- 1 Role of sulphur in canola plants
- 2 Sulphur uptake by canola
- 3 Sulphur supply from the soil
- 4 Important tips for sulphur fertilizer management
- 5 Canola response to fertilizer sulphur
- 6 Fate of applied sulphur fertilizer
- 7 Identifying deficiency symptoms
- 8 Placement of fertilizer
- 9 Timing of application
- 10 Product choices
- 11 Nitrogen: Sulphur ratio
- 12 Footnotes
Sulphur (S) is the fourth macronutrient, but ranks as the third most limiting nutrient on the Prairies. Sulphur deficiency in western Canada was first identified in 1927 on Gray Wooded soils in Alberta. Canola is more sensitive than cereals to sulphur deficiency and frequently responds to fertilizer sulphur addition. Therefore, pay equal attention to nitrogen, phosphorus and sulphur.
Role of sulphur in canola plants
Canola contains large amounts of sulphur. Sulphur is part of structural and enzymatic components. Sulphur is a key component of two essential amino acids (cysteine and methionine) and is needed for protein synthesis. Chlorophyll synthesis also requires sulphur. Both of these amino acids are also precursors for coenzymes and secondary plant substances. Glutathione, an important antioxidant in plants and animals, is synthesized from cysteine. Glutathione contents are higher in leaves than roots. It’s found primarily in the chloroplasts where its anti-oxidant ability is needed to detoxify free radicals generated during photosynthesis. Glutathione also functions as transient sulphur storage, and a precursor of phytochelatins (compounds which detoxify heavy metals in plants). Thioredoxins, another important group of sulphur compounds related to glutathione, help activate several enzymes in carbon metabolism. Sulphur also is part of several enzymes and coenzymes such as ferrodoxin, biotin (vitamin H), coenzyme A, urease, and thiamine (vitamin B1).
An important group of secondary plant sulphur compounds in canola are glucosinolates. Plants contain over 100 different glucosinolate compounds. These secondary compounds, although not well understood, probably have a number of functions. Glucosinolates are stored in cell vacuoles, and can be broken down by an enzyme (myrosinase) to yield glucose, sulphate and volatile compounds such as isothiocyanate. Glucosinolates contribute to defense or attractant systems for certain insects and diseases. When plant cells are destroyed by insect feeding, glucosinolates are broken down, releasing various deterrents/attractants.
Glucosinolate levels are highest in growing points, roots, and youngest leaves, all of which are most vulnerable to insects and diseases. The role of glucosinolates as sulphur reserves to maintain plant sulphur during periods of high demand (such as bolting, flowering, podding and seed fill) is controversial. However, research in Europe showed that glucosinolates comprised a small sulphur pool in leaves, and under induced sulphur deficiency, sulphate (SO4-2) mobilization from storage in cell vacuoles was about 10 times greater than contributions from glucosinolates.
Sulphur is also a constituent of sulpholipids, which are membrane components.
Sulphur uptake by canola
The main sulphur form absorbed by canola roots is sulphate. In industrial areas, atmospheric sulphur compounds dissolved in rain can be absorbed by leaves. However, this amount is quite small and is decreasing with better air pollution control. Sulphate absorption is accomplished with active transport systems across membranes. The uptake rate increases as the sulphate level increases in the soil water. Low plant sulphur contents also increase the root uptake rate. Negative feedback signals for sulphur uptake may be sulphate or glucosinolate levels in vacuoles, or the levels of organic sulphur compounds such as cysteine, methionine or glutathione. Sulphate uptake faces competition from molybdenum and selenium. Therefore, soils high in these minerals will antagonize sulphur uptake.
