days from seeding to initial emergence increased. With severe salinity, plants emerged rapidly, but over time the percentage that survived dropped off.
Salinity under both moderate and severe conditions reduces average plant height, shoot and root biomass, number of leaves, leaf area, evapotranspiration, and crop yield at harvest. For example, the average harvest height in the study shown in this graph averaged 167, 132 and 84 centimetres (66, 52 and 33 inches), respectively for the 0.6, 5.6 and 12.4 deciSiemens per metre treatments. Root growth is also hindered at salinity above six deciSiemens per metre. At 5.6 deciSiemens per metre, canola yields in this study were reduced by 60 per cent and with severe salinity, canola yields were reduced by 80 per cent. However, several other research studies have shown there was little effect on canola yields with salinity levels up to 10 deciSiemens per metre.
Soil tests will show the extent of a salinity problem. When sampling, collect representations from both affected and non-affected areas of the field. Perform analyses for electrical conductivity, pH, cationA positively-charged ion (ion with a net positive charge). More base saturation, and calcium, magnesium, sodium and organic matter content.
When sodium levels are high in relation to calcium and magnesium in the soil, another soil problem occurs. These soils are very sticky and slippery when wet, and are very hard, cloddy and prone to crusting when they dry. The sodium adsorptionThe process of electrical attraction that holds cations to negative surfaces of soil colloids. More ratio (SAR) is the ratio of sodium to the beneficial soil structural cations, calcium and magnesium. When the SAR value exceeds 13, the soil is referred to as sodic. If the SAR exceeds 13 and the EC is greater than four, it is considered a saline sodic soil. Use a soil test to determine the SAR of soils.
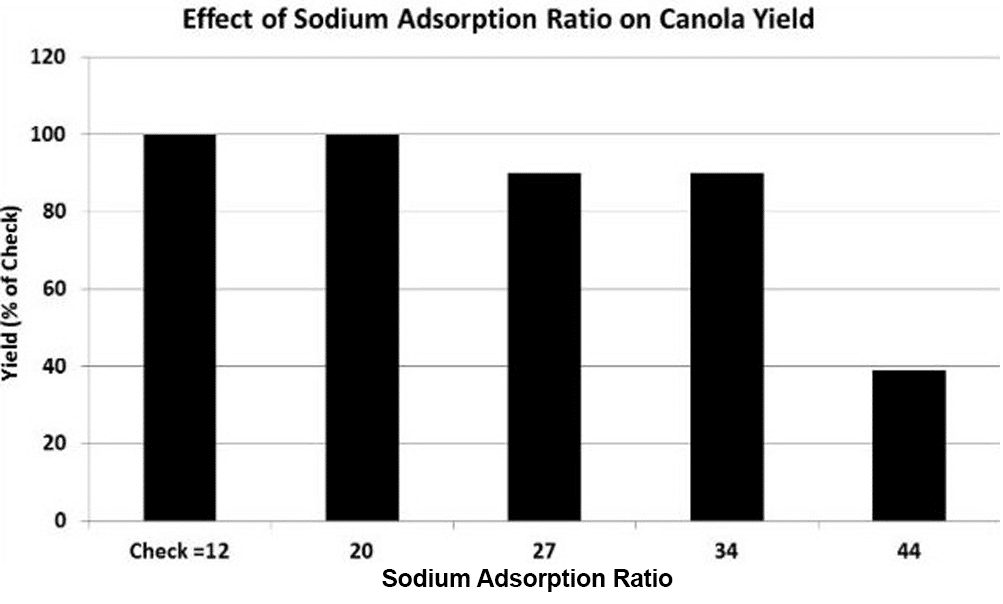
An Argentinian study found that sodium, even with no soil crusting, can cause a slight reduction in canola emergence at SAR values of 20 and increase to a 60 per cent reduction at SAR values above 34. At SAR values greater than 20, main stem growth and yield decreased slightly but an increase in the number of secondary branches compensated for this reduction. Reductions in number of stems, seeds per pod on the main stem, pods per stem and canola yields tend to decrease dramatically when SAR values exceed 34. This may be partly due to induced calcium deficiency from excessive sodium32.
