

Browse the Canola Meal Feed Guide by chapter:

Canola meal is fed to all types of poultry throughout the world. The meal provides an excellent amino acid profile and is an alternative to, or complement to other protein ingredients such as soybean meal. Canola meal provides excellent value in diets where the greatest emphasis in formulation is placed on amino acid balance. Canola meal can also be a cost effective alternative to other proteins in high energy broiler diets. Care must be taken to formulate diets on a digestible amino acid basis to ensure performance is optimal when canola meal is included in diets for poultry.
| wdt_ID | DIET TYPE | INCLUSION LEVELS |
|---|---|---|
| 1 | Chick starter | Intakes may be reduced with inclusion over 20% |
| 2 | Broiler grower | High performance reported at 30% inclusion |
| 3 | Broiler finisher | High performance reported at 40% inclusion |
| 4 | Layers | No data beyond 24% |
| 5 | Broiler breeders | High performance to 30%. No data beyond 30% |
| 6 | Turkey starter | High performance to 24%. No data beyond 24% |
| 7 | Turkey grower | High performance to 24%. No data beyond 24% |
| 8 | Turkey finisher | High performance to 24%. No data beyond 24% |
| 9 | Ducks starter | High performance with 10%. More data needed |
| 10 | Ducks grower | High performance to 21%. No data beyond 21% |
| 11 | Geese starter | No data found |
| 12 | Geese grower | High performance with 16%. No data beyond 16% |
| 13 | Quail grower | High performance reported at 15% |
| 14 | Quail egg layers | High performance with 18.5%. No data beyond 18.5% |
| 15 | Ostrich | High performance with 20.0%. Only one trial |
The rate of production is the basis for ingredient comparison in most academic trials. However, in industry, cost/unit of gain is the integration of several factors, including ingredient cost, production, health, and survival. While feeding canola meal to poultry may not always result in the maximum rates of gain, there can be reductions in the cost/unit of production when canola meal is incorporated in diets.
In an elaborate study evaluating both rapidly growing and slowly growing broilers, Berger et al. (2021) showed that broiler chickens readily adapt to diets containing alternative protein sources to soybean meal and included canola meal in their evaluation. They determined that replacement may result in a slight increase in the cost of production (under 2%) due to greater feed intake but can result in major reductions in greenhouse gas emissions, often associated with the production systems used to produce and acquire soybean meal. While this may not consistently be the case, better information is being generated to allow sustainability to be predicted with greater accuracy.
Birds have been demonstrated to be averse to bitter compounds (Yoshida et al., 2022). Glucosinolates tend to impart a bitter taste and older varieties of canola meal, that contained higher concentrations of these compounds resulted in reduced intakes (Khajali and Slominski 2012).
In general, poultry maintain appropriate feed intake levels if given diets high in canola meal that are properly formulated for available amino acids. However, studies in raising poultry suggest that canola meal should be limited during the starter period to 20% for broilers and turkeys, and 10% for more less studied species such as ducks, geese and quail. Concentrations of 30% to 40% of the diet are readily tolerated at later stages of growth. Oryschak and Beltranena (2013) and Rogiewicz et al. (2015) demonstrated that proper diet formulation allows canola meal to be included at 20% of the diet with no negative effect on feed intake in laying hen diets. Feed intake was maintained for broilers fed up to 20% canola meal from days 1 to 35 of life (Naseem et al., 2006), and broiler growers can be given diets with up to 30% canola meal (Newkirk and Classen, 2002; Ramesh et al., 2006).
Using canola meal at high levels in poultry diets is best accomplished by balancing diets for available amino acids. Extensive research has been conducted in recent times to determine the standardized ileal digestibility (SID) of amino acids from solvent extracted canola meal. Results for broilers are provided in Table 1 and SID results for laying hens, turkeys and ducks are shown in Table 2.
Table 1. Standardized ileal digestibility (SID) of amino acids in canola meal for broiler chickens1.
| wdt_ID | AMINO ACIDS | AVERAGE, % (2) | STANDARD DEVIATION |
|---|---|---|---|
| 1 | Indispensable | ||
| 2 | Arginine | 87.23 | 2.33 |
| 3 | Histidine | 76.51 | 11.31 |
| 4 | Isoleucine | 82.66 | 3.71 |
| 5 | Leucine | 83.68 | 2.58 |
| 6 | Lysine | 79.32 | 3.06 |
| 7 | Methionine | 88.93 | 2.10 |
| 8 | Phenylalanine | 83.89 | 2.21 |
| 9 | Threonine | 76.48 | 3.68 |
| 10 | Tryptophan | 87.15 | 5.18 |
| 11 | Valine | 78.85 | 3.94 |
| 12 | Dispensable | ||
| 13 | Alanine | 82.96 | 3.04 |
| 14 | Aspartic acid | 77.59 | 4.17 |
| 15 | Cysteine | 76.47 | 3.77 |
| 16 | Glutamic acid | 89.00 | 3.54 |
| 17 | Glycine | 78.80 | 4.08 |
| 18 | Proline | 77.45 | 3.31 |
| 19 | Serine | 80.51 | 4.73 |
| 20 | Tyrosine | 82.66 | 5.28 |
1 Adewole et al., 2017; Agyekum and Woyengo, 2022; Ariyibi, 2019; Chen et al., 2015; Gallardo et al.; 2017; Kim et al., 2012; Kong and Adeola, 2013; Osho et al., 2019; Park et al., 2019, Ross et al., 2019
2 Average of 41 Values
Huang et al. (2006) found that there were no differences in apparent ileal digestibility of amino acids between broiler chicks, laying hens and adult roosters, which is not the case for all feed ingredients (Adedokun et al., 2009; Huang et al., 2006). Therefore, the values shown may be useful in extrapolating SID values for other poultry species.