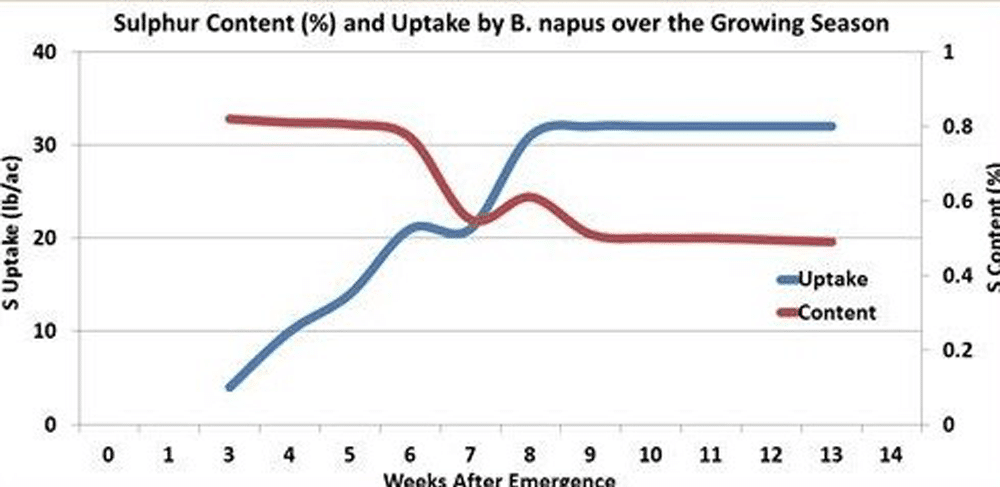
The sulphur level in canola plants is highest in the early seedling stage when young leaves comprise most of the dry matter.
As plants develop, the overall sulphur level declines but not as dramatically as with nitrogen. By maturity, canola straw contains approximately 0.3 to 0.4 per cent sulphur while pod chaff contains slightly more sulphur (0.5 to 0.6 per cent). Canola seed contains about 0.4 to 0.6 per cent sulphur. At harvest, canola straw and pod chaff contain roughly twice as much sulphur per acre as that in the seed.
Sulphur uptake increases rapidly after germination and peaks three to four weeks after emergence 1. This highlights the importance of early season sulphur availability, but also indicates that sulphur deficiency can be corrected if top dressing of ammonium sulphate occurs early enough, before the canola bolts.
There has been limited research on the complex sulphur partitioning into the various compounds of different plant parts over the growing season. Most of the plant sulphur ends up in protein and stored sulphate. As leaves senesce, protein sulphur is readily remobilized, while stored sulphate remobilization is slow and more limited. Therefore, overall, sulphur has medium mobility.
Sulphur supply from the soil
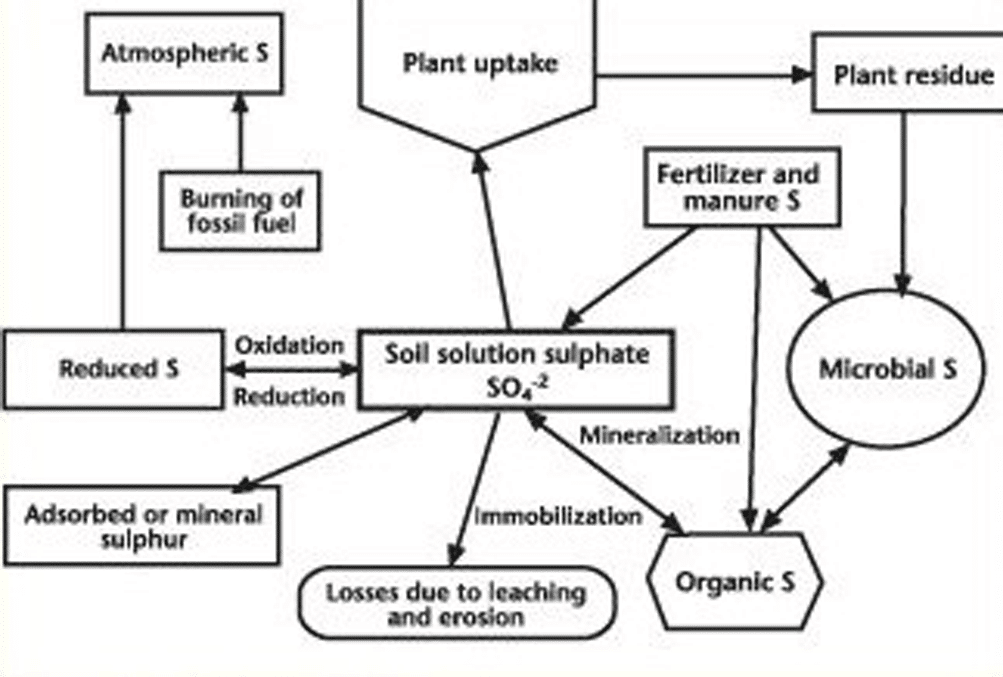
The organic portion of the sulphur cycle in soil is closely tied to nitrogen due to their association in protein. Like nitrogen, the main sulphur reserve in soil is in organic matter. Although there is considerable variability in the relative proportions or ratio of carbon, nitrogen and sulphur (C:N:S) in soil organic matter, the ratios are quite similar for each soil group. In a study of Saskatchewan farm soils, the C:N:S ratio ranged from 58:6:1 in Brown soils, to 63:7:1 in Dark Brown, 83:8:1 in Black, 100:8:1 in Gray Black, and 129:11:1 in Gray soils.