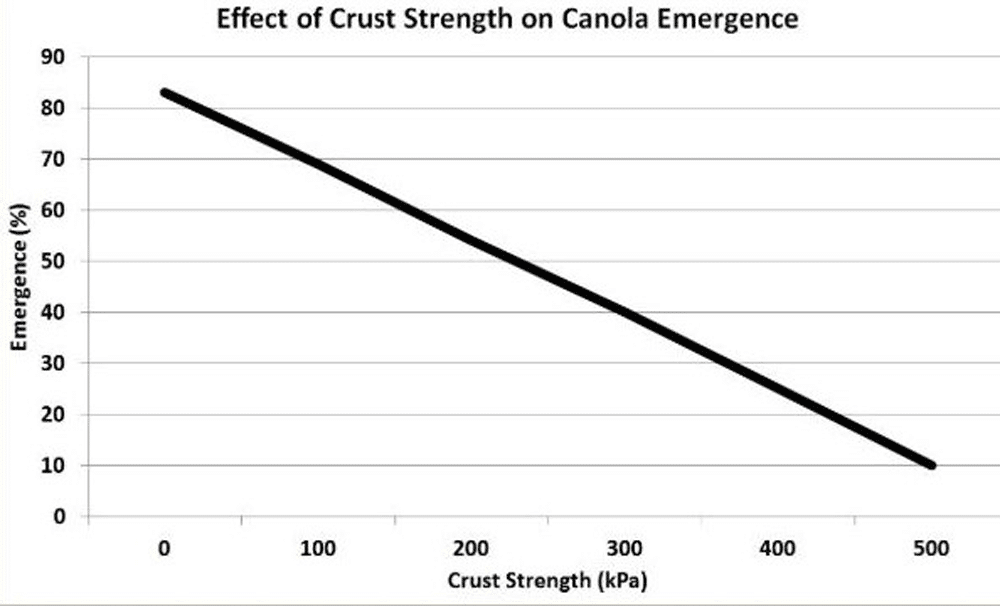
However, the most important effect of soil sodium is through increased crusting that reduces emergence. The amount of sodium required to affect emergence is much higher than that necessary for clay dispersion and crusting. The Argentinian study showed SAR values of eight and even lower can encourage clay dispersion and crusting when raindrops impact the soil surface. Canola has a small seed size and at germination the cotyledons are pushed through the soil to the surface, making emergence more difficult in crusted soils. Crusting is also affected by soil particle size distribution as a high proportion of fine particles enhance soil susceptibility to crusting. Very fine seedbeds, resulting from excessive tillage, increase the susceptibility of a soil to form a crust. Crust strength will greatly influence emergence.
On soils with moderate salinity, use continuous cropping or at least lengthen the rotation as much as possible. Crops use the water rather than allowing it to move deeply into the soil or accumulate near the soil surface and contribute to salt movement.
On soils with moderate salinity, use continuous cropping or at least lengthen the rotation as much as possible. Crops use the water rather than allowing it to move deeply into the soil or accumulate near the soil surface and contribute to salt movement.
Use shallow tillage and maintain all possible crop residues at the soil surface. Deep tillage may, in many cases, simply bring more salts to the surface and make problems worse. Early in the spring seed canola shallow so that seeds may germinate when surface salt levels are temporarily lowered.
If soil salinity has already reached the stage where cereal and oilseed crops cannot be grown, consult qualified soil specialists on management procedures to combat and reclaim saline land. Salinity can be removed mechanically, but it requires irrigation on top and tile drains underneath to wash salinity out of the soil and drain it away.
Environmental sensitivity
Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More has an explanation on soil texture and water quality related to sustainable agriculture.