Table 2. Standardized ileal digestibility (SID) of amino acids in canola meal for laying hens, turkeys and ducks1.
| wdt_ID | AMINO ACIDS | LAYING HENS (1) | TURKEYS (2) | DUCKS (3) |
|---|---|---|---|---|
| 1 | Indispensable | |||
| 2 | Arginine | 88.23 | 88.57 | 85.30 |
| 3 | Histidine | 82.97 | 79.67 | 81.75 |
| 4 | Isoleucine | 77.70 | 76.29 | 78.40 |
| 5 | Leucine | 80.63 | 78.51 | 83.50 |
| 6 | Lysine | 80.60 | 79.86 | 75.50 |
| 7 | Methionine | 88.67 | 84.19 | 88.60 |
| 8 | Phenylalanine | 81.70 | 83.49 | 84.00 |
| 9 | Threonine | 73.50 | 75.13 | 74.60 |
| 10 | Tryptophan | 82.30 | 95.00 | 87.40 |
| 11 | Valine | 77.73 | 74.39 | 77.55 |
| 12 | Dispensable | |||
| 13 | Alanine | 80.00 | 81.83 | 80.05 |
| 14 | Aspartic acid | 77.20 | 80.92 | 74.90 |
| 15 | Cysteine | 77.67 | 73.59 | 79.80 |
| 16 | Glutamic acid | 86.75 | 88.07 | 85.15 |
| 17 | Glycine | 76.70 | 82.01 | 75.55 |
| 18 | Proline | 75.70 | 75.68 | 83.10 |
| 19 | Serine | 75.60 | 80.04 | 82.05 |
| 20 | Tyrosine | 78.30 | 79.02 | 80.45 |
1 Goudarzi et al., 2017; Oryschak et al., 2020. Mean of 4 values;
2 Adedoken et al, 2008; Koslowski et al., 2011; Koslowski et al., 2018; Zhang et al, 2020. Mean of 28 values; 3 Kong and Adeola, 2013; Zhang et al, 2020. Mean of 30 values.
The SID values obtained with expeller extracted canola meal are provided in Table 3. Values are available from studies using broiler chickens only. Due to the similarities in SID values between broilers, laying hens, turkeys and ducks for solvent extracted canola meal, it is most likely that the SID values given in Table 3 can be used for all these commercial species until more information becomes available.
Table 3. Standardized ileal digestibility (SID) of amino acids in expeller extracted canola meal for broiler chickens1.
| wdt_ID | AMINO ACIDS | AVERAGE, % (2) | STANDARD DEVIATION |
|---|---|---|---|
| 1 | Indispensable | ||
| 2 | Arginine | 85.49 | 4.69 |
| 3 | Histidine | 74.97 | 12.14 |
| 4 | Isoleucine | 80.13 | 7.85 |
| 5 | Leucine | 81.41 | 3.96 |
| 6 | Lysine | 80.79 | 4.46 |
| 7 | Methionine | 87.60 | 4.09 |
| 8 | Phenylalanine | 83.00 | 4.29 |
| 9 | Threonine | 77.43 | 4.30 |
| 10 | Tryptophan | 83.27 | 8.37 |
| 11 | Valine | 78.79 | 4.53 |
| 12 | Dispensable | ||
| 13 | Alanine | 79.56 | 5.49 |
| 14 | Aspartic acid | 80.04 | 7.72 |
| 15 | Cysteine | 87.01 | 6.18 |
| 16 | Glutamic acid | 80.57 | 4.87 |
| 17 | Glycine | 76.84 | 3.97 |
| 18 | Proline | 79.97 | 3.73 |
| 19 | Serine | 77.93 | 6.94 |
| 20 | Tyrosine | 79.56 | 5.49 |
1 Agyekum and Woyengo, 2022; Bryan et al., 2019; Park et al., 2019; Kong and Adeola, 2016; Toghyani et al., 2014; Woyengo et al., 2010
2 Average of 19 samples.
The energy value of canola meal for poultry is lower than that of soybean meal, the most common vegetable protein used in poultry diets. In certain diets, where the energy value of the diets is of great importance, such as for broilers, canola meal inclusion levels may be restricted. Egg layer diets and early-phase, high-protein turkey diets based on least cost formulation may at times restrict canola meal inclusion levels if high energy ingredients are unavailable.
The values for AMEn shown in Table 4 for solvent extracted canola meal reflect results from recent experiments, and may differ from published values where older varieties, no longer available, were tested. Some of the variability may be associated with the season and/or the location where the canola was grown. However, Georgia researchers (Veluri and Olukosi, 2020; Wu et al., 2020) revealed that the reference diet used in the determination of energy, as well as the calculation method (difference or regression) can impact the values obtained and may account for some of the variability shown. The physical form and degree of processing also impact the energy value of the meal (Khalil et al., 2021).