A key component of the soil sulphur cycle for plant growth is the mineralization path. Soil organic matter and plant residues are decomposed by soil microbes, releasing sulphate. The sulphur mineralization rate is quite slow (much slower than nitrogen), and cannot match the uptake rate of growing plants. Also like nitrogen, the sulphate amounts released from residues will depend on the sulphur content. When plant residues contain more than about 0.15 per cent sulphur (C:S ratio about 300:1), there will be a net release of sulphate through mineralization. Below 0.15 per cent sulphur, decomposition is slower and there will be immobilization of soil sulphate by soil microbes. The ability of soil to mineralize sulphate from organic matter has been found to be independent of the total amount of carbon, nitrogen or sulphur, and of the C:N or N:S ratios in soils. However, research has also found that the initial amounts of sulphate mineralized from soil are closely correlated with the initial amounts of nitrogen mineralized in short-term incubation.
Another important aspect of the soil sulphur cycle is the oxidation path. In soils, sulphides, elemental sulphur and thiosulphate can be oxidized to sulphate by various soil microbes, but the main actors are bacteria from the genus Thiobacillus. The oxidation of these inorganic sulphur compounds produces considerable sulphuric acid. Sulphur oxidizing bacteria are most active under warm, moist, well aerated conditions. It is the oxidizing ability of these bacteria that permits the agricultural use of elemental sulphur for crop growth.
Although sulphur reduction is shown in the soil sulphur cycle diagram, it generally is not significant in aerated agricultural soils. In flooded soils, sulphate can be reduced by soil microbes to sulphides in a process analogous to denitrification. However, soil microbes will utilize nitrate, iron and manganese compounds before reducing sulphate.
In many western Canadian soils, there is a subsoil salt (gypsum) and/or a lime (calcium carbonate) layer. This subsoil layer contains considerable sulphate, often as co-precipitates with lime. Although this subsoil sulphate solubility is reduced, it still can contribute to plant needs if it exists within the rooting zone. However, the length of time that canola grows in sulphur deficient topsoil before rooting to the subsoil sulphur will affect the yield response to fertilizer sulphur. Also, the depth to subsoil sulphur tends to vary greatly across the field. Total sulphur amounts (organic and sulphate) generally increase from upper to lower slope positions.
In most prairie soils, sulphate is not held by organic matter and clay particles since they are both negatively charged. Therefore, sulphate is vulnerable to leaching losses.
Important tips for sulphur fertilizer management
- Sulphur is very important for meeting yield expectations. Typically it is recommended to give canola at least 10 to 20 pounds per acre of sulphur, regardless of the soil test results. Due to highly variable sulphur levels within fields, composite soil test may show sufficient levels even though large parts of the field are deficient.
- Use ammonium sulphate to address sulphur needs in the year of application. Elemental sulphur will not usually be converted to available sulphate form in time for adequate uptake in sufficient quantities in the year of application.
- Ammonium sulphate (AS) should be placed away from the seed row. Save the seed row location for phosphorus fertilizer, as it provides a known early season benefit to stand establishment. Adding AS to the seed row in addition to ammonium phosphate pushes the nitrogen levels too high for seedling safety in many cases.
