Beneficial insects provide valuable contributions to canola production, both in terms of pollination of crops and predation or parasitism of insect pests. Most of the thousands of insects found in canola fields are either beneficial or nonthreatening. Observing economic thresholds for pest management and protecting pollinators and parasitoids while spraying canola fields is vitally important. Accurately identifying beneficial insects (in addition to the pest insects they may be feeding on) as well as maintaining undisturbed habitat for them to maintain healthy populations are management strategies to utilize in optimizing canola production.
Important tips for best management of beneficial insects

- Use insecticides judiciously.
- Only spray insecticides when the pests have clearly surpassed the economic thresholds.
- Use trap crops or strips to lure pest to a localized area where targeted insecticide application can be made.
- Management strategies in the field that may increase populations of some beneficial insects and reduce mortality of some parasitoids include reducing tillage, leaving tall stubble to improve overwinter survival, and intercropping.
- Perennial crops like alfalfa offer a stable environment and may increase the overall populations of beneficial insects in the area.
- Time and choose insecticidal applications to minimize impacts on pollinators.
- Choose insecticides with low hazard potential to bees.
- Discuss spray plans with beekeepers prior to spraying.
- Alert the spray applicator to the exact location of the beehives within the intended spray area.
- Give careful attention to wind direction and velocity in relation to bee yard locations.
- Do not spray a crop in flower unless absolutely necessary.
- If spraying a crop in flower is necessary, do the spraying when there will be minimal bee activity in the fields.
- Explore research on beneficial insects on the Canola Research Hub.
- Learn scouting tips for beneficial insects from Field Heroes.ca.
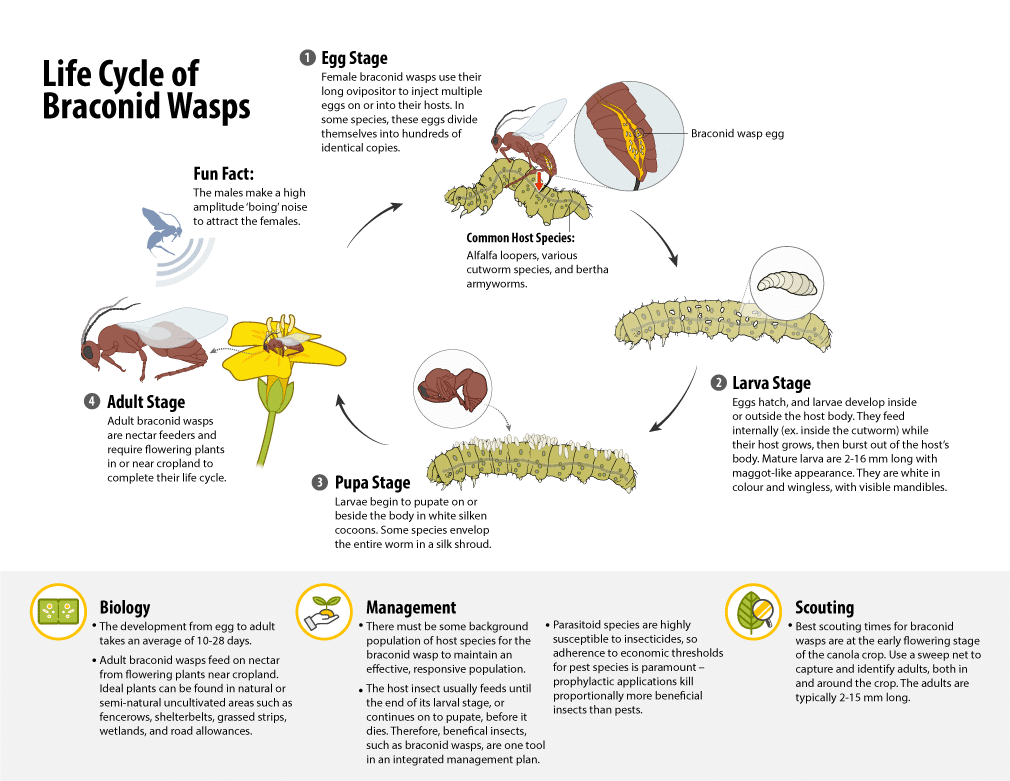
Beneficial insects – Natural enemies of pest insects
Natural enemies of pest insects provide an important service to farmers in the form of biological control – a key component of an integrated pest management system. Relatively few insect species actually classify as a pest and many are beneficial.