Table 4. Energy values of canola meal for poultry (AMEn, Kcal/kg).
| wdt_ID | REFERENCE | SPECIES | AS FED BASIS | DRY MATTER BASIS |
|---|---|---|---|---|
| 1 | Adewole et al, 2017 | Broilers | 1,777 | 2,019 |
| 2 | Agyekum and Woyengo | 1,608 | 1,909 | |
| 3 | Chen et al.,2015 | 1,983 | 2,254 | |
| 4 | Gallardo et al | 1,822 | 2,071 | |
| 5 | Gorski et al, 2017 | 1,851 | 2,217 | |
| 6 | Jayaraman, 2016 | 2,144 | 2,437 | |
| 7 | Jia et al., 2012 | 1,810 | 2,057 | |
| 8 | Rad-Spice, 2018 | 1,834 | 2,084 | |
| 9 | Rahmani et al, | 1,789 | 2,032 | |
| 10 | Wise and Adeola 2022 | 1,763 | 2,003 | |
| 11 | Woyengo et al., 2010 | 1,584 | 1,801 | |
| 12 | Zhang and Adeola, 2017 | 2,011 | 2,286 | |
| 13 | Zhong and Adeola, 2019 | 1,689 | 1,919 | |
| 14 | Jia et al., 2012 | Layers | 1,936 | 2,200 |
| 15 | Oryschak et al, 2020 | 1,928 | 2,192 | |
| 16 | Kozlowski et al., 2018 | Turkeys | 1,886 | 2,143 |
| 17 | Jia et al., 2012 | 1,766 | 2,007 | |
| 18 | Noll et al., 2017 | 2,010 | 2,284 | |
| 19 | Wickramasuriya, 2015 | Ducks | 1,885 | 2,142 |
| 20 | Mandal et al., 2005 | Quail | 1,852 | 2,105 |
Table 5 provides AMEn values for expeller extracted canola meal. As the table indicates, the lipid content of the meal can be variable depending on the source, and is expected to impact the energy value of the meal.
Table 5. Energy value of expeller extracted canola meal for poultry (AMEn, Kcal/kg).
| wdt_ID | REFERENCE | SPECIES | AS FED | DRY MATTER BASIS | LIPID % OF DRY MATTER |
|---|---|---|---|---|---|
| 1 | Agyekum and Woyengo | Broilers | 1,994 | 2,265 | 15.3 |
| 2 | Bryan et al, 2019 | 2,623 | 2,997 | 11.4 | |
| 3 | Bryan et al., 2019 | 2,917 | 3,314 | 15.9 | |
| 4 | Sessingnong et al., 2022 | 2,043 | 2,322 | - | |
| 5 | Toghyani et al., 2014 | 2,258 | 2,566 | - | |
| 6 | Woyengo et al., 2010 | 2,370 | 2,694 | 12 | |
| 7 | Zhong and Adeola, 2019 | 2,584 | 2,937 | 18.1 | |
| 8 | Oryschak et al, 2020 | Layers | 2,556 | 2,904 | 13.2 |
The use of dietary enzymes is common in poultry feeds, especially those containing barley and wheat. These additives have been demonstrated to improve carbohydrate digestibility. Canola meal contains a significant portion of cell wall components that are undigested by poultry. Extensive research has been conducted at the University of Manitoba to investigate cell wall composition and non-starch polysaccharide (NSP) digestion with the inclusion of NSP degrading enzymes. Meng and Slominski (2005) examined the effects of adding a multi-enzyme complex (xylanase, glucanase, pectinase, cellulase, mannanase and galactonase) to broiler diets. The enzyme combination increased total tract NSP digestibility of canola meal, but no improvements were observed in other nutrient digestibility values or animal performance. Jia et al. (2012) fed broiler diets containing canola meal and a multi-carbohydrase enzyme to determine their effect on AMEn values and found a 6% increase in AMEn. Gallardo et al. (2017) calculated an 8% improvement in the energy value of canola meal. An improvement in AMEn of 6.6% with the use of multi-carbohydrase enzymes was witnessed and reported by Rad-Spice (2018). Most recently, Niu et al. (2022) determined that the inclusion of a multi-carbohydrase enzyme cocktail increased NSP digestion from zero to 20%. Bodyweight gain by broilers in the same study was improved by 5%. Although the data are not completely conclusive, moderate enhancement of canola meal digestion may occur. The enzymes may also improve the digestibility of other dietary ingredients.
The lipid content of canola meal is higher than many other vegetable protein sources, making a significant contribution to the energy value of the meal (Newkirk, 2011). A study by Barekatain et al. (2015) revealed that the digestibility of the oil in canola seed was as digestible as added canola oil is for broiler chickens.
The complete mineral and vitamin profile from canola meal is provided in Chapter 2. These values can be used as guidelines in formulations.
Canola meal is notably a rich source of phosphorus, which is a critical nutrient for all classes of poultry. In the past, only the non-phytate portion of the phosphorus in canola meal was assumed to be available, which is approximately 35% of the total phosphorus of the meal. Using the ileal digestibility technique, Mutucumarana et al. (2015) calculated that 47% of the phosphorus in canola meal was digestible by broilers, and that a portion of the phytate phosphorus was also digested by birds. In a more recent experiment, Munoz et al. (2018) using the precision fed rooster bioassay technique found phosphorus retention to be 44% of the total when intakes approached requirements, but declined when requirements were exceeded. The authors suggested a value of 38% based on their studies.
Phytase has been shown to be effective in improving phosphorus bioavailability in rapeseed meal varieties (Czerwiński et al., 2012) and more recently in canola meal for broilers. In an Australian study (Moss et al, 2018), the availability of phosphorus was increased from 32% to 52% with the inclusion of phytase and 69% when both phytase and xylanase were added. Phytase also improved the availability of calcium. A 40% improvement in phosphorus digestibility by broilers noted by David et al. (2021).