- An in-crop application of sulphate fertilizer can be effective at rescuing most of the crop’s yield potential if canola shows signs of deficiency and fertilizer is applied early enough to allow sufficient uptake by early flowering at the latest.
Canola response to fertilizer sulphur
Yield
Canola has a higher sulphur requirement than any other major crop grown in Western Canada. Expansion of canola acres, increased yield through improved cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and production practices, and tightening rotations have increased sulphur removal from soils to the point where fields or parts of fields that were once well supplied with sulphur are becoming depleted.
Canola generally responded to sulphur fertilizer when the water soluble soil sulphate content to a 60 centimetres (two-foot) soil depth was less than 22 to 34 kilograms per hectare (20 to 30 pounds per acre).
Canola yield response to sulphur fertilizer is dramatic under extreme sulphur deficiency, but canola will also often respond to sulphur fertilizer on soils where a composite soil sample shows sulphur levels to be sufficient. This may be because sulphur levels can be highly variable across a field.
For example, extreme sulphate variability was found on a 24 hectare (60 acre) solonetzic field near Stettler, Alberta in 1994. Composite samples were taken from each 0.5 hectare (acre) and tested separately. The average worked out to be 1,076 kilogram sulphate per hectare (960 pounds of sulphate per acre) in the 60 centimetres (two-foot) depth, which is excessive from the soil test standpoint. However, the average value is heavily skewed by the few samples with extremely high sulphate values. The mode (or result that occurred most frequently in this field), was just nine kilogram (20 pounds) of sulphate, which is deficient. The majority of this field would likely respond to sulphur fertilizer.
The Stettler example illustrates that single composite soil samples from fields with high sulphur variability can be difficult to interpret. A soil testing deficient for sulphur is likely deficient (unless underlain by a subsoil sulphate salt layer), while soils testing medium or high for sulphur may have deficient areas that are skewed by areas with excessive sulphur.
For fields testing medium or high for sulphur, a blanket application of 11-22 kilograms per hectare (10-20 pounds per acre) of sulphur will ensure ample sulphur in low-sulphur areas to meet canola yield expectations. For fields on most soils testing low for sulphur, seed yields are usually optimized with application of 15-30 kilograms per hectare (13-26 pounds per acre) sulphur as sulphate, at seeding.
Canola needs 0.5 to 0.7 pounds of sulphur per bushel of yield. A 50 bushel per acre canola crop needs 25-35 pounds per acre of available sulphur. If the 10 to 20 pounds per acre application rate is insufficient to meet a grower’s yield expectation, accounting for soil residual levels, then a higher rate will be warranted.
Canola response to sulphur fertilizer varies greatly, depending on:
- soil sulphate levels (amount, spatial and temporal distribution)
- availability of other nutrients (especially nitrogen, phosphorus and possibly boron)
- soil moisture
- amount, type, and method of sulphur fertilizer applied
Quality
A Saskatchewan study found that optimal yield response for sulphur fertilizer on canola occurred at a sulphur rate of 10 kilograms per hectare (nine pounds per acre), while seed quality (oil, protein and sulphur concentration) responded up to the 15 kilogram per hectare (13 pounds per acre) rate.
Some studies have found that sulphate (sulphur) applications increase concentration of oil in canola seed while other studies found a decrease or no change.
Application of sulphur has been found to decrease chlorophyll concentration in seed, suggesting that seed quality can be improved by correcting sulphur deficiency in canola.
Sulphur fertilization can increase protein content of the meal, which is desirable, but can also increase glucosinolate contents. Glucosinolates increase with excessively high sulphur fertilizer rates, but are usually well below the standard canola quality limit (30 micromoles per gram).
Economic
Canola yield response to the first 10-20 pounds per acre of applied sulphur can be dramatic, especially for parts of the field where sulphur is deficient. These areas will need sulphur in order to generate an economic gain from any amount of nitrogen fertilizer. But once a baseline amount of soil sulphate sulphur is reached, the economic return for subsequent fertilizer will be much higher for nitrogen than for sulphur.
Fate of applied sulphur fertilizer
The organic portion of the sulphur cycle in soil is closely tied to nitrogen due to their association in protein. Each undergoes mineralization from organic matter, immobilization, oxidation and reduction of inorganic compounds.
