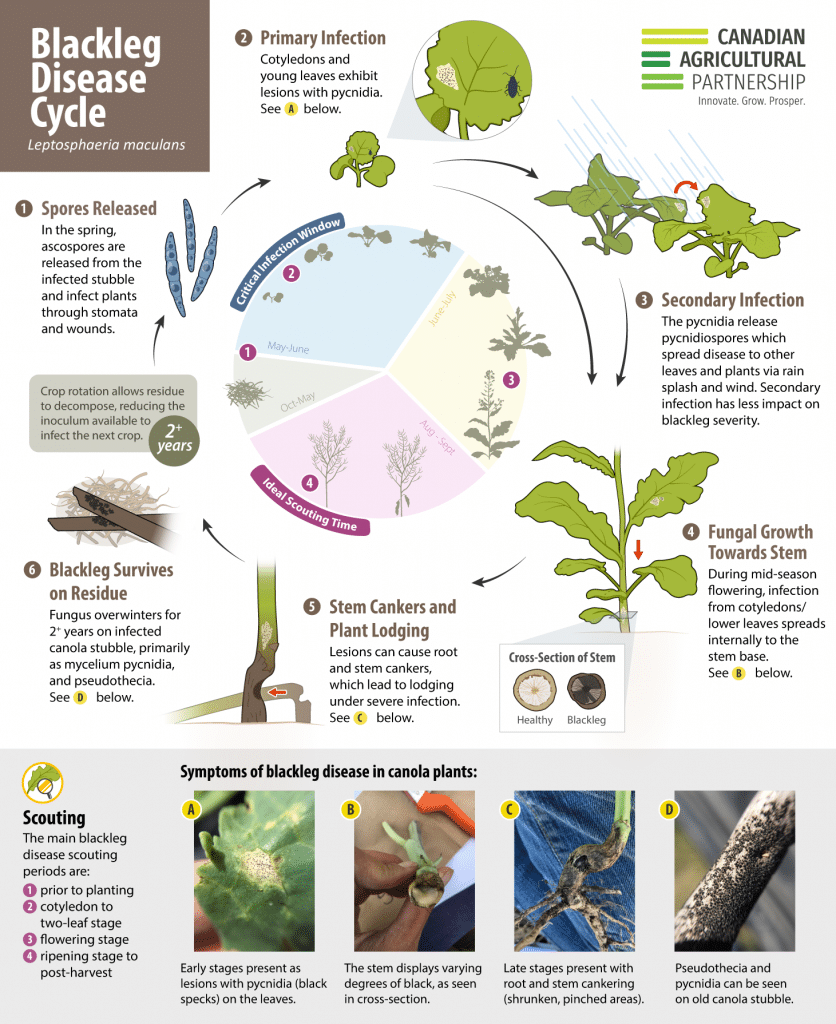
Blackleg is a serious disease of canola across all canola growing areas. Field scouting and accurate identification of the plant symptoms is required to diagnose this disease. Blackleg requires an integrated management strategy utilizing the best agronomic practices to minimize yield loss and maintain the effectiveness of genetic resistance in canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More.
Important tips for best management of blackleg
- Scout crops for accurate identification of blackleg, its severity and the proportion of plants affected by it, to estimate economic impact and assist blackleg management planning.
- Select resistant (R) rated cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and rotate resistance genetics. Utilizing the same resistance genes repeatedly in the field over time can select for races of the fungus that are virulentA form which causes infection (and can result in a disease) More against that resistance. The higher the frequency of growing the same cultivar, the greater the risk.
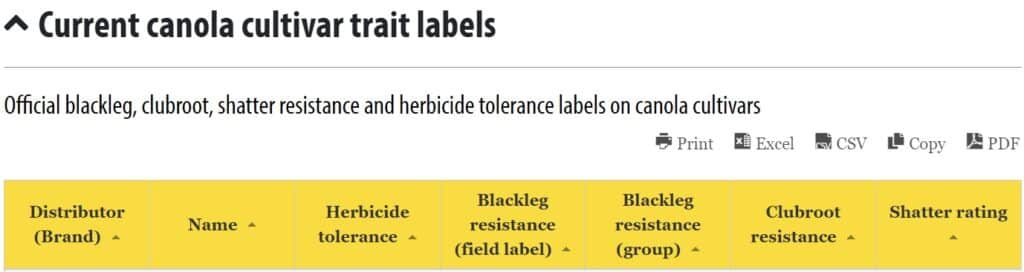
- See the Current canola cultivar trait labels for a list of cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and their corresponding resistance field labels and resistance groups.
- Increasing the number of years between canola crops in the rotation reduces incidence and severity of blackleg in fields. Maintain a minimum break of two years between canola crops, to protect against the breakdown of current blackleg resistance in cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and allow effective long-term blackleg disease management.
- Use certified seed and control Brassica weeds and volunteers.
- Use the Blackleg Management Guide and Understanding Blackleg Resistance to effectively integrate a range of tools into a cohesive blackleg management strategy.
Blackleg overview
Blackleg disease of canola is caused primarily by the fungus Leptosphaeria maculans. It is a serious disease of canola and can cause significant yield losses in susceptible cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More.
Blackleg, also known as stem canker or phoma stem canker, is actually attributed to a complex of two species, L. maculans and L. biglobosa. These species were previously referred to as highly and weakly virulentA form which causes infection (and can result in a disease) More forms of L. maculans. Leptosphaeria biglobosa, a mild or weakly virulentA form which causes infection (and can result in a disease) More species, is often associated with upper stem lesions and is widespread in western Canada. This species usually infects plants late in the season, rarely causing significant yield losses and is therefore considered a minor problem 1, 2.
However, L. maculans is the virulentA form which causes infection (and can result in a disease) More species causing blackleg that infects canola from the seedling stage onward. It progressively damages the crop as the season progresses, by girdling stems and restricting moisture and nutrient uptake, and eventually leading to yield loss. Leptosphaeria maculans was first detected in 1975 in north east Saskatchewan and in 1987 in Ontario. Since then L. maculans has become widespread throughout western and eastern Canada.
In western Canada, yield losses up to 50 per cent have been reported in individual fields. In the early 1990s, canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with high levels of genetic resistance to the disease were introduced and cultural controls such as rotation were adopted by producers. These measures reduced the yield and quality losses associated with the disease, but did not eliminate the pathogenA disease-causing organism (such as a fungus or bacteria). More. By the late 1990s, evidence of virulence changes in the pathogenA disease-causing organism (such as a fungus or bacteria). More was observed, and in recent years producers have reported higher than expected disease severity in previously resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More.
It is not unusual to observe blackleg symptoms in canola crops, even when resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More are grown. It is important to be familiar with blackleg symptoms, the disease cycle, and to know the disease management practices that prevent yield and seed quality losses.