It is common practice to formulate poultry diets based on cation-anion balance. Diets are generally formulated to a positive cation-anion balance. Canola meal has a negative cation-anion balance and is high in sulfur, which can interfere with calcium absorption. Supplementing the diet with extra calcium helps to a certain extent, but care is advised, as too much dietary calcium can depress feed intake. Adding potassium bicarbonate to diets is a better alternative.
Unlike rapeseed meal, canola meal does not need to be restricted on the basis of the glucosinolate contribution to the diet. The very low levels of glucosinolates that are present in Canadian canola meal have eliminated concerns for this anti-nutrient in practical feeding situations.
Canola meal contains less potassium and more sulfur that soybean meal (Khajali and Slominski, 2012). Feed intake in broilers has been correlated with the cation-anion balance of the diet based on some pioneering investigations into feeding canola meal to poultry (Summers and Bedford, 1994). This can be overcome by providing diets with higher levels of potassium carbonate or sodium chloride. Improvements in understanding requirements of broilers have resulted in the development of routine formulation procedures that have permitted greater amounts of canola meal to be included in today’s diets for broilers. As noted, it is now common practice to formulate diets based on cation-anion balance.
In addition, formulating diets based on SID has resulted in weight gains that are nearly identical to those found with other protein ingredients, particularly during the grower period. Recent research suggests that up to 30% canola meal can be used in broiler diets. Gorski et al. (2017) provided starter diets (1-21 days of age) to broilers that contained 0, 10, 20, 30 and 40% canola meal. Weight gains were reduced with the 30 and 40% inclusion rates, due to lower feed intakes for these diets. Grower diets, provided from 21 to 37 days of age contained 0, 10, 20 or 30% canola meal. There were no differences in average daily gain or feed intake between diets during the growing period. Gopinger et al. (2014) formulated diets with 0, 10, 20, 30 and 40% canola meal, which was provided to the birds from 7 to 35 days of age. Growth rates were greater with the 10, 20 and 30% canola meal diets than with the soybean meal control but declined with the 40% canola meal diet to the same level as with soybean meal. Ariyibi (2019) fed diets to broilers that contained 6 incremental levels of canola meal ranging from 0 to 15% from 1 through 7 days of age, 0 to 18% from 7 through 14%, 0 to 25% from 14 through 21 days of age, and 0 to 35% from 21 through 28 days of age. Increasing levels of canola meal had no effect on growth performance. Looking at these three studies, canola meal inclusion levels of up to 20% for 0-7 days, 30% from 7-14 days and up to 40% at ages beyond are possible. Rad-Spice et al. (2018) concluded that canola meal can be used effectively and can replace soybean meal in diets for broiler chickens.
Canola meal is a commonly fed and economically effective feed ingredient in commercial egg layer diets. As with broiler diet formulation, SID amino acids must be considered. Early research, where diets were formulated on a crude protein basis, showed a reduction in egg weight when canola meal was substituted for soybean meal. Diets formulation on a crude protein basis resulted in insufficient lysine content in the canola meal diet (Kaminska, 2003). Previous published research by Novak et al. (2004) supported the hypothesis that insufficient lysine can affect egg weight. These researchers increased lysine intake from 860 mg/d to 959 mg/d and observed an increase in egg weight from 59.0 g to 60.2 grams. Another outdated concept suggested that feeding high levels of canola meal to brown-shelled egg layers could result in eggs with a fishy flavor. This was associated with a genetic error in the hens, and has since been resolved.
Oryschak and Beltranena (2013) demonstrated that when properly formulated, diets can include canola meal at 20% of the diet with no negative effects on egg production, hen health, egg quality or egg fatty acid content. As Figure 1 shows, egg weights and laying percentage were maintained for the duration of the 36 week-long study. There were also no differences in liver hemorrhage in the hens, and there was no detectible fishy odor in the eggs. Rogiewicz et al. (2015) similarly demonstrated excellent performance of hens fed 15-20% canola meal. Gorski (2015) provided hens from 33 to 49 weeks of age with diets containing 0 (soybean meal control) 8, 16, or 24% canola meal. They found no differences between treatments in feed intake, egg production, egg weight, or change in weight of the hens over the course of the 16-week study.