Like nitrogen, the main sulphur reserve in soil is in organic matter. Growers can expect roughly two to three pounds per acre of available sulphur for each percentage point of organic matter. Soil with four per cent organic matter could provide eight to 12 pounds of available sulphur to the crop.
However, the sulphur mineralization rate is quite slow, and cannot match the uptake rate of growing plants.
In most Prairie soils, sulphate is not held by organic matter and clay particles since they are both negatively charged. Therefore, sulphate is vulnerable to leaching losses.
Identifying deficiency symptoms
Sulphur deficiency symptoms include:
- Top leaves are small and narrow, and are often cupped.
- New leaves are pale yellow.
- Prolonged flowering may occur if the crop is having trouble setting seed.
- Small flowers that have very pale yellow colouration.
- Short pods with little or no seed set.
- Patchy look to the field. Sulphur is highly variable across a field, so deficiencies will usually show up in patches.
- Sulphur deficiency is more typical in sandy soil with low organic matter.
Sulphur has several effects on canola growth. Since chlorophyll synthesis requires sulphur, sulphur deficiency will affect visible leaf colour and photosynthesis. Protein synthesis requires sulphur containing amino acids, and, therefore, sulphur deficiency affects rapidly growing parts, especially reproductive structures.
Mild sulphur deficiency often does not result in noticeable symptoms, but still can reduce yield. Medium deficiencies do not show symptoms until bolting, flowering and podding. Under severe deficiency, symptoms show up about two weeks after germination.
By the bolting stage, sulphur deficiency begins to affect yield parameters such as branches per plant, fertile flowers per plant, seeds per pod and individual seed weight. Under mild to moderate sulphur deficiency, the thousand kernel weight is normally not significantly affected since plants compensate by reducing the seed number per pod. Nitrogen deficiency affects pods per plant more than sulphur deficiency, while sulphur deficiency reduces the seeds per pod.
Sulphur deficiency symptoms vary depending upon the severity and timing of the deficiency relative to crop growth stage. In the vegetative stage, foliar symptoms show up under severe sulphur deficiency. Since sulphur has a low mobility within the plant, symptoms are observed more readily on the youngest leaves, which are greenish-yellow compared to the normal bluish-green in B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More. The yellowing (chlorosis) starts from the leaf edges and the tissue around leaf veins remains green.
Subsequently, the leaf edges and bottoms may turn purple as may be caused by other plant stresses. Besides the leaf colour, sulphur deficiency in young plants causes smaller leaves as well as upward cupped leaves.
By the bolting stage, new leaves of sulphur-deficient plants show chlorosis, purpling and spoon-like leaf cupping. The purple colour is caused by enhanced pigment (anthocyanin) synthesis due to sugar accumulation resulting from sulphur limited amino acid and protein synthesis. The degree of leaf cupping is highly dependent on the timing of the sulphur deficiency. There is significant cupping when sulphur deficiency occurs before half of the leaf weight is attained.
By flowering, sulphur deficiency symptoms can show up in the petals, which may be smaller and lighter yellow. If severe sulphur deficiency occurred in the vegetative stage, symptoms can be found both in foliage and flowers. However, if sulphur deficiency occurs around flowering, leaf symptoms may not be obvious, but flower petals may become paler. Yellow and white petals may even exist side by side on a single flower.
The life span of sulphur-deficient petals is shortened to one day from two or three, and pollen production is greatly reduced. In addition, sulphur-deficient petals are egg shaped compared to more round petals on sufficient sulphur plants. Flowering is often delayed and prolonged. Reports suggest that sulphur-deficient plants do not attract honey bees, perhaps due to lack of pollen. By podding, sulphur deficiency becomes more distinctive. Pod number and size, and seed number per pod are reduced significantly. Pods may be pale green, often with purpling and can be compressed or flattened.
Proper diagnosis of nutrient deficiency should use the following assessments:
- Soil test. Consider if any obvious shortages are evident. Consider soil quality variation within the field. Determine if the symptom severity is consistent with typical differences in soil sulphur levels (e.g. less sulphur on hilltops leading to more severe symptoms).