Honey and leaf-cutter bees also provide a valuable service in the production of hybrid canola. In addition, approximately 80 per cent of Canada’s honey crop is from canola1.
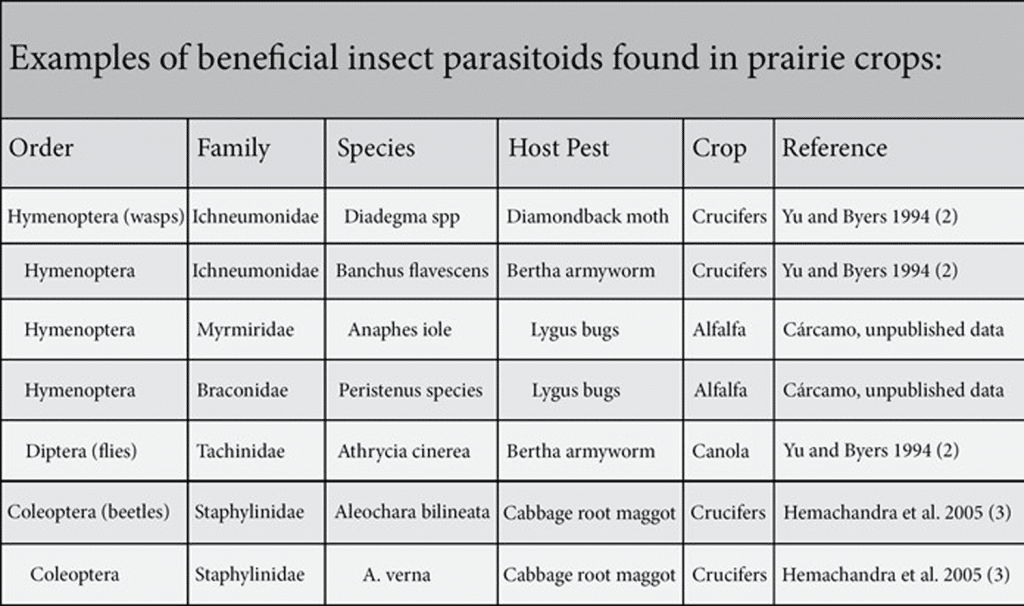
Natural enemies of pests include predators and parasitoids that feed on pests from within or attached to their skin.
Predatory insects
Some key predatory insects that may be found in and around canola fields include:
- ladybugs
- lacewings
- hover fly larvae
- minute pirate bugs
- ground beetles
- spiders
- predatory mites
- thrips (which feed on mite eggs)
Parasitoids
Many of the parasitoids that could be found in and around canola fields include:
- wasps
- flies (certain species)
These beneficial insects help to control pest insects during non-outbreak situations when the pest infestation is below economic thresholdAn economic threshold is the level of infestation (ex. pest insect density) at which lost yield (ex. due to feeding/insect pest damage) exceeds the cost of the chemical and its application. More. These insects serve a positive role in the production of canola. A healthy population of these insects is good for canola health and yield. While insecticides are valuable in controlling pest insect outbreaks, they are toxic to natural enemies. Insecticides should always be used judiciously and economic thresholds should be observed to help mitigate their impact on beneficial insects.
When scouting fields with sweep nets it is common to find beneficial insects such as:
- hover fly
- Lacewing adult, on canola flower
- Lacewing larva, consuming lygus bug
- Banchus flavanecens parasitic wasp, a parasite of bertha armyworm
- Diadegma insulare, a parasite of diamondback moth
- honey bee
Biocontrol approaches

Biocontrol utilizes non-insecticidal approaches to control insect pests. Classical biocontrol is the most common approach, where the natural enemies of an alien pest are imported from its native range to suppress the pest below economic levels. In Canada, at least 50 insects, including some agricultural pests, have been targeted from 1981 to 2000 using mostly the classical approach2.