Disease cycle of blackleg
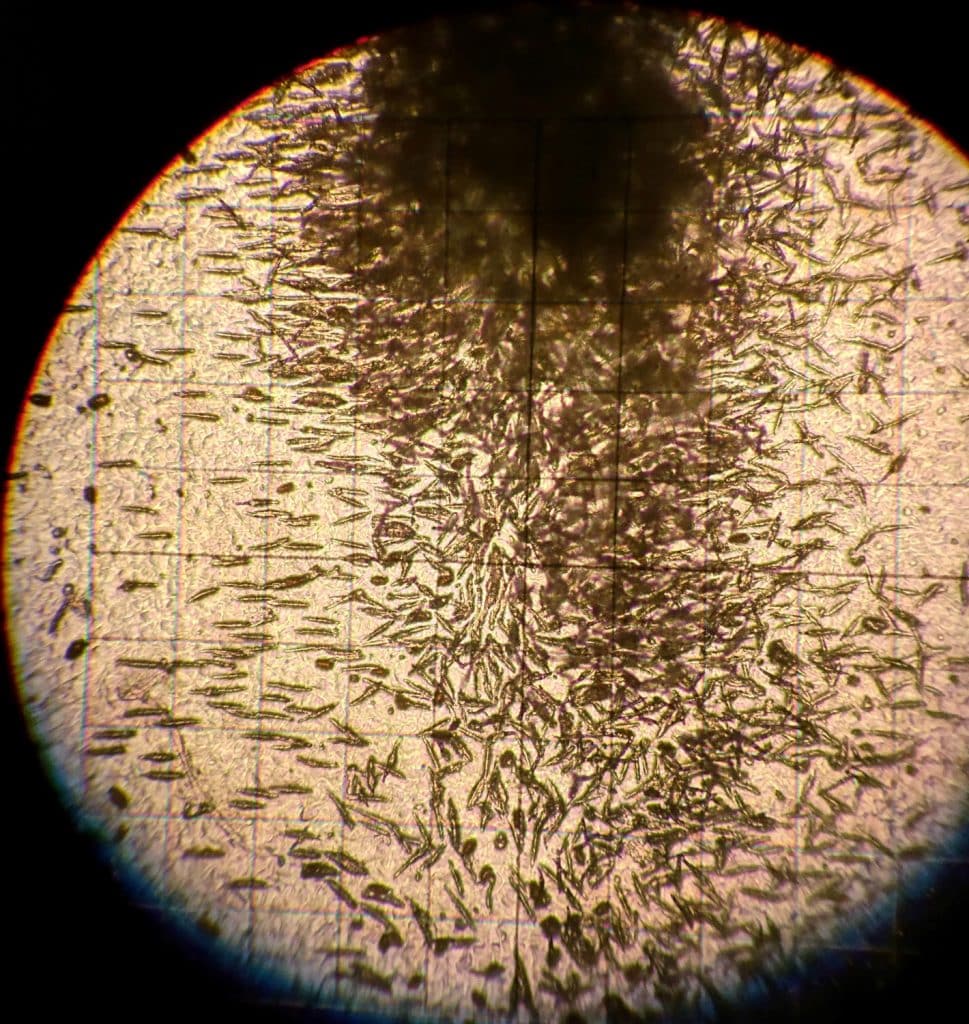
The L. maculans fungus overwinters on infected canola residue. The spores produced from diseased stubble, especially the infected lower stem and upper root pieces, are the major source of the pathogenA disease-causing organism (such as a fungus or bacteria). More that contributes to widespread field infection and yield loss. In the spring, the fungus produces fruiting bodies, called pseudotheciaFruiting bodies produced by fungus which cause disease (as the L. maculanspseudothecia can be produced on infected canola residue, which can then infect live canola plants nearby). It is plural for pseudothecium. More and pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More, on infected canola residue. PseudotheciaFruiting bodies produced by fungus which cause disease (as the L. maculanspseudothecia can be produced on infected canola residue, which can then infect live canola plants nearby). It is plural for pseudothecium. More may continue to be produced on infected residue for several years, or until the infected residue breaks down.
Small microscopic sexual spores, called ascosporesMicroscopic asexual spores that typcially become airborne. More, are released from the pseudotheciaFruiting bodies produced by fungus which cause disease (as the L. maculanspseudothecia can be produced on infected canola residue, which can then infect live canola plants nearby). It is plural for pseudothecium. More and become airborne, resulting in long distance dispersal of the disease to newly planted canola crops. AscosporesMicroscopic asexual spores that typcially become airborne. More are considered the primary inoculum. They initiate the disease and serve to spread new races of the pathogenA disease-causing organism (such as a fungus or bacteria). More. AscosporesMicroscopic asexual spores that typcially become airborne. More are dispersed by wind and rain, and can be released from infested residue for at least three years. Ascospore release can begin as early as May, causing leaf lesions from the seedling stage onward.
During the growing season, the pathogenA disease-causing organism (such as a fungus or bacteria). More also produces another type of fruiting body called pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More that appear as pepper-like spots within lesions. From the pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More ooze masses of tiny spores called pycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More. These spores spread short distances by rain splash and wind, and cause secondary infection within a crop. Infected stubble can continue to produce pycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More for three to five years.
Seedling infection may arise from infected seed or from airborne and rain-splashed spores. Plants infected at this stage are usually the most severely affected (resulting in higher disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More and severity at maturity) 3 and may be lost or stunted. Seed-treated certified seed can control seed-borne blackleg infection, which will reduce the chance of new pathogenA disease-causing organism (such as a fungus or bacteria). More races being introduced to the field with the seed. Newer seed treatments protect the cotyledon to one-leaf stage from airborne blackleg infection, but do not protect plants from later infection by airborne spores.
Once the leaves are infected, the fungus grows down the petiole into the stem, eventually leading to the most damaging phase of the disease, stem cankering, usually at ground level. This restricts moisture and nutrient movement in the plant. Flowering plants may lodge as the fungus girdles stem bases, often completely severing or cutting off the plant.
The earlier plants are infected, the greater the likelihood of the development of basal stem canker. Infection before the two-leaf stage is often associated with serious yield loss. Infections initiated after the two-leaf stage usually cause less damage than early infections. Less severely affected plants may survive but produce less seed of poor quality. Wounded stems from hail or insects may be infected directly from spores germinating in the wound.
Influence of environment
Climate has an impact on blackleg incidence and severity. Regions with warm, humid conditions and frequent rain showers tend to have higher incidence and severity of blackleg. Warm, dry conditions slow disease development, while prolonged moist weather favours rapid spread and development. However, the disease is present in drier regions of the Prairies and can be a problem, even in dry years, provided early season showers occur to disperse the spores for initial infection.
Infection of canola is due to ascosporesMicroscopic asexual spores that typcially become airborne. More and pycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More of the pathogenA disease-causing organism (such as a fungus or bacteria). More, whose production and dispersal depends on environmental factors such as rain and wind. Therefore, the severity of blackleg disease can vary between regions and years. AscosporesMicroscopic asexual spores that typcially become airborne. More are responsible for the spread of new races of the pathogenA disease-causing organism (such as a fungus or bacteria). More, as these are wind-borne and can move relatively long distances (at least 10 kilometres). Although most blackleg infection comes from canola residue within the same field, ascosporesMicroscopic asexual spores that typcially become airborne. More and even some pycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More that become air-borne after rain-splash may land on canola or volunteer canola plants in nearby fields to begin new infections. In the management of blackleg disease, it is recommended that canola crops in western Canadian should be separated from fields that were planted to canola the previous year by at least 50 to 100 metres.