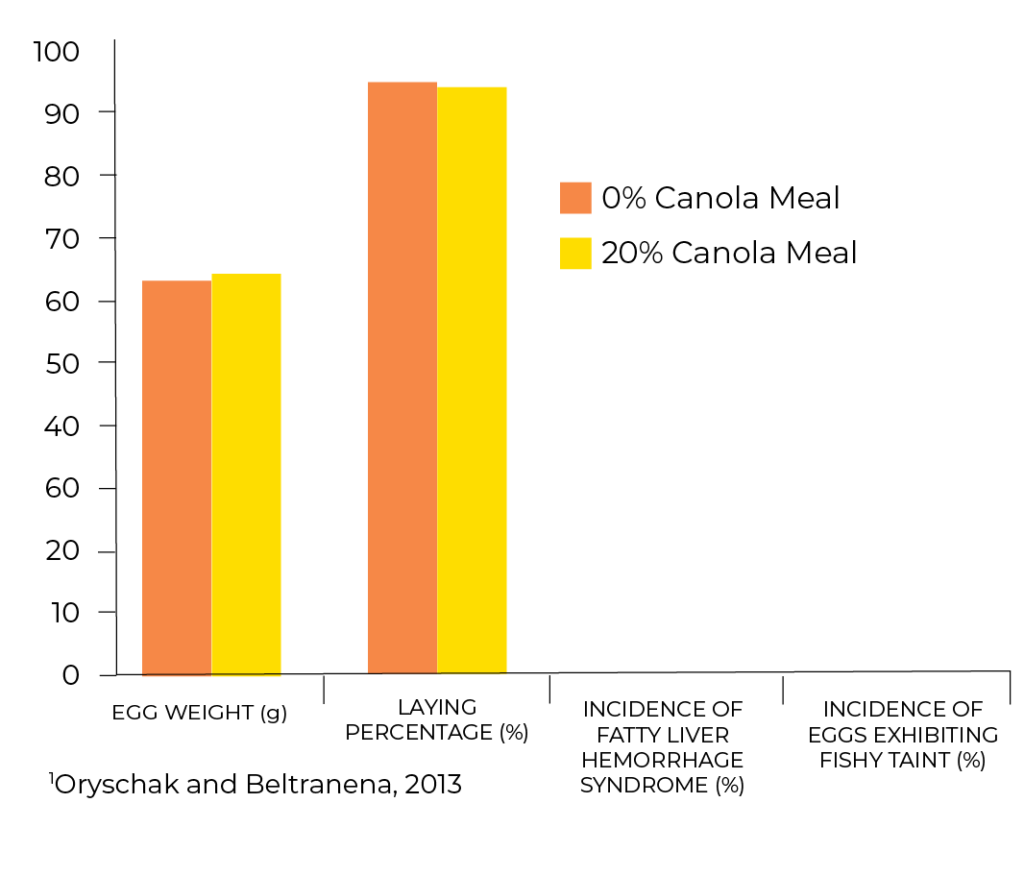
Figure 1: Performance results from feeding canola meal (CM) to laying hens on egg weight, laying percentage, incidence of fatty liver hemorrhage syndrome and presence of fishy taint in eggs. (Average over 36weeks of production)1.
In an experiment conducted by Dalhousie University (Savary et al., 2017), brown-egg laying hens were given diets containing soybean meal as the major protein source, or 10 or 20% canola meal. The experiment was analyzed for 4 feeding periods: 30 to 41, 42 to 49, 50 to 61 and 62 to 78 weeks of age. There were no statistical differences in egg production, feed efficiency, or mortality for any of the feeding phases. The researchers noted that there were no differences in egg quality or hen weights. A similarly designed follow up trial confirmed these results (Savary et al., 2019).
Most recently, a comprehensive trial conducted by Oryschak et al. (2020) clearly demonstrated that the inclusion of solvent extracted meal in the diet of brown-shelled egg layers at an inclusion level of 20* supported excellent lay performance and egg quality for a 36-week laying cycle. Based on these recent findings, canola meal can be fed effectively at elevated levels in laying hen diets without negatively affecting egg production, egg weight, egg quality or fatty acid content as long as the diets are formulated on digestible amino acid content.
There is limited new research on the use of canola meal in broiler breeders, likely because much of the results from laying hens are applicable to these birds. The high-protein and high-fiber content of canola meal makes it an ideal feedstuff to manage weight gain in broiler breeder birds. Older research showed that canola meal has no negative effects on egg fertility or hatchability of leghorn breeders (Kiiskinen, 1989; Nasser et al., 1985). A more recent study by Ahmadi et al. (2007) evaluated the effects of adding 0%, 10%, 20% or 30% rapeseed meal to the diet of broiler breeders. It is unclear as to what the glucosinolate content of the diets was; however, they concluded that rapeseed meal can be used effectively in broiler breeder diets without affecting production, egg weight or chick quality. Use of canola meal for broiler breeders can be justified due to the extensive information available for laying hens and other poultry.
Canola meal is an excellent protein source for growing turkeys. It is common commercial practice to feed high dietary concentrations of canola meal to growing and finishing turkeys. It has long been known that the key to successful use of canola meal for turkeys is to ensure that diets are balanced for amino acids. Early on, Waibel (1992) demonstrated that when canola meal was added at 20% of the diet without maintaining equal energy and essential amino acid levels, growth and feed conversion efficiency were decreased. However, when extra fat was added and amino acid levels were kept constant, performance was equal to or superior to the control diet. As with other species, it is important that diets be formulated on a digestible amino acid basis.
More recently, Kozlowski et al. (2018) verified that starter and grower diets with 20% canola meal resulted in growth rates that were not different than those obtained with soybean meal. Feed to gain was found to be slightly higher in the starter phase for the canola meal diet (1.43 for canola meal as compared to 1.36 for soybean meal) but this could be reduced to 1.37 with the inclusion of multicarbohydrase enzymes. There were no differences in average daily gain, feed intake or feed efficiency due to treatment over the length of the 8 week-long study. Similarly, Noll et al. (2017) provided starter turkeys with diets containing 0 (soybean meal control), 8, 16 or 24% canola meal. The researchers found no differences in any performance parameters measured. A follow-up shorter study, conducted during the very sensitive first 3 weeks of life noted that up to 24% canola meal could be provided to starter turkeys (Noll et al., 2017).
Commercially, canola meal is often included in turkey diets at levels beyond the 20% level. In this case, it is important to ensure the dietary electrolyte balance of the final diet is in the appropriate range. The dietary electrolyte balance of canola meal (Na + K–Cl) is approximately 307mEq/kg. However, canola meal contains a significant amount of sulfur, and this should also be considered. (Khajali and Slominski, 2012) recommend the equation (Na + K) – (Cl + S), which results in an electrolyte balance of approximately 100 mEq/kg.
Ducks and geese represent the third largest source of poultry meat, after chickens and turkeys. These birds are also prized for their eggs and feathers. Canola meal is commonly fed to ducks and geese, and with no reported issues resulting from the use of the meal.