Fertilizer runoff
The two key nutrients for runoff risk are nitrogen and phosphorus. Nitrates and phosphates in water systems can promote the growth of aquatic plants and algae, which can reduce the aesthetic value of lakes and rivers and can reduce oxygen levels in the water, threatening fish numbers. When algal blooms die and decompose, oxygen in the water is depleted and fish and other aquatic organisms suffocate33.
Nitrogen: The nitrate form of nitrogen — a form that plants take up — is very mobile and can leach through the root zone and get into ground water. Coarse textured (sandy) soils increase the risk of nitrate leaching to ground water, especially in depressional areas of the landscape and in regions with large amounts of precipitation.
Phosphorus: On the relatively flat Canadian Prairies, phosphorus is more likely to enter waterways as dissolved phosphorus in surface-water runoff, and less likely through soil erosion of particulate phosphorus. Measures to reduce soil erosion, while providing other clear benefits, do very little to reduce phosphorus loading in Prairie lakes and rivers. Phosphate runoff is not generally regarded as a major issue on the Prairies, but this risk is substantial where tile drainage is used in areas where large amounts of phosphate have been or are being applied (ex. tile-drained, heavily-manured sandy soils). The key management tool to prevent dissolved phosphorus runoff is to avoid manure and fertilizer application on frozen soils34.
To limit fertilizer runoff:
- Do not apply fertilizer or manure on frozen soils. When manure is applied on frozen soils or when nitrogen fertilizer is applied on snow, spring runoff will remove many of those nutrients before they can enter the soil and be available to crops. These nutrients end up in rivers and lakes.
- Match nitrogen rates to crop uptake. Applying more nitrogen than a crop can use could increase surface water and groundwater contamination.
- Nitrogen fertilizer products that slow the rate of fertilizer conversion to nitrate may also help reduce leaching losses.
- Test manure for nitrogen and phosphorus content and apply manure at rates that match crop use.
Pesticide runoff
Overland runoff and leaching of pesticidesPesticides (herbicides, insecticides or fungicides) used to protect against or reduce the amount of damage caused by weeds, pest insects or plant diseases. More into water systems is more common in certain soil types.
Sandy soil. Water moves through sandy soil quicker than through soils with higher clay content and smaller pores. Pesticide mixing and tank rinsing should be avoided on sandy soils, if possible.
Clay soil. While clay soils may have lower rates of leaching, clay soils may result in higher rates of surface runoff losses. Clay soils are more prone to compaction and crusting, which limit infiltration and increase runoff.
Soil health
Soil health is a general term that can include good soil organic matter levels, granular soil structure, nutrient release and microbial activity. Soil texture is more difficult to change.
A soil’s ability to produce crops can be sustained over time, or degraded, or improved. By following practices outlined in the previous content, growers can build soil organic matter, maintain soil nutrient levels, and keep salinity in check. These steps, as well as reducing soil erosion, will contribute to overall soil health.
- Canadian Society of Soil Science. 2020. Soils of Canada. Available online at: soilsofcanada.ca [June 2020] [↩]