Research at the University of Manitoba found that peak ascospore and pycnidiospore dispersal was associated with rain events. Peak ascospore dispersal occurred several hours after rainfall of greater than two millimetres and persisted for approximately three days after such events. Peak pycnidiospore dispersal occurred during the same time as the rainfall. Spores are spread by wind, with more spores carried in the direction of prevailing winds. PycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More were found to travel further than first thought (45 metres) and may even move by wind. The research also found that on days without rain, air temperature and relative humidity (RH) had an effect on spore dispersal. More ascosporesMicroscopic asexual spores that typcially become airborne. More and pycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More were trapped between 9:00 pm and 4:00 am, when temperatures were between 13 to 18 degrees Celsius and RH was greater than 80 per cent, than at other times of the day, on days without rain 4.
Blackleg and yield loss
Research from the University of Alberta/Alberta Agriculture & Forestry found that for every unit of increase in disease severity, a 17.2 per cent loss in plant seed yield can be expected 5. Pod number and seed yield both declined linearly as blackleg severity increased. An updated yield loss model from the University of Alberta indicated that yields dramatically decrease when disease severity is equal to or greater than a rating of two 6. Use the online tool to calculate your blackleg yield loss.
Identifying blackleg
Symptoms
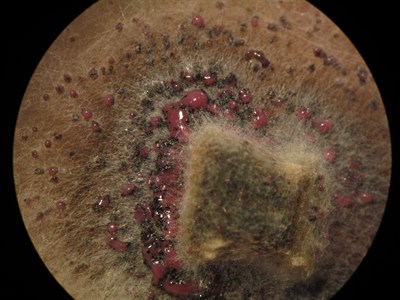
Disease lesions may occur on cotyledons, leaves, stems and pods. Leaf spots are dirty white, round to irregularly shaped, and usually dotted with numerous small, black pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More. PycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More appear as tiny round specks that may be seen more easily with the aid of a hand lens. Under moist weather conditions, a viscous pink liquid carrying the pycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More oozes from the pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More. Canola plants are susceptible to blackleg infection throughout their lifecycle (all growth stages).
On stems, blackleg lesions can be quite variable, but are usually found at the base of the stem or at points of leaf attachment. Stem cankers, which occur most frequently if infection of plants happens before the two-leaf stage, are associated with serious yield loss. Stem lesions may be up to several inches in length, and are usually white or grey with a dark border. Numerous pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More form in the center of the lesion. Basal stem lesions may also appear as a general blackening at the base, again with numerous pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More, with stem tissue constricted or pinched at the soil surface.
Lesions may also be observed where physical damage has occurred. Damage due to hail, insect feeding, tire tracks, etc. may provide an open wound where the pathogenA disease-causing organism (such as a fungus or bacteria). More can successfully establish, resulting in infection and lesion development.
Severe infection usually results in a dry rot or canker at the base of the stem. If basal infection begins early, stem cankers may appear from flowering onwards. As the season progresses, cankers penetrate, deepen and may girdle stem bases, often completely severing the plant. By mid-July, plants ripen prematurely and may lodge. Less severely affected plants remain standing but have restricted moisture and nutrient flow. Slicing through the canker or base of the root perpendicular to the stem should reveal black and/or brown necroticA symptom of plant cell or plant tissue death. More discolouration in the interior of the root of plants affected with virulentA form which causes infection (and can result in a disease) More blackleg.
PseudotheciaFruiting bodies produced by fungus which cause disease (as the L. maculanspseudothecia can be produced on infected canola residue, which can then infect live canola plants nearby). It is plural for pseudothecium. More, which are usually slightly larger than pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More, may form on the canola residue within the basal stem cankers. This occurs as a result of crossing of alternate mating types and takes place in autumn or the spring of the following year.
Pods and seeds may also be infected, although this is uncommon to find in Canada, especially in resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Infected pods ripen and shatter easily at harvest, resulting in seed loss. The seed formed close to a pod lesion may be sunken or shriveled and pale grey. The earlier pod infection occurs, the less likely that viable seeds will be produced (as the fungus infects the seed coat and embryo). The weakly virulentA form which causes infection (and can result in a disease) More species L. biglobosa usually infects plants later in the growing season as the crop matures, resulting in shallow stem lesions but rarely forming cankers that girdle the stem and cause yield loss.
Scouting tips
Scouting is an important management tool for blackleg management. There are four main scouting periods for blackleg disease during the growing season:
- Prior to planting:
- Examine canola residue to see if pseudotheciaFruiting bodies produced by fungus which cause disease (as the L. maculanspseudothecia can be produced on infected canola residue, which can then infect live canola plants nearby). It is plural for pseudothecium. More or pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More are present.
- Vegetative stage (cotyledon to two-leaf)
- Scout young canola plants for leaf lesions.
- If lesions are found, an early season, foliar fungicide application may be warranted.
- Vegetative stage (three to six-leaf)
- Scout using a ‘w- pattern’ through field, starting at field edge. Focus on edges where new canola residue may be present in adjacent field.
- Examine at least 50 plants for the presence of lesions. If lesions are found on more than 10 per cent of plants, then there is a risk of significant disease development.
- Look for pinching of the root at the soil surface; roots may appear hardened and woody with pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More starting to develop.
- At swathing:
- The best time to scout for the disease as the basal cankers, which cause significant yield loss, are easy to see.
- Pull up at least 50 plants in a w-pattern as described above.
- Clip with clippers at the base of stem/top of root and look for blackened tissue inside the crown of the stem. The amount of infection present will help identify the level of risk and the best management practices for that field in following years.
- Use the zero to five blackleg disease rating system to identify severity of the infection along with the incidence of the disease.
Testing
Suspected plant samples can be submitted to diagnostic labs to identify if blackleg species are present. Specific tests can also determine if Leptosphaeria maculans or Leptosphaeria biglobosa is the causal agent. Blackleg raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More identification testing is also available through several labs.
Manitoba
Follow these blackleg sampling guidelines to submit samples to the Pest Surveillance Initiative (PSI) lab.
As the founding supporter of the Pest Surveillance Initiative, Manitoba Canola Growers are offering free testing for their members. Agronomists are welcome to submit tests on a farmer’s behalf. See more details from the Manitoba Canola Growers.
For general diagnostics options (including plant diseases, weeds and insects) in Manitoba, visit Manitoba Agriculture’s Crop Diagnostic Services.