Wickramasuriya et al. (2015) determined that the first limiting amino acid for ducks is methionine and found that canola meal represented a well-balanced amino acid profile for these birds. The amino acid digestibility of canola meal in ducks is shown in Table 2. Canola meal and soybean meal have similar amino acid digestibility in ducks (Kluth and Rodehutscord, 2006). In addition, the higher available phosphorus as compared to soybean meal is a desirable attribute.
Bernadet et al. (2009) studied the effects of rapeseed meal on the growth of mule ducks and noted that inclusion level would be limiting due to glucosinolates concentrations, which were not measured in their study. They did, however, determine that concentrations of 7% rapeseed meal in the starter period, and 21% in the grow finish period allowed for excellent growth. This suggests that at least these amounts of canola meal can be included in diets for ducks.
Zhu et al. (2019) provided starter ducks with “canola quality” rapeseed meal at levels of 0, 5, 10, 15 or 20% of the diet. The meal contained 25 umol/g of glucosinolates, a level that is approximately 5 times that of canola meal. There were no differences in growth rate for the feeding period (7 to 21 days). Feed/gain improved linearly with inclusion of the rapeseed meal, suggesting that starter feeds could contain more than the 7% proposed by Bernadet et al. (2009).
Fewer research programs are available for geese compared to other poultry species. Interestingly, geese have a greater digestive capability than other types of poultry, and appear to digest canola meal efficiently (Jamroz, et al., 1992). A dose titration study comparing graded levels of rapeseed meal to soybean meal was found (Fu et al., 2021). Isonitrogenous diets containing 0, 4, 8, 12 and 16% rapeseed meal were provided to growing geese from 35 to 70 days of age. There were no differences due to diet for growth, feed intake or feed efficiency. Dressing percentage and carcass component yields were likewise unaffected by diet. These results would suggest that 16% canola meal could be fed in the grow-finish period for geese.
Quail are raised for eggs as well as meat. Saki et al. (2017b) evaluated canola meal for quail hens at 10, 20 or 30% of the diet from 46 to 56 weeks of age. Production declined at the 20 and 30% level of inclusion, but there were no differences in performance at the 10% inclusion rate. The authors noted that this would allow 1/3 of the soybean meal to be replaced by canola meal. In an earlier study (Sarıçiçek et al., 2005), researchers replaced 0, 25 or 50% of the soybean meal in the diet for quail hens (0, 9.25 or 18.5% of the total diet as canola meal). In this 126 day-long study, there were no differences in hen body weight change, feed efficiency, % lay or egg mass.
A growth study was described by Minisi and Mlambo (2018). In the study, 6-week old quail were given isonitrogenous diets containing 0, 2.5, 5.0, 12.5 and 17.5% canola meal, replacing soybean meal on a protein basis. There were no differences in weight gain, but feed intake was lowest for the diet containing 17.5% canola meal. Sarıcicek et al. (2005) also compared canola meal to soybean meal in a quail growth study (Table 4). Again, 0, 25 or 50% of the protein from soybean meal was replaced with protein from canola meal, resulting in diets with 0, 12.15 and 24.3% total canola meal. In addition, multi-carbohydrase and phytase enzymes were tested for their ability to improve digestibility. Growth rates with the 50% canola meal were lower than the control when no enzymes were added to the diet. Dressing percentages and carcass characteristics were not different due to the diets. Combined these two trials suggest that 15% canola meal can safely be given to growing quail.
Table 6. Growth of quail in a study where canola meal partially relaced soybean meal1.
| wdt_ID | DIET | Soybean meal | Low canola meal | High canola meal |
|---|---|---|---|---|
| 1 | Replacement of soy protein, % | 0 | 25.00 | 50.00 |
| 2 | Canola meal in diet, % | 0 | 12.15 | 24.30 |
| 3 | No enzymes added | |||
| 4 | Weight gain, g | 150 | 140.00 | 132.00 |
| 5 | Feed intake, g | 761 | 751.00 | 740.00 |
| 6 | Feed/gain | 5.06 | 5.22 | 5.59 |
| 7 | Multi-carbohydrase enzymes added | |||
| 8 | Weight gain, g | 143 | 142.00 | 147.00 |
| 9 | Feed intake, g | 738 | 753.00 | 755.00 |
| 10 | Feed/gain | 5.16 | 5.13 | 5.16 |
1 Sancicek et al, 2015.
One novel study showed that ostriches can be grown to market weights using canola meal (Brand et al., 2020). Ostriches from 75 through 337 days of age were given diets with varying percentages of canola meal, with the canola meal replacing soybean meal and wheat grain on an amino acid basis (Table 7).
Table 7. Growth of ostriches in a study where canola meal was provided at varying levels1.
| wdt_ID | Feeding Program | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|---|
| 1 | Starter diet (76-146 days) | |||||
| 2 | Canola, % | 0.00 | 7.80 | 15.60 | 23.40 | 31.30 |
| 3 | Soybean meal, % | 17.90 | 13.40 | 9.00 | 4.50 | 0.00 |
| 4 | Grower diet (147-230 days) | |||||
| 5 | Canola, % | 0.00 | 5.00 | 10.00 | 15.00 | 20.00 |
| 6 | Soybean meal, % | 13.50 | 10.00 | 6.70 | 3.70 | 0.00 |
| 7 | Finisher diet (231- 377 days) | |||||
| 8 | Canola, % | 0.00 | 5.00 | 10.00 | 15.00 | 20.00 |
| 9 | Soybean meal, % | 10.40 | 7.90 | 5.20 | 2.60 | 0.00 |
1 Brand et al., 2020.
There were no differences in intake between the diets, and there were no differences in average daily gain from the start to the completion of the trial. Average daily gains during the starter phase increased with the 15.6 and 23.4% canola meal inclusion levels, but the advantage was not maintained for the duration of the trial. There were no differences in carcass weights or dressing percentages that could be associated with the trial.