Conservation biocontrol works to maintain or improve the effectiveness of native beneficial insects. This natural control of insect pests probably has the most economic impact by keeping many pest insects from reaching pest status. This approach is the one most readily available to growers and the one that they can influence significantly through agronomic practices, since all agroecosystems can harbour a healthy population of beneficial insects3.
In some cases, parasitoids arrive with the imported insect pest, such as in the cases of cabbage seedpod weevil and the cabbage root maggot, where the European parasitoids arrived in Canada co-incidentally with the pest.
Root maggot parasitism

Aleochara bilineata (Coleoptera: Staphylinidae) is an important natural enemy of root maggots and has been studied in western Canada. In general, greater root maggot incidence and damage, and greater population density of A. bilineata, occur in conventional tillage compared to zero tillage. Relatively greater parasitism of root maggot pupariaCocoons that overwinter in the soil (such as those which are used by root maggots). More by A. bilineata occurs in plots in zero tillage than in conventional tillage. No consistent effects are observed on A. bilineata activity in relation to seeding rate and row spacing4.
To enhance A. bilineata parasitism, in the context of integrated crop management in canola cropping systems, canola growers should utilize zero tillage in conjunction with adopting the currently recommended seeding rates of between 5.6 to 9.0 kilogram per hectare (five to eight pounds per acre) and row spacing of 30 centimetres (12 inches) because this can bring advantages in terms of improved management of root maggots and other important canola pests like flea beetles and weeds
Pollinators
Beekeeping is a major industry in the canola growing areas of Canada. Caution must be taken to avoid killing bees or beneficial insects with insecticides. Although it may be difficult to protect native pollinating insects, honeybee kill can be minimized through co-operation with beekeepers. Keep the following information in mind when spraying canola fields to maximize pollination benefits to your canola and ensure continued good working relationships with commercial beekeepers:

- Carefully sample sites in a field to be absolutely sure that insect population levels are high enough to require control measures.
- Discuss spray plans with beekeepers prior to spraying. Since the hives may have to be moved, give beekeepers at least two days of notice prior to spraying.
- If hives cannot be adequately protected (moved or covered) before insecticide spraying begins, alert the spray applicator to the exact location of the beehives within the intended spray area, so they can avoid direct spraying or spray drift contamination of the hives.
- Give careful attention to wind direction and velocity in relation to bee yard locations.
- Do not spray a crop in flower unless absolutely necessary.
- If spraying a crop in flower is necessary, do the spraying when there will be minimal bee activity in the fields, preferably during the evening hours. During most summer evenings, honeybees leave fields by 8:00 p.m. and do not return until 8:00 a.m. or later. Warm temperatures, however, can “hold” bees in flowering fields for periods longer than normal.
- Whenever possible, choose insecticides with low hazard potential to bees. Consult the table below for options but always confirm with current guide to crop protection, product labels, or the local agricultural representative or district agriculturist or apiarist.
- Do not spray crops of uneven maturity which are partly in flower or which contain flowering weeds when bees are active.5
How to preserve and enhance beneficial insects

Learn beneficial insect habits, and time insecticide application to avoid beneficial activity whenever possible. Here are some more ways to preserve and enhance beneficial insects:
Economical thresholds: Only spray insecticides when the pests have clearly surpassed the economic thresholds.
Trap crops/strips: Provide the opportunity to lure the pest to a local area where targeted insecticide applications can be made. For example, cabbage seedpod weevil can be lured to earlier flowering strips of canola where it can be sprayed in an area of less than 10 per cent of the field, thereby, sparing the rest of the beneficials6.
Reduced tillage: This can increase populations of some ground beetles and spiders and also reduces the mortality of many parasitoids that overwinter as pupae such as the Peristenus wasps that attack lygus bugs.
Tall stubble: Leaving taller standing stubble by using a stripper header has been shown to increase the overwintering survivorship of parasitoids in other crops (e.g. Bracon cephi, the parasitoid of wheat stem sawfly)7.