Saskatchewan
For visual diagnosis of plants, submit suspicious samples to the Crop Protection Laboratory. To submit, send a complete sample (the entire plant, preferably more than one) along with the proper form with as much information filled out as possible to the Crop Protection Laboratory. Note: when downloading and using the Internet version of the form, four copies of the fully completed form are required, along with the sample, for submission to the laboratory. For more information or to submit a complete sample:
Crop Protection Laboratory
346 McDonald Street
Regina, Saskatchewan
S4N 6P6
(306) 787-8130
Alberta
Producers finding suspicious plants can contact and send samples to one of four commercial labs (20/20 Seed Labs, SGS Biovision, Discovery Seed Labs or Exova Edmonton) for testing. Samples free of charge can be submitted by Alberta Agriculture and Forestry staff, Applied Research Associations, Agricultural Fieldmen, or by Municipal Pest Management to the Alberta Plant Health Lab.
Alberta Plant Health Lab
Crop Diversification Centre North
17507 Fort Road NW
Edmonton Alberta T5Y 6H3
780-638-3999
planthealthlab@gov.ab.ca
Labs
20/20 Seed Labs
Alberta Laboratory
507 11th Ave
Nisku, AB T9E 7N5
(780) 955-3435
Manitoba Laboratory
3489 Pembina Hwy
Winnipeg, MB, R3V 1A4
(204) 261-3755
SGS Canada
Sherwood Park Laboratory
#310 – 280 Portage Close
Sherwood Park, AB T8H 2R6
1-800-952-5407
Grande Prairie Laboratory
Unit 106, 10136 128 Ave
Grande Prairie, AB, T8V 1E9
1-877-532-8889
Discovery Seed Labs
450 Melville Street
Saskatoon, SK S7J 4M2
(306) 249-4484
Pest Surveillance Initiative (PSI)
5A 1325 Markham Road
Winnipeg, MB R3T 4J6
(204) 813-2171
Management
Managing blackleg disease of canola in western Canada requires an integrated management strategy including agronomic best practices and stewardship of blackleg resistance. Reliance on any one strategy alone, such as genetic resistance, is not a viable long-term strategy. The concerns and issues related to blackleg management go beyond yield, quality and resistance stewardship, to international market barriers. Follow these steps to help control blackleg 7.
Blackleg is a disease that can increase with some crop management choices, therefore assessing your risk is important. For example, growing canola in a tight rotation (canola more frequently than one in three years) will increase the risk for the development of blackleg and the risk of blackleg resistance being eroded or overcome.
For an integrated management strategy that utilizes a range of tools to minimize the risk to canola, utilize the Blackleg Management Guide.
Scout for disease
It is important to accurately identify the disease, as there are other diseases that can be confused with blackleg (e.g. sclerotinia, verticillium stripe, grey stem, root rots).
Look for blackleg early in the season and just prior to crop maturity. Fields that are highly infected indicate that action should be taken to reduce the risk of blackleg. Highly infected fields indicate that the rotation may be too short, which results in the buildup of large amounts of infected residue. It may also indicate that the resistance carried by the cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More grown is no longer effective and different cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More should be selected in future years.
Crop rotations
From a blackleg management standpoint, crop rotation allows for the decomposition of infected canola residue, reducing the spores available to infect the next canola crop. This could require a delay of up to three years between canola crops. Even where disease issues are less severe and genetic resistance is still working reasonably well, trends in yield and economic returns suggest canola should not be grown more often than one year in three. This is important not only for blackleg disease management, but also to maintain and protect the blackleg resistance present in canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More.
Crops should be isolated from infected fields, and the recommendation is that canola crops should be planted at least 50 to 100 metres from each other in western Canada. Reducing the movement of ascosporesMicroscopic asexual spores that typcially become airborne. More and pycnidiosporesMasses of tiny spores (such as those from the blackleg-causing Leptosphaeria maculans pathogen, the which ooze out from pycnidia in a viscous pink liquid). More between fields may be accomplished by maintaining an effective buffer distance between future canola fields and fields containing infected residue. Fields on which canola has been grown in the previous two to three years will likely contain significant quantities of canola residue that may harbour the pathogenA disease-causing organism (such as a fungus or bacteria). More until this residue fully decomposes.
Resistant cultivars
Grow B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More that carry at least a moderately resistant (MR) blackleg disease resistance rating, or better yet an ‘R’ rating. Resistance reduces blackleg infection to specific races of L. maculans, but does not mean the cultivar is immune to the disease.
A rotation study conducted at Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More (AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More) in Melfort and Scott, Saskatchewan from 2000-2006 found that blackleg severity of canola was reduced significantly as the length of rotation increased from two to four years even when growing an R-ratedResistant rating used to describe the level of resistance to plant disease, such as blackleg or clubroot. More hybrid. Intensive rotations of every two or even three years duration will increase the risk that disease resistance will break down and that disease severity will increase over the longer term 8.
See the Current canola cultivar trait labels table for a list of cultivars and their corresponding resistance field labels and resistance groups.
Resistance stewardship
As a result of the introduction of resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, changes in the races of the pathogenA disease-causing organism (such as a fungus or bacteria). More have been observed. New races have been isolated that can overcome the resistance of some Australian, European and Canadian canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Most of the resistance genes identified at the present time have been overcome in one or more continents.
The use of management strategies such as cultivar selection or rotation of resistance genes over time, in combination with good quantitative resistance and best agronomic practices such as crop isolation, and four-year rotations of canola crops to minimize the impact of these new strains, should provide the protection necessary for successful canola production. Stewardship of blackleg resistance and agronomic practices designed to control blackleg are recommended to be used together 2, 7, 9, 10.
To learn more about an integrated blackleg management strategy, see the Blackleg Management Guide.
Rotate cultivars
Many genes are responsible for blackleg resistance in canola and at least 16 described virulent L. maculans races are currently found on the Prairies. These races and populations are highly dynamic. Some seed companies are identifying their resistance genes for blackleg, but not all. Growing the same cultivar repeatedly on the same field will select for races of L. maculans that can overcome genetic resistance in that cultivar. The cultivar Q2, for example, went from an ‘R’ rating when it was originally launched to an ‘MS’ (moderately susceptible) or ‘S’ (susceptible) rating in some geographic areas today, because races of L. maculans virulentA form which causes infection (and can result in a disease) More to this resistance carried by Q2 became more predominant. There are other examples of resistance genes failing because of changing races of L. maculans too. Rotating cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More or resistance gene groups creates the opportunity to bring a mix of resistance genes to the field over time, which can reduce selection pressure and improve durability.
Certified seed
With certified seed, each canola plant should have the same blackleg resistance genes and be equally resistant. With bin run seed from hybrids, this second generation (F2) seed may not have inherited the same mix of resistance genes, and therefore may be more at risk to blackleg. As well, seed treatments on certified seed can control seed-borne blackleg infection, which will reduce the chance of blackleg being introduced to the field with the seed. But keep in mind that seed treatments do not protect seedlings and adult plants from later infection.
Weigh the benefit of fungicide
Seed treatments
Use of a seed treatment can protect plants during the critical infection window (cotyledon or early true leaves) from blackleg 3. New fungicide seed treatments may replace foliar application and can protect plants early in the season when blackleg is most harmful to yield. Seed treatments can reduce the disease severity substantially on susceptible cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More or cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More no longer acting resistant 11. Most fungicide products within seed treatments do not have protection against airborne blackleg, until 2021 when succinate dehydrogenase inhibitor (SDI) fungicides were launched.