- http://publications.gc.ca/collections/collection_2015/aac-aafc/A53-1391-1980-eng.pdf [↩]
- Lickacz, J. 1993. Management of Solonetzic Soils. Alberta Agriculture, Food & Rural Development publication Agdex 518-8 [↩] [↩] [↩]
- Toogood, J.A. and Cairns, R.R. (co-editors). 1973. Solonetzic soils technology and management. Bulletin B-73-1. University of Alberta. [↩]
- Ross McKenzie, research scientist, agronomy, Alberta Agriculture and Rural Development, personal communication, March 2012 [↩]
- Rob Dunn, Agriculture Land Management Specialist, Alberta Agriculture and Rural Development, personal communication, March 2012 [↩]
- Grevers, M. C. J. and De Jong, E. 1993. Soil structure and crop yield over a 5-year period following subsoiling Solonetzic and Chernozemic soils in Saskatchewan. Canadian Journal of Soil Science, 73(1), 81-91. doi.org/10.4141/cjss93-008 [↩]
- Grevers, M. C. J. and De Jong, E. 1993. Soil structure and crop yield over a 5-year period following subsoiling Solonetzic and Chernozemic soils in Saskatchewan. Canadian Journal of Soil Science. 73 (1): 81-91. doi.org/10.4141/cjss93-008 [↩]
- Penney, D.C., Nyborg, M., Hoyt, P.B., Rice, W.A., Siemens, B. and Laverty, D.H. 1977. An assessment of the soil acidity problem in Alberta and northeastern British Columbia. Can. J. Soil Sci. 57:157-164. doi/pdfplus/10.4141/cjss77-020 [↩]
- Penney, D.C., Nyborg, M., Hoyt, P.B., Rice, W.A., Siemens, B. and Laverty, D.H. 1977. An assessment of the soil acidity problem in Alberta and northeastern British Columbia. Can. J. Soil Sci. 57:157-164. doi.org/10.4141/cjss77-020 [↩]
- Marschner, H. Mechanisms of adaptation of plants to acid soils. Plant Soil 134, 1–20 (1991). doi.org/10.1007/BF00010712 [↩]
- Delhaize, EaPRR & Ryan, Peter. (1995). Aluminum Toxicity and Tolerance in Plants. Plant physiology. 107. 315-321. doi.org/10.1104/pp.107.2.315. [↩]
- Nyborg, M. and Hoyt, P.B. 1978. Effects of soil acidity and liming on mineralization of soil nitrogen. Canadian Journal of Soil Science. 58(3): 331-338. doi.org/10.1016/S0038-0717(97)00094-1 [↩]
- Edmeades, D. C., Judd M. and Sarathchandra, S. U. 1981. The effect of lime on nitrogen mineralization as measured by grass growth. Plant and Soil. 60(2): 177-186. doi.org/10.1007/BF02374102 [↩]
- Haynes, R.J. and Naidu, R. 1998. Influence of lime, fertilizer and manure applications on soil organic matter content and soil physical conditions: a review. Nutrient Cycling in Agroecosystems. 51(2): 123-137. https://doi.org/10.1023/A:1009738307837 [↩]
- Arshad, M.A., Gill, K.S., Turkington, T.K. and Woods, D.L. 1997. Canola root rot and yield response to liming and tillage. Agron. J. 89:17-22. doi.org/10.4141/S04-023 [↩] [↩] [↩] [↩]
- Arshad, M. A. Soon, Y. K. Azooz, R. H., Lupwayi, N. Z. and Chang, S. X. 2012. Soil and Crop Response to Wood Ash and Lime Application in Acidic Soils. Agronomy Journal. 104(3): 715-721. Doi.org/10.4141/CJSS08007 [↩] [↩] [↩] [↩]
- https://www1.agric.gov.ab.ca/$department/deptdocs.nsf/all/agdex3684/$file/534-1.pdf [↩]
- Warren, D. 1983. Organic Carbon, Nitrogen, and Phosphorus Concentrations and pH in Soil Profiles as Affected by Tillage Intensity1. Soil Science Society of America Journal. 47. 10.2136/sssaj1983.03615995004700010021x [↩]
- Arshad, M.A., Schnitzer, M., Angers, D.A. and Ripmeester, J.A. 1990. Effects of till vs no-tillA type of tillage management technique where soil is undisturbed from harvest through planting, seeding with narrow knives/hoes or discs. More on the quality of soil organic matter. Soil Biology and Biochemistry. 22(5): 595–599. Doi.org/10.1016/0038-0717(90)90003-I [↩]