Intercropping: Intercropping adds vegetation diversity, which has been shown to increase populations of some natural enemies and reduce pest problems8. Incorporating perennial forage crops such as alfalfa, which offer a stable habitat with a high diversity of insect hosts may increase the overall populations of beneficials in an area.
Habitat conservation: Leaving undisturbed areas in and around cropland can provide valuable habitat for these beneficial insects and natural enemies to complete their life cycles, unhindered. Shelterbelts, road allowances, forested bluffs, grassed strips, wetlands, all play valuable roles in providing this habitat9,((Galpern, P., Vickruck, J., Devries, J., Gavin, M. 2019. Landscape complexity is associated with crop yields across a large temperate grassland region. Agriculture Ecosystems & Environment, 290. 10.1016/j.agee.2019.106724.)).
Lady beetle (Coleoptera; Family Coccinelidae)

There are over 450 species of lady beetles (also commonly called ladybugs or ladybird beetles) in North America. Most lady beetles are beneficial as both adults and larvae, primarily feeding on aphids, mites, small insects and insect eggs.
Identifying lady beetles
Adult lady beetles have the familiar red or orange back with black markings or spots. Some lady beetles can be black and often with red markings. The adults are small, round to oval, and dome-shaped. The color and markings can vary between species.
Lady beetle larvae are dark and alligator-like with three pairs of prominent legs.
Depending on the species and availability of prey, larvae grow from less than one millimetre (1/25 of an inch) to about one centimetre (3/8 of an inch) in length, typically through four larval instars, over a 20 to 30 day period. Large larvae may travel up to 12 metres (about 40 feet) in search of prey. The larvae of many species are gray or black with yellow or orange bands or spots10.
Scouting techniques
Lady beetle adults may be commonly caught in sweep nets, especially in fields with aphid populations. Early season adults may be seen on aphid infested shrubs or perennials.
Managing benefits
Lady beetles eat voraciously, and need to eat many aphids per day to produce and lay eggs. Some species may eat their weight in aphids each day as a larva, and can consume upwards of 200 to 300 aphids per day. Once aphid populations fall, the lady beetle will fly in search of additional food.
Lady beetle adults and larvae may feed on the eggs of moths and beetles, and mites, thrips, and other small insects, as well as pollen and nectar when aphids are scarce. This ability to survive on a wide range of prey makes them a valuable beneficial insect.
Tolerance to insecticides is variable among lady beetle species. Good application practices to minimize the impact on lady beetles are important. Only spray when pest populations indicate application will save enough yield to be economical.
Life cycle
Lady beetles overwinter as adults under protected areas such as leaf litter, hedgerows and field debris. In the spring, the adults disperse in search of prey and egg laying sites. Female lady beetles may lay from 20 to more than 1,000 eggs over a one to three month period, commencing in spring or early summer. Eggs are usually deposited near prey such as aphids, often in small clusters in protected sites on leaves and stems. The eggs of many lady beetle species are small (about one millimetre or 1/25 of an inch), cream, yellow, or orange-coloured, and spindle-shaped10 .
The last larval instarA developmental stage within one life stage (ex. of an insect) More remains relatively inactive before attaching itself by the abdomen to a leaf or other surface to pupate. Pupae may be dark or yellow-orange. The pupal stage may last from three to 12 days depending on the temperature and species. The adults emerge, mate, and search for prey or prepare for hibernation, depending on the availability of prey and time of year. Adults may live for a few months to over a year. The more common species typically have one to two generations per year10.
Carabid beetle (Coleoptera; Family Carabidae)
There are nearly 400 species of carabid beetles (commonly called ground beetles) in Canada. Most are beneficial, and are voracious general predators of nearly all insect pests, including cutworms, diamondback moths, mites, small insects and insect eggs. Additionally, several are weed seed consumers. Adult carabid beetles have been observed consuming four to five cutworms in a 24-hour period, but in fields are frequently not this adept at locating them11.