- Fertilizer history. In past years, consider which fertility rates and sources have been applied on that field. Include crop yields and consider whether rates have been adequate to match removal. Since sulphur is mobile in the soil, and prone to leaching, so consider if the current year’s sulphur rate was adequate to meet crop needs.
- Tissue test. Use in conjunction with other tests. Consider that different cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More may have considerably different critical levels at which sulphur deficiency is observed. A comparative test between healthy looking and deficient looking plants in the field is always more preferable. Make sure to record growth stage as criteria change with stage.
- Environment. Cold, wet, hot and dry can all stress canola plants, creating symptoms that may look like nutrient deficiencies. If neighbors have similar symptoms, the cause is probably environmental (e.g. frost, excess moisture, etc.).
- History of the land. Recently broken forage land is likely to be depleted in a lot of nutrients. For example, alfalfa uses significant quantities of sulphur, so if the field has been baled, removing all top growth over many years, sulphur levels could be very depleted.
- Look at other fields for similar symptoms. When diagnosing for a specific nutrient, target the crop that tends to be most sensitive to that nutrient. Canola is more sensitive to low sulphur levels than cereal crops are.
Placement of fertilizer
Seed row
Applying ammonium sulphate in the seed row along with monoammonium phosphate can increase seedling toxicity and reduce plant stand affecting maturity, and final seed yield. Phosphorus should be the top priority for seed row placement, because unlike sulphur it has demonstrated crop response benefits from this type of placement at safe seed-placed rates.
Only limited amounts of sulphur fertilizer can be safely applied near the seed. The safe amount will vary with the degree of seedbed utilization, moisture conditions, soil type, fertilizer source, and amounts of other fertilizer nutrients. Until research has determined the safe amount of seed row sulphur fertilizer under various combinations of the above factors, limit seed-placed ammonium sulphate to no more than 12 kilograms per hectare (10 pounds per acre) of sulphate. Include nitrogen amounts in the sulphur fertilizer source when calculating total nitrogen fertilizer amounts to be safely seed-placed.
Band
The best yield response to sulphate fertilizer is at or before seeding, as was determined from a six site-year study at the Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More research centre in Melfort, Saskatchewan. This research also showed that side banding or pre-seed incorporation increased yield more than seed row placement of 30 kilogram per hectare (27 pounds per acre) of sulphate, probably due to seedling toxicity.
Broadcast
Under average to good moisture conditions, sulphate-containing fertilizer can be broadcast applied in the spring with good results. Under dry spring conditions, broadcast sulphate-containing fertilizer can be stranded and result in poor uptake. However, under such dry conditions, canola germination and establishment will also be severely affected. Sulphate does not pose a risk for volatilization which makes broadcast applications more acceptable than for many forms of nitrogen fertilizer.
In-crop
Applications at seeding are generally more effective than at bolting and early flowering stages, but in-crop applications can correct sulphur deficiencies. An Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More study found that in-crop applications of sulphur can restore seed yield substantially at bolting and moderately at early flowering. This study applied sulphur in-crop at sulphate rates of 15 kilograms per hectare (13 pounds per acre) and 30 kilograms per hectare (26 pounds per acre). The results are in the table below. Topdressing, in this case, is applying granular potassium sulphate at the sulphur rates listed. Foliar application, in this case, is a liquid potassium sulphate solution at 200 litres per hectare (20 gallons per acre).
Table 6. Impact of sulphur treatment and rate on yield
Elemental sulphur is not a good choice for in-crop application since its slow conversion to sulphate means the sulphur may not be available to the crop soon enough. Elemental sulphur is better suited for long-term maintenance.
When doing an in-crop application, growers could target only those areas (such as hill tops or sandy areas) that tend to be sulphur deficient.
An in-crop application can make financial sense for canola if:
- Growers could not put the desired rate on at seeding.
- Yield potential improved and growers want to add sulphur to their nitrogen top dress. If field conditions have been excessively moist, sulphur may have moved lower in the soil profile. As canola plants grow, their roots will extend into these reserves. For that reason, growers who have been applying recommended rates of sulphur may not see as much economic return from a sulphur top up compared to a grower who has cut sulphur rates in recent years.