- Beckie, H., & Ukrainetz, H. (1996). Lime-amended acid soil has elevated pH 30 years later. Canadian Journal of Soil Science, 76, 59-61. doi:10.4141/cjss96-009 [↩]
- Gregorich, E.G., Angers, D.A., Campbell, C.A., Carter, M.R., Drury, C.F., Ellert, B.H., Groenevelt, P.H., Holmstrom, D.A., Monreal, C.M. Rees, H.W., Voroney, R.P. and Vyn, T.J. 1995. The Health of Our Soils: Toward sustainable agriculture in Canada. Changes in Soil Organic Matter (Chapter 5). Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More. Retrieved from: https://atrium.lib.uoguelph.ca/xmlui/bitstream/handle/10214/15123/FDMR_Health_of_Our_Soils.pdf [↩]
- Schnitzer, M. and Monreal, C.M. 2011. Chapter three–Quo vadis soil organic matter research? A biological link to the chemistry of humification. Adv. Agron. 113: 143– 217. Retrieved from https://doi.org/10.1016/B978-0-12-386473-4.00003-8 [↩]
- ALS Labs, Saskatoon, personal communications with Dianne Kemppainnen [↩]
- Gregorich, E.G., Angers, D.A., Campbell, C.A., Carter, M.R., Drury, C.F., Ellert, B.H., Groenevelt, P.H., Holmstrom, D.A., Monreal, C.M. Rees, H.W., Voroney, R.P. and Vyn, T.J. 1995. The Health of Our Soils: Toward sustainable agriculture in Canada. Changes in Soil Organic Matter (Chapter 5). Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More. Retrieved from: https://atrium.lib.uoguelph.ca/xmlui/bitstream/handle/10214/15123/FDMR_Health_of_Our_Soils.pdf [↩]
- Monreal, C. M., Zentner, R. P. and Robertson, J. A. 1997. An analysis of soil organic matter dynamics in relation to management, erosion and yield of wheat in long-term crop rotation plots. Canadian Journal of Soil Science. 77(4): 553-563. doi/10.4141/S95-076 [↩] [↩]
- Harker, K. N., O’Donovan, J. T., Turkington, T. K., Blackshaw, R. E., Lupwayi, N. Z., Smith, E. G., Klein-Gebbinck, H., Dosdall, L. M., Hall, L. M., Willenborg, C. J., Kutcher, H. R., Malhi, S. S., Vera, C. L., Gan, Y., Lafond, G. P., May, W. E., Grant, C. A. and McLaren, D. L. 2012. High-yield no-tillA type of tillage management technique where soil is undisturbed from harvest through planting, seeding with narrow knives/hoes or discs. More canola production on the Canadian prairies. Can. J. Plant Sci. 92: 221–233. doi.org/10.4141/cjps2011-125 [↩]
- Marla Riekman, Manitoba Agriculture, Food, and Rural Initiatives. 2017. Personal communication [↩] [↩] [↩]
- Ashraf, M. and McNeilly, T. 2004. Salinity tolerance in Brassica oilseeds. Crit. Reviews in Plant Sci. 23:157-174. doi.org/10.1080/07352680490433286 [↩]
- Steppuhn, H., Volkmar, K.M. and Miller, P.R. 2001. Comparing canola, field pea, dry bean, and durum wheat crops grown in saline media. Crop Science 41:1827-1833. doi.org/10.2135/cropsci2001.1827 [↩]
- Steppuhn, H, Volkmar, K.M. and Miller, P.R. 2001. Comparing canola, field pea, dry bean, and durum wheat crops grown in saline media. Crop Science 41:1827-1833. doi.org/10.2135/cropsci2001.1827. [↩]
- Carter, M.R., Webster, G.R. and Cairns, R.R. 1979. Calcium deficiency in some Solonetzic soils of Alberta. J. Soil Sci. 30:161-174. doi.org/10.1111/j.1365-2389.1979.tb00974.x [↩]
- Alberta Agriculture and Rural Developoment. Managing Nitrogen to Protect Water Quality. 2000. Agri-Facts: Agdex 576-1. Retrieved from: https://www1.agric.gov.ab.ca/$department/deptdocs.nsf/all/agdex928/$file/576-1.pdf [↩]
- Salvano, E., Flaten, D.N., Rousseau, A.N. and Quilbe, R. (2009), Are Current Phosphorus Risk Indicators Useful to Predict the Quality of Surface Waters in Southern Manitoba, Canada?. J. Environ. Qual., 38: 2096-2105. doi:10.2134/jeq2008.0159 [↩]