Identifying carabid beetles

Although the Carabidae family is widely diverse in size and colouration, they share the same basic morphological features. Antenae are long, threadlike, and 11 segmented11. Mandibles, or external jaws, are long, project away from the head, and are somewhat curved. The thorax is usually wider than the head, but narrower than the abdomen which is prominent and covered by the elytra, or hardened forewing.
The larvae are long and slender, bearing three pairs of legs on the thorax and a 10-segmented abdomen. The mandibles project forward.
Scouting techniques
Sampling for carabid beetles can be performed actively, by turning over rocks, stones, and coil clods where beetles typically shelter during daylight hours. Additionally, “spot and stalk” methods may be utilized whereby exposed beetles are procured by hand, with nets, or with pails. However, passive pitfall trapping is likely to yield the greatest numbers and diversity. This method involves burying a cup or dedicated pitfall trap to soil level, and leaving it for a period of days to catch beetles as they move about. Larger beetles tend to move farther and faster than smaller species, so pitfall trapping tends to over-represent these species11.
Life cycle
Most carabid species in Canada have one defined breeding period within the year, and development from egg to adult will take at least one year, with larval forms frequently taking several years to reach adulthood. Some species overwinter as larvae, but most overwintering occurs as adults, buried in soil, undisturbed residues, or field margins. Many species can survive multiple years, if habitat and food supplies are adequate.
- Pollinating hybrid canola- the southern Alberta experience. Clay, H. Hivelights (August 2009).14-16. Retrieved from: http://www.honeycouncil.ca/documents/CanolaSthAB2009.pdf [↩]
- Mason, P.G. & Huber, J.T. (eds). 2002. Biological Control Programmes in Canada 1981-2000. CABI Publishing, Wallingford, Oxon, UK, 583 [↩]
- Giberson, D.J. & Cárcamo, H.A. (eds). 2014. Arthropods of Canadian Grasslands. Volume 4: Biodiversity and Systematics, Part II. Biological Survey of Canada. ISBN: 978-0-9689321-7-9. [↩]
- Dosdall, L. M., Identifying Agronomic Practices that Conserve and Enhance Natural Enemies of Insect Pests in Canola. CARP Project: 2007-03 [↩]
- Saskatchewan Agriculture and Food. 2020. Guide to Crop Protection. Retrieved from: www.agriculture.gov.sk.ca/Guide_to_Crop_Protection [↩]
- Dosdall L.M., Moisey, D., Cárcamo, H.A., & Dunn, R. 2001. Cabbage seedpod weevil fact sheet. Alberta Agriculture Food and Rural Development. Agdex622-21.4 [↩]
- Meers, S. 2005. Impact of harvest operations of parasitism of the wheat stem sawfly, Cephus cinctusNorton (HymenopteraThe insect order (the scientific classification more broad than Family) which includes sawflies, wasps, bees, and ants. More: Cephidae). M.Sc. Thesis, Montana State University. Bozeman, Montana, U.S.A. [↩]
- Vandermeer, J. 1989. The Ecology of Intercropping. Cambridge University Press, Cambridge, 237. https://doi.org/10.1017/CBO9780511623523 [↩]
- Vickruck, J., Best, L., Gavin, M., Devries, J. & Galpern, P. 2019. Pothole wetlands provide reservoir habitat for native bees in prairie croplands. Biological Conservation, 232, 43-50. 10.1016/j.biocon.2019.01.015. [↩]
- Hoffmann, M.P. & Frodsham, A.C. 1993. Natural Enemies of Vegetable Insect Pests. Cooperative Extension, Cornell University, Ithaca, NY. 63. [↩] [↩] [↩]
- Holliday, N. J., Floate, K. D., Cárcamo, H., Pollock, D. A., Stjernberg, A., & Roughley, R. E. 2014. Ground beetles (Coleoptera: Carabidae) of the Prairie Grasslands of Canada. In Arthropods of Canadian Grasslands (Volume 4). Biodiversity and Systematics Part 2. Chapter 1. 1-85. Edited by D. J. Giberson and H. A. Cárcamo. Biological Survey of Canada. © 2014 Biological Survey of Canada. ISBN 978-0-9689321-7-9. [↩] [↩] [↩]