- Canola shows signs of sulphur deficiency.
Application prior to rainfall is best. Surface applied ammonium sulphate requires rain to move it into the root zone. Sulphur is not volatile like nitrogen fertilizer, so losses will be minimal if rain is not immediately forecast.
If dribbling on liquid ammonium sulphate (with the nitrogen-phosphorus-potassium blend of 8-0-0-9) or ammonium thiosulphate (with the nitrogen-phosphorus-potassium blend of 12-0-0-26), damage from leaf burn tends to be more severe when canola is smaller than the five-leaf stage and when the crop canopy is dry. Always use streamer nozzles (dribble band) liquid forms of sulphur in-crop.
Applying sulphate fertilizer in a tank mixCombining two or more crop protection products (that are compatible) in one application. More with herbicide/fungicide may not provide enough sulphur to provide an economic benefit to the crop. Flat fan nozzles that cover the leaf are far more toxic to the crop than fertilizer dribbled on the soil surface. Plus, the crop can’t take up large amounts of fertilizer nutrients through the leaves.
Timing of application
Fall broadcast elemental sulphur fertilizer left on the surface until next spring will improve the oxidation and canola yield response compared to spring broadcast. However, this practice should still be initiated two years ahead of the canola crop to ensure consistent response, and is unlikely to match the availability of spring applied sulphate fertilizer.
Spring, before or at seeding, tends to be the best time to apply sulphate fertilizer. Highest fertilizer use efficiency generally results when sulphate fertilizer is placed in the soil for easy access, and just before the period of plant uptake. Under dry spring conditions, broadcast sulphate fertilizer can be stranded and result in poor uptake. However, under such dry conditions, canola germination and establishment will also be severely affected. Under average to good moisture conditions, sulphate fertilizer can be broadcast-incorporated in the spring with good results. On sandy soils, sulphate leaching can occur during wet periods.
Product choices
Table 7. Effect of sulphur fertilizer form and placement on canola yield
Many different sulphur fertilizers are available, and each requires a different management system to maximize the nutrient potential. Sulphates are the best choice to meet immediate needs of the crop.
Available forms
Sulphate fertilizer forms should be used when canola needs sulphur for immediate crop uptake. Choices:
- Dry ammonium sulphate (20.5-0-0-24 granular and 21-0-0-24 crystalline)
- Liquid ammonium thiosulphate (12-0-0-26). This requires a short time period for oxidation to sulphate.
Fertilizer use efficiency is highest when sulphate fertilizer is placed in the soil for easy access, and just before the period of plant uptake.
Elemental sulphur (0-0-0-90 or 0-0-0-95) has a significant disadvantage in that sulphur is unavailable to the crop until soil bacteria oxidize it into sulphate form.
The most consistent response to elemental sulphur fertilizer will be achieved by surface broadcasting the granules, allowing time for granule breakdown by rain, frost or snow, and then mixing the particles with soil by tillage. Therefore, apply elemental sulphur fertilizer at least the fall before seeding canola. In some cases, elemental sulphur fertilizer application needs to be two years before seeding canola. Research found that elemental sulphur fertilizers became effective in the second to fourth year, but seed yield and sulphur uptake were still less than those obtained with sulphate fertilizer, particularly when the sulphur fertilizers were applied in spring.
Conversion rate from elemental sulphur to sulphate depends on the particle size, the degree of dispersion in the soil, and the growing conditions for the bacteria. Common elemental sulphur fertilizers are formulated as granules or pastilles (split pea shape) for ease of shipping and handling, each consisting of thousands of individual particles. The surface area of these individual particles is the access where the soil bacteria feed, converting the elemental sulphur to sulphate. Small particles have the largest surface area and the fastest oxidation rate.
Another factor that influences the oxidation rate is previous use of elemental sulphur in the field. Exposure to elemental sulphur in the past has been shown to increase oxidation rates of subsequent applications, probably due to stimulation of the sulphur-oxidizing bacterial population.