Table 1. Seed treatments for blackleg in canola
Source: 2024 Saskatchewan Ministry of Agriculture Guide to Crop Protection
Foliar Fungicides
Use Table 2 to search foliar fungicide product names registered in Canada. They only have protectant activity and little or no eradicant activity, so ideally fungicides should be applied before blackleg symptoms are present. Consult the label or a current guide to crop protection for more information on registered products, rates, etc.
In an AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More rotation study from Melfort and Scott, there was generally little or no reduction in blackleg severity or improved yield when applying a fungicide on ‘R’ rated cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Even on highly susceptible cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, fungicides typically only depressed incidence by 20 per cent and severity ratings by about one severity point, which often did not translate into a significant or economical yield benefit 8.
Table 2. Foliar fungicides for blackleg in canola
Source: 2024 Saskatchewan Ministry of Agriculture Guide to Crop Protection
Control weeds and volunteers
Canola volunteers and related weed species such as wild mustard can host blackleg and other canola diseases in non-canola years. If not controlled, these volunteers and weeds act as a disease bridge, reducing the effectiveness of rotation and cultivar resistance for disease management.
No solid benefit from tillage or burning
Published studies from AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More at Melfort, Saskatchewan looked at tillage and burning as ways to manage canola diseases. Neither measure significantly reduced blackleg severity in canola in that region of the Prairies. The report concluded that differences in disease severity among tillage treatments were minimal, and the use of burning was ineffective 12.
Consider your climate
Blackleg incidence and severity tend to be higher in regions that are humid and warm with frequent rain showers. Therefore tight rotations in areas with conditions conducive to disease development will tend to be less sustainable, from a blackleg management perspective, than tight rotations in areas that are less humid with lower rainfall.
While most blackleg infection comes from canola residue within the same field, blackleg ascosporesMicroscopic asexual spores that typcially become airborne. More can be dispersed by wind. Therefore, canola growing in fields that have never had canola can still be infected from infested fields immediately adjacent. In that case, infection tends to be highest in canola plants closest to this infested field.
Genetic resistance
Blackleg resistance has been available in commercial canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More since the early 1990s, which has protected the crop against the predominant races of L. maculans. The use of a race-specific resistance strategy to manage this disease has been of great value in Canada, Australia and Europe. However, large-scale surveys in Europe and the more limited analysis of Canadian and Australian populations of the pathogenA disease-causing organism (such as a fungus or bacteria). More indicate that races with the ability to overcome most of the known specific resistance (R) genes can be found in many pathogenA disease-causing organism (such as a fungus or bacteria). More populations. In some cases the virulence of the local pathogenA disease-causing organism (such as a fungus or bacteria). More population changed in a matter of three years (two cycles of selection) to defeat the resistance. Stewardship of blackleg resistance and agronomic practices designed to control blackleg must be managed together 2, 7, 9, 10, 13.
Blackleg testing protocols and resistance ratings
The Western Canada Canola/Rapeseed Recommending Committee (WCC/RRC) has established Blackleg Testing Protocols and ratings to assess resistance. The resistance ratings currently available for commercial cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More are based on their field performance, in terms of disease incidenceThe severity of the plant disease on a specific plant (referred to as a percentage of plants infected). More and severity relative to check cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More in disease testing sites. Resistance reduces infection to certain races of L. maculans, but does not provide immunity. Normally the final resistance rating is based on trials from at least three locations each year in western Canada 14.
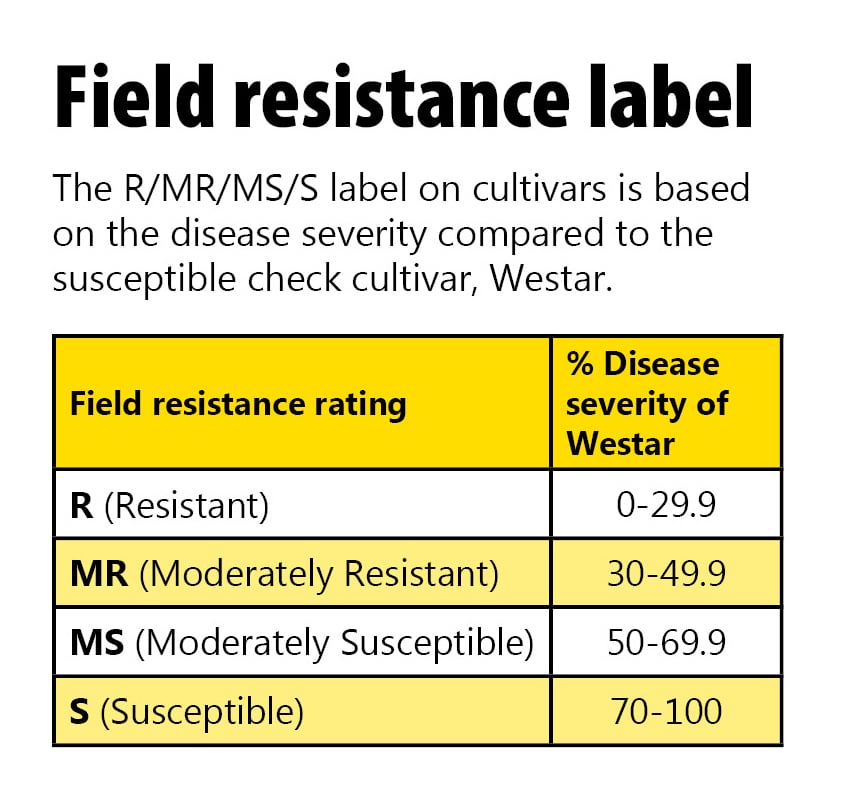
The following scale is used to describe the level of resistance based on average severity ratings compared to the highly susceptible cultivar Westar:
R (Resistant) = up to 30 per cent of the severity of Westar
MR (Moderately Resistant) = 30-49 per cent of the severity of Westar
MS (Moderately Susceptible) = 50-69 per cent of the severity of Westar
S (Susceptible) = 70-100 per cent of the severity of Westar
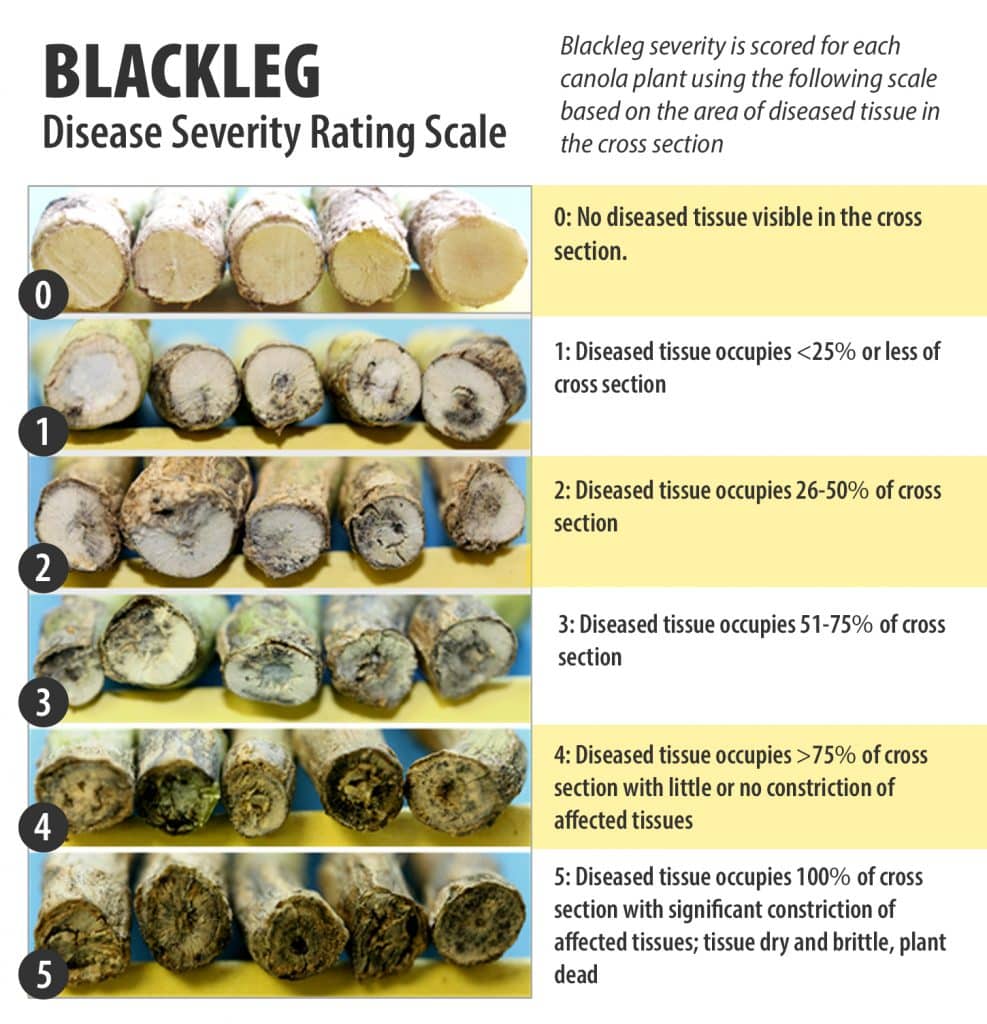
The severity of blackleg infection is evaluated on a minimum of 100 plants averaged over four replicates at crop maturity. Individual plants are uprooted, cut through the basal part of the stem and scored on the basis of the amount of disease using the following zero to five (0-5) scale.