Much of the canola used for poultry is solvent extracted, but there has been growing interest in the use of expeller canola meal due to its greater energy content. One item that hinders use is oil content, which can vary with the source of the meal (Woyengo et al., 2010), making it important to know the oil content for feed formulation.
Several studies support the use of expeller canola meal for broilers. An expeller pressed yellow variety of canola was evaluated in a Dalhousie study (Bryan et al., 2019a) with and without inclusion of fiber digesting enzymes. The canola meal was substituted for soybean meal and corn with an inclusion level was 30%. Additionally, expeller meals with two levels of residual oil (10 Vs 14%) were evaluated. The energy value of the meal increased with residual oil and was further increased with the inclusion of carbohydrase enzymes.
Inglis et al. (2021) provided boilers with diets containing 20% expeller meal in the starter, grower and finisher rations. Expeller canola meal and canola oil were incorporated in the diet in exchange for soybean meal and corn to provide the birds with isoenergetic and isonitrogenous diets. The researchers found no differences in average daily gain or feed conversion for the duration of the 35- day feeding trial.
A feeding trial was conducted by Oryschak et al. (2020) to evaluate the effects of expeller canola meal compared to solvent extracted canola meal and soybean meal. Two varieties of meal were also included in the 36-week evaluation with a 20% level of inclusion. There were no differences in percentage lay or hen body weights with any of the diets. Egg to feed ratio decreased slightly with the expeller meal. Diets with canola meal increased the percentage of mono-unsaturated fatty acids in the eggs. These results were similar to findings of Savary et al. (2017; 2019).
Spent hens can play a role in food security and are often used in commercial soups and specialty dishes requiring chicken with more texture. An exploratory study was conducted by Semwogerere et al. (2019) to compare nutritional characteristics of meat obtained from spent hens from a flock receiving a soybean meal diet (40 weeks of lay), and from a flock that had been given a diet with 20% expeller canola meal (48 weeks of lay). No differences in sensory attributes were found. The hens that had received the canola meal diet had less saturated fat (34.0% as compared to 38.7% of total fatty acids for the birds given the soybean meal diet) and a greater omega-3 fatty acid (5.1 % for the canola-fed birds as compared to 3.4% for the controls).
Canola seed is rich in oil and can be used as an energy source. Toghyani et al. (2017) analyzed six samples of seed, representing the range in composition. AMEn for growing broilers ranged from 4,501 to 4,791 and averaged 4,554 kcal/kg (dry matter basis). The variation could largely be explained by the variability in oil content, which ranged from 40.8 to 47.9% of the seed. This recently determined energy value for the seed was similar to the previously determined value (Barekatain et al., 2015) of 4,691 kcal/kg of dry matter.
Canola oil is routinely fed as an energy source to broiler chickens. In addition to its energy value, it is an excellent source of unsaturated fatty acids. Kanakri et al. (2018) fed broiler chickens diets containing approximately 3% added fat from beef tallow, flaxseed oil, corn oil, canola oil, macadamia oil or coconut oil. While there were no differences in growth performance between the different types of fat provided, the tissue fatty acid compositions of the birds reflected the varying fat sources provided. Muscle tissues from birds given canola oil had the lowest concentrations of saturated fatty acids and were second only to birds fed flax oil in omega-3 fatty acid content of muscle.
The ratio of linoleic acid (omega-6) to linolenic acid (omega-3) is approximately 2:1, as compared to 7:1 for soybean oil and 50:1 for corn oil. This is of importance because a common desaturase enzyme is used to elongate both fatty acids. Birds can elongate linolenic acid to docosahexaenoic acid (DHA). Excess linoleic acid limits the conversion (Cachaldora et al., 2008).
With the hens’ability to synthesize DHA from linolenic acid, eggs commonly provide an important and economical dietary supply of long chain omega-3 fatty acids. The fatty acid profile of the basal diet is the key to the success of producing DHA enriched eggs when the diets are supplemented with linolenic acid from sources such as flax oil or chia oil. Canola-based diets have been shown to be superior to diets where major ingredients contribute competing linoleic acid (Gonzalez-Esquerra and Leeson, 2001; Goldberg et al., 2016). In addition, Rowghani et al. (2007) showed that adding between 3 to 5% canola oil to corn-soybean meal diets resulted in over 8 times greater concentrations of DHA in eggs than diets without oil addition.
As the use of chemical growth promoting agents continues to diminish in the poultry industry, more and more information comes to light concerning the role of ingredients and specific nutrients upon health maintenance. Canola meal has been highlighted in several feeding trials as an ingredient that may contribute to gut health and digestion.
The fiber in canola meal is partially digested in the ceca of birds, resulting in the production of volatile fatty acids (VFA). These fatty acids, and in particular butyrate, serve to inhibit pathogenic bacteria (Elnesr et al., 2020) and supply required nutrients for colonocytes in the ceca and large intestine. Kozlowski et al. (2018) found that turkeys given diets with 20% Canadian canola meal as a replacement for soybean meal experienced a shift in the proportion of butyrate as a percentage of total VFA. The amount of total VFA in the cecal contents was enhanced when multi-carbohydrase enzymes were also added to the diet. Inglis et al. (2021) reported greater fermentation in growing broilers when diets contained canola meal. These researchers also reported differences in the bacterial profile of the cecal contents.