However, repeated applications of elemental sulphur fertilizer can negatively impact agricultural soil. Saskatchewan research on Gray Wooded soil found that repeated elemental sulphur application decreased soil pH, organic carbon, and microbial biomass. Negative effects on soil enzymes involved with nutrient transformations were also found.
Gypsum is available from mines but its low analysis (about 17 per cent sulphur) limits its transportation and use. Gypsum also contains impurities and so many jurisdictions restrict land application. In addition, gypsum is only slightly soluble, which limits its usefulness as an immediate sulphate source. One possible advantage is that gypsum contains calcium, which can promote good soil structure — a potential benefit on soils prone to crusting.
Potassium sulphate (K2SO4) is more widely used for tobacco, fruit and vegetables, and on lawns. Potassium sulphate should perform similarly to ammonium sulphate, in terms of sulphur availability.
Special formulations
Micro-essentials S15 (with a nitrogen-phosphorus-potassium blend of 13-33-0-15) is a granule with nitrogen, phosphorus and sulphur in layers. Sulphur is in both elemental and ammonium sulphate forms.
For powder elemental sulphur, applying very fine particles as a powder on a field scale is just not practical given fertilizer delivery systems (drills, spreaders) in use on the Prairies. Powder forms do convert to sulphate faster, but field scale use is, so far, impractical. Powder sulphur is also a skin and eye irritant and can be explosive.
Nitrogen: Sulphur ratio
A proper nitrogen to sulphur balance (N:S ratio) is important for canola production. When nitrogen is in excess and sulphur is deficient (high N:S ratio), there is insufficient sulphur to combine with the nitrogen to make protein, and thus non-protein nitrogen accumulates.
An outdated guideline is to add nitrogen and sulphur fertilizer in a 7:1 (N:S) ratio. At the moderate to high levels of nitrogen fertilizer needed in areas with high crop yields, sulphur sufficiency is reached far sooner than nitrogen sufficiency, and thus the fixed 7:1 (N:S) ratio would overapply sulphur. Further, the practice of balancing applied nitrogen and sulphur in a fixed ratio on soils containing sufficient sulphur levels appears unnecessary and wasteful 2.
Also, canola yield is highly dependent on nitrogen fertility. Optimum yields of canola are derived so long as individual nitrogen, phosphorus and sulphur requirements are fulfilled. Nitrogen requirements to obtain optimum yield of hybrid canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More are higher than that of conventional canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Once a nitrogen or sulphur deficiency is corrected, there appears to be little need for balancing nitrogen and sulphur application rates at any particular ratio with hybrid canola 3.
There has been research into using the N:S ratio during tissue testing to determine sulphur status. However, the N:S ratio of canola tissue has not proved reliable for predicting sulphur status. The N:S ratio only indicates the relative proportions of nitrogen and sulphur in the plant, and does not indicate their actual magnitudes. Therefore, if canola tissue tests show an optimal ratio of 7:1 (N:S), there are three possibilities: both nitrogen and sulphur levels are optimal, excessive, or deficient. At the rosette stage, tissue testing canola for sulphur status should include several criteria to improve the reliability:
- sulphur greater than 0.25 per cent
- N:S ratio of 10 or less
- a sulphate: total sulphur ratio (as indicated by hydriodic acid reducible sulphur: total sulphur) greater than 0.38
Research has shown a decrease in canola yield as a result of nitrogen application when nitrogen was applied alone to sulphur deficient soils.
Footnotes
- Malhi, S.S., Brandt, S., Ulrich, D., Lafond, G.P., Johnston, A.M., & Zentner, R.P. 2007. Comparative nitrogen response and economic evaluation for optimum yield of hybrid and open-pollinated canola. Can. J. Plant Sci., 87, 449-460.[↩]
- Karamanos, R.E., Goh, T.B., & Flaten, D.N. 2007. Nitrogen and sulphur fertilizer management for growing canola on sulphur sufficient soils. Can. J. Plant Sci., 87, 201-210.[↩]
- Karamanos, R.E. Goh, T.B., &Poisson, D.P. 2005. Nitrogen, Phosphorus and Sulfur Fertilization of Hybrid Canola. J. Plant Nutr., 28, 1145 – 1161.[↩]