0 – No diseased tissue visible in the cross-section.
1 – Diseased tissue occupies up to 25 per cent of the cross-section.
2 – Diseased tissue occupies 26-50 per cent of the cross-section.
3 – Diseased tissue occupies 51-75 per cent of the cross-section.
4 – Diseased tissue occupies greater than 75 per cent of the cross-section with little or no constriction of affected tissues.
5 – Diseased tissue occupies 100 per cent of the cross-section with significant constriction of affected tissues; tissue dry and brittle; plant dead.
For the trial to be valid, the severity rating of Westar must score between 2.6 and 4.5.
Development of commercial cultivars resistant to blackleg
Resistance to L. maculans in Brassica spp. is of two types:
quantitative (or minor gene or adult plant resistance) and
qualitative (or major gene or race-specific) resistance.
Quantitative resistance is expressed at the adult plant stage as reduced development of necroticA symptom of plant cell or plant tissue death. More tissue at the stem base compared to that found in susceptible cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, and is believed to be due to many genes, each with a relatively small effect. Qualitative resistance in Brassica spp. is usually effective at the site of initial infection on the cotyledons and leaves and is controlled by race-specific resistance genes. A resistant response is presumed to result from recognition of the corresponding L. maculans avirulence genes in a gene-for-gene manner, according to the theory developed by Harold Flor in 1946. In Canada, all commonly grown cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More of B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More canola and high erucic acid rapeseed carry moderate to high resistance to L. maculans, although the type of resistance or the specific genes for resistance present in a cultivar are still becoming better known.
Canola breeders likely use both quantitative and qualitative resistance to develop blackleg resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Qualitative disease resistance results from the interactions between products of pathogenA disease-causing organism (such as a fungus or bacteria). More avirulence (Avr) genes, such as those from L. maculans that causes blackleg, and matching resistance (R) genes in the plant. Therefore, if a pathogenA disease-causing organism (such as a fungus or bacteria). More has an Avr gene and tries to infect a host plant with the corresponding R gene, the R gene recognizes the pathogenA disease-causing organism (such as a fungus or bacteria). More and causes the plant to initiate a defense or immune system response. Resistance only results if both the Avr gene in the pathogenA disease-causing organism (such as a fungus or bacteria). More and the R gene in the host are present. Otherwise a susceptible reaction results and disease symptoms are observed.
Research at AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More Melfort, Saskatchewan and the University of Manitoba have isolated and identified new virulentA form which causes infection (and can result in a disease) More races of L. maculans, threatening the current blackleg resistance available in current cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. A recent study of western Canadian isolates indicated variation for eight of the ten Avr genes tested in a sample of 96 L. maculans isolates. In total, 16 races of L. maculans were identified based on ten Avr genes, with seven races accounting for 90 per cent of the isolates. There were similarities in the frequency of various Avr genes between the European and the Canadian studies, but also major differences. Unfortunately, surveys in Europe, Canada and analysis of Australian isolates of the pathogenA disease-causing organism (such as a fungus or bacteria). More have identified races of L. maculans with the ability to overcome most of these R genes 2, 7, 9, 10, 13. Using a specific set of races of L. maculans, the specific R genes in cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More or breeding material of B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More and other Brassica spp. can be determined. Knowledge of host R genes and pathogenA disease-causing organism (such as a fungus or bacteria). More Avr genes is beneficial to the development of blackleg disease resistance strategies that might prolong the effectiveness of specific resistance genes. Knowledge of the frequency of Avr genes in the pathogenA disease-causing organism (such as a fungus or bacteria). More population across the Canadian Prairies and in other oilseed rape growing regions of the world will allow pathologists, breeders and farmers to consider strategies to manage specific resistance.
R-gene rotation
Current labels for blackleg are based on field ratings of blackleg in comparison to a susceptible cultivar (Westar) for blackleg. Typically only resistant (R, zero to 29.9 per cent of Westar) and moderately resistant (MR, 30-49.9 per cent of Westar) are on the market. In recent years, some growers have noticed increased blackleg severity in their R and MR-rated cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. This is where the addition of more detailed labels will help to provide more accurate information.