Older studies suggested that canola meal might alter the integrity of the gut lining in poultry. Gopinger et al. (2014) provided male broiler chickens with starter and grow-finisher diets that provided 0, 10, 20, 30 and 40% canola meal. They found no loss in integrity of the mucosal lining.
Researchers at the University of Georgia recently evaluated the effects of expeller rapeseed meal, solvent extracted canola meal and a metabolite of glucosinolate degradation (allyl isothiocyanate, AITC) in two challenge studies with Eimeria maxima and Salmonella typhimurium. The same diets were used in both studies: soybean meal control, 10% expeller rapeseed meal, 30% expeller rapeseed meal, 20% solvent extracted canola meal, 500 ppm AITC and 1000 ppm AITC. The Eimeria (coccidiosis) study (Yadav et al., 2022a) revealed that there were differences in growth rates that were not related to the Eimeria challenge that was imposed. Therefore, this test was not a meaningful measure of the outcome of the two trials. However, the intestinal permeability increased to a greater extent with the soybean meal and rapeseed meal than with the canola meal diet. Furthermore, the protective ability of the canola meal was numerically improved when the diet contained 40%, as compared to 20% canola meal. The AITC was also highly effective at reducing intestinal permeability. In the second study (Yadav et al., 2022) broilers were challenged with Salmonella at hatch. Gut permeability was not elevated by exposure to Salmonella. Intestinal villus height was greatest with the 30% rapeseed meal diet, with other treatments being the same as the control. Mortality was highest with the soybean meal challenge treatment. Therefore, canola meal may provide advantages when suffering from Eimeria infections and may provide an advantage over soybean meal through lower mortality when birds are exposed to Salmonella.
Adedokun, S.A., Adeola, O., Parsons, C.M., Lilburn, M.S. and Applegate, T.J., 2008. Standardized ileal amino acid digestibility of plant feedstuffs in broiler chickens and turkey poults using a nitrogen-free or casein diet. Poultry Science, 87(12), pp.2535-2548.
Adewole, D.I., Rogiewicz, A., Dyck, B. and Slominski, B.A., 2017. Effects of canola meal source on the standardized ileal digestible amino acids and apparent metabolizable energy contents for broiler chickens. Poultry Science, 96(12), pp.4298-4306.
Agyekum, A.K. and Woyengo, T.A., 2022. Nutritive value of expeller/cold-pressed canola meal and pre-pressed solvent-extracted carinata meal for broiler chicken. Poultry Science, 101(1), p.101528.
Ahmadi, A.S., Shivazad, M., Zagharim M and Shahneh, A.Z., 2007. The effect of different levels of rapeseed meal (with or without enzyme) on the broiler breeder flocks’ performance. Proceedings of the 2nd Animal Science Congress, Tehran, Iran. pp. 576-579.
Ariyibi, S., 2019. High inclusion levels of canola meal in broiler chicken nutrition (Master’s thesis, University of Manitoba).
Barekatain, M.R., Wu, S.B., Toghyani, M. and Swick, R.A., 2015. Effects of grinding and pelleting condition on efficiency of full-fat canola seed for replacing supplemental oil in broiler chicken diets. Animal Feed Science and Technology, 207, pp.140-149.
Barua, M., Abdollahi, M.R., Zaefarian, F., Wester, T.J., Girish, C.K. and Ravindran, V., 2020. Standardized ileal amino acid digestibility of protein sources for broiler chickens is influenced by the feed form. Poultry Science, 99(12), pp.6925-6934.
Berger, Q., Guettier, E., Urvoix, S., Bernard, J., Ganier, P., Chahnamian, M., Le Bihan-Duval, E. and Mignon-Grasteau, S., 2021. The kinetics of growth, feed intake, and feed efficiency reveal a good capacity of adaptation of slow and rapid growing broilers to alternative diets. Poultry Science, 100(4), p.101010.
Bernadet, M.D., Peillod, C., Lessire, M. and Guy, G., 2009. Incorporation of Rapeseed meal in mule duck diets. In Proceedings of IV World Waterfowl Conference, Thrissur, India (Vol. 161).
Bhaskare, G.V., 2020. Determination of metabolisable energy and digestible amino acids of canola meals and canola seed for broilers: a thesis presented in partial fulfilment of the requirements for the degree of Master of Science in Animal Science at Massey University, Manawatu, New Zealand (Doctoral dissertation, Massey University).
Brand, T. S., J. van der Merwe, and L. C. Hoffman. “Effect of including canola meal in diets of slaughter ostriches (Struthio camelus var. domesticus).” South African Journal of Animal Science 50, no. 4 (2020): 537-551.
Bryan, D.D.S.L., Abbott, D.A., Van Kessel, A.G. and Classen, H.L., 2019. In vivo digestion characteristics of protein sources fed to broilers. Poultry Science, 98(8), pp.3313-3325.
Bryan, D.D., MacIsaac, J.L., McLean, N.L., Rathgeber, B.M. and Anderson, D.M., 2019a. Nutritive Value of Expeller-Pressed Yellow Canola Meal for Broiler Chickens Following Enzyme Supplementation. Journal of Applied Poultry Research, 28(4), pp.1156-1167.
Cachaldora, P., Garcia-Rebollar, P., Alvarez, C., De Blas, J.C. and Mendez, J., 2008. Effect of