Using the same disease resistance genetics over and over causes a shift in pathogenA disease-causing organism (such as a fungus or bacteria). More population, which can then overcome the resistance in available cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More – similar to herbicide resistance in weeds. Knowing the resistance genetics used in previous years will allow growers to rotate to a different resistance gene and reduce the blackleg infection within a field. As many as 10 new blackleg resistance labels will be applied to cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More in the coming years. They will use these letters: A, B, C, D, E₁, E₂, F, G, H, X to identify major resistance genes present.
Labels to identify the resistance group (RG) based on the major genes in a canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More:
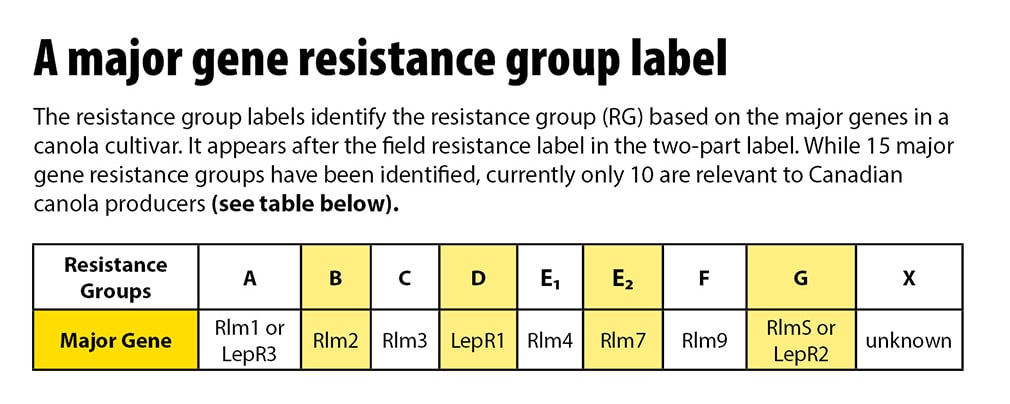
RG A = Rlm1 or LepR3*
RG B = Rlm2
RG C = Rlm3
RG D = LepR1
RG E1 = Rlm4**
RG E2= Rlm7**
RG F = Rlm9
RG G = RlmS or LepR2
RG X = unknown
Notes:
*Rlm1 and LepR3 have been placed into RG A because Canadian research indicates that these two genes generally recognize both AvrLm1 and AvrLep3 genes in the pathogenA disease-causing organism (such as a fungus or bacteria). More.
**Rlm4 and Rlm7 have been placed in two subgroups of RG E because they both typically recognize the AvrLm4-7 gene. The pathogenA disease-causing organism (such as a fungus or bacteria). More can be virulentA form which causes infection (and can result in a disease) More towards Rlm4 and still avirulentA form which doesn’t infection (or result in a disease) More towards Rlm7, but the reverse is less common. Therefore, the Blackleg Management Guide recommends that one can rotate Rlm4 (RG E1) to Rlm7 (RG E2) but the reverse is discouraged.
No resistance groups have used these letters: I, M, O, R, or S – to prevent confusion with the R/MR/MS/S system or 0.
How to use the labels
Blackleg management starts with scouting and identifying the disease on the previous canola crop’s stubble. If a grower has a rotation break longer than two years without canola and has been growing R-ratedResistant rating used to describe the level of resistance to plant disease, such as blackleg or clubroot. More cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, the risk of severe blackleg infection is minimal. But if a grower uses a tighter canola rotation and the disease is present and/or severe, the new additional label will come into play.
Picking an R-ratedResistant rating used to describe the level of resistance to plant disease, such as blackleg or clubroot. More cultivar with at least one different resistance gene group (to what was used previously) will help to provide protection from the aggressive, new virulentA form which causes infection (and can result in a disease) More blackleg race(s) within the field. In those cases where the disease is not present/evident in the field, then changing resistance groups would not be necessary.
The most effective means for reducing blackleg disease is reducing inoculum or spore production in a field. This is accomplished by lengthening the break between canola crops in a field. But spores can also migrate from adjacent fields. Under these circumstances, using a canola cultivar with a different resistance gene can be very beneficial in protecting the field and reducing blackleg disease.
In conclusion, the most important step is to scout for disease. If a grower does not have blackleg, they should choose cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with the highest probability of profitability and reduced production risk on their farm. If a grower does have blackleg disease that is increasing, they should use the tools available to manage and reduce the disease. Switching to cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with a different resistance gene(s) is one tool.
Examples of how the new blackleg label will work
Cultivar alpha: R (BC)
The traditional R rating means average field performance of blackleg resistance was below 30 per cent of Westar, the susceptible check. The additional “(BC)” designation means the cultivar contains the resistance genes Rlm2 and Rlm3.
Cultivar beta: MR (A)
The traditional MR rating means average field performance of blackleg resistance was 30-49.9 per cent of Westar check. The additional “(A)” means it contains the resistance gene LepR3 or Rlm1.
Cultivar Charlie: R (CX)
As an R-ratedResistant rating used to describe the level of resistance to plant disease, such as blackleg or clubroot. More cultivar, average field performance of blackleg resistance was below 30 per cent of Westar check. “(CX)” means it contains the resistance gene Rlm3 and an unidentified major resistance gene.
Reminder: The use of blackleg R-gene labels is a voluntarily process for seed companies to include on their cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. The industry is committed to providing the best genetics and advice to producers, so it will take some time to effectively deploy and incorporate these labels. Talk with the Canola Council of Canada agronomy specialist in your region for more information.
Learn more in Understanding Blackleg Resistance and the Blackleg Management Guide.
Blackleg race identification tests
Steps for the test
Step 1. Sample collection
The ideal time to pull and assess plants for blackleg infection is at 60 per cent seed colour change, which is around swath timing. Plants should still be green. Cut just below the crown of the plant into the root material to assess.
“Look for black discolouration within the hypocotyl tissue, often appearing in a wedge pattern. If a stem looks to be infected, keep a two to three inch piece for testing. Allow samples to air dry overnight before packaging to avoid excessive fungal growth.” From Discovery Seed Labs’ Blackleg Sampling Factsheet
Note: Old canola residue samples that contain pycnidiaA type of fruiting body (produced by a pathogen, such as the blackleg-causing Leptosphaeria maculans pathogen) that appears as pepper-like spots (which are spore-bearing structures) within lesions. More can be submitted, but these samples may show many other fungal species that are more saprophytic than L. maculans.
Step 2. Lab analysis
The lab will determine which blackleg species is present: L. biglobosa or L. maculans.
L. biglobosa, a mild or weakly virulentA form which causes infection (and can result in a disease) More species, is often associated with upper stem lesions and is widespread in western Canada. This species usually infects plants late in the season, rarely causing significant yield losses and is therefore considered a minor problem.
L. maculans is the virulentA form which causes infection (and can result in a disease) More species causing blackleg. It infects canola from the seedling stage onward, and progressively damages the plant by girdling stems and restricting moisture and nutrient uptake, leading to yield loss. Determining the species is difficult to complete visually. DNA must be taken to determine species.
Step 3: Interpreting the results
Lab reports typically report both the genotype and phenotype of the blackleg races identified from the samples collected.
- Genotype is the genetic content or genes/DNA present in the organism – in this case the blackleg fungus.
- Phenotype is the physical expression of genes; it may or may not be influenced by environment.
Producers need to look at the estimated phenotype when making cultivar selections. The phenotype is estimated because a physical expression test is not completed where different canola lines are grown out and tested with the inoculum collected. The phenotype is estimated based on published literature looking at gene interactions.
Lab reports will identify the predominant raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More of blackleg or give a frequency breakdown of blackleg races found within the samples collected. Major genes within canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More need to match up to avirulence genes (Avr) within the pathogenA disease-causing organism (such as a fungus or bacteria). More to be effective.
Example of lab results
Lab result, example one:
Phenotype: AvrLm4-5-6-7-11
Lab result, example two:
Phenotype: AvrLm2-4-5-6-7-11 (25%)
AvrLm4-5-6-7-11 (50%)
AvrLm1-4-5-6-7 (25%)
In example two, you want to find a major gene that corresponds with an avirulence gene found in all the races listed. If a diversity of blackleg races are presented, find a major gene that matches to the most frequent avirulence gene. In this example, avirulence gene 4 (AvrLm4) is found in all blackleg races identified. Using a cultivar deploying a major gene of Rlm4 (or Gr E1) would be a suitable match.
Application of lab results
Example scenario
A producer grew a canola cultivar from resistance group C and saw severe disease symptoms. So the producer submitted samples to a lab. The lab results showed Genotype AvrLm3-4-5-6-7-9-11 and Phenotype AvrLm4-5-6-7-11 present in the samples.
Phenotype results show five options to match a corresponding R-gene within a cultivar. The Avr genes are 4, 5, 6, 7 and 11, which means the producer can choose canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with one of these major genes: Rlm4, Rlm5, Rlm6, Rlm7, Rlm11.
Currently, the only major gene available on this list is Rlm4 (from resistance group E1). Cultivar options with Rlm4 or resistance group E1: CS2000, 6090RR, 6076CR, 5545CL, V14-1, V12-3, V12-1, V24-1, or V33-1CL. If the seed this producer wants does not provide this option, then the producer should still switch cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and look to grow a strong quantitative resistant cultivar.
The producer could take a risk and continue growing group-C cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, but if the environmental conditions are conducive to disease development, the crop is at risk for infection as the major gene being deployed (Rlm 3, in this example) does not match up to the predominant blackleg raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More in the field and therefore is not effective.
Testing labs for blackleg race identification
Labs can test fresh stems (taken from this year’s crop) or old stem pieces found in fields that will be seeded to canola next year.
Manitoba’s Pest Surveillance Initiative (PSI) Lab
PSI will provide a blackleg raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More identification test for about $200. Members of Manitoba Canola Growers, which helps fund the lab, can get one free test this year and then, like anyone else, pay $200 for extra tests if they want to check more than one field. Read more on how to take and submit samples.
Discovery Seed Labs
If stems seem to be infected, send in a two to three inch length of that infected stem. Include up to 12 stem samples. Discovery will test to confirm the presence of the blackleg pathogenA disease-causing organism (such as a fungus or bacteria). More, and it will test to identify the specific raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More. Read more.
20/20 Seed Labs
These labs have a blackleg stubble test and will also provide a raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More identification test upon request. See details at 20/20 Seed Labs.
Footnotes
- Fitt, B.D.L. , Brun, H., Barbetti, M.J., & Rimmer, S.R. 2006. World-wide importance of phoma stem canker (Leptosphaeria maculans and L. biglobosa) on oilseed rape (Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More). European Journal of Plant Pathology, 114, 3-15[↩]
- Kutcher, H.R., Yu, F., & Brun, H. 2010. Improving blackleg disease management of Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More from knowledge of genetic interactions with Leptosphaeria maculans. Canadian Journal of Plant Pathology, 32(1), 29-34.[↩][↩][↩][↩]
- Peng, G. 2023. Understanding the critical infection window that causes blackleg of canola in western Canada. Available on the Canola Research Hub.[↩][↩]
- Guo, X.W. & Fernando, W.G.D. 2005. Seasonal and diurnal patterns of spore dispersal by Leptosphaeria maculans from canola stubble in relation to environmental conditions. Plant Dis., 89, 97-104.[↩]
- Hwang, S., Strelkov, S.E., Peng, G., Ahmed, H., Zhou, Q., & Turnbull, G. 2016. Blackleg (Leptosphaeria maculans) Severity and Yield Loss in Canola in Alberta, Canada. Plants, 5, 31.[↩]
- Wang, Y., Strelkov, S.E., & Hwang, S.F. 2020. Yield losses in canola in response to blackleg disease. Canadian Journal of Plant Science, 0, 0. https://doi.org/10.1139/CJPS-2019-0259.[↩]
- Kutcher, H.R., Fernando, W.G.D., Turkington, T.K. & D.L. McLaren. 2011. Best Management Practices for Blackleg Disease of Canola. Prairie Soils & Crops Journal, 4, 122-134.[↩][↩][↩][↩]
- Smith, E.G., Favret, M.L., Brandt, S.A. & Kutcher, H.R. 2008. Economics of Shorting Canola Rotations. Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More poster.[↩][↩]
- Kutcher, H. R., Balesdent, M. H., Rimmer, S. R., Rouxel, T., Chèvre, A. M., Delourme, R., & Brun, H. 2010. Frequency of avirulence genes in Leptosphaeria maculans in Western Canada. Canadian Journal of Plant Pathology, 32(1), 77 – 85.[↩][↩][↩]
- Fernando, W.G.D. 2010. Managing Blackleg Resistance Breakdown and Trade Barriers through Blackleg Resistance Stewardship in Canola. Manitoba Agronomists Conference, December. University of Manitoba.[↩][↩][↩]
- Peng, G., Liu, X., Mclaren, D.L., Mcgregor, L. & Yu, F. 2020. Seed treatment with the fungicide fluopyram limits cotyledon infection by Leptosphaeria maculans and reduces blackleg of canola. Canadian Journal of Plant Pathology, 42(4), 480-492. https://doi.org/10.1080/07060661.2020.1725132. CARP 2015-12 CARP 2019-10.[↩]
- Kutcher, H.R. & Malhi, S.S. 2010. Residue burning and tillage effects on diseases and yield of barley (Hordeum vulgare) and canola (Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More). Soil & Tillage Research, 109(3), 153-160.[↩]
- Van de Wouw, A., Cozijnsen, A., Rayner, J., & Howlett, B. 2009. Monitoring of virulence in Australian populations of the blackleg fungus. School of Botany, University of Melbourne. (Slide deck).[↩][↩]
- Western Canada Canola/Rapeseed Recommending Committee (WCC/RRC). 2011. Procedures of the Western Canada Canola/Rapeseed Recommending Committee Incorporated for the Evaluation and Recommendation for Registration of Canola/Rapeseed Candidate CultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More in Western Canada. Appendix B: Disease Testing Protocols, 14.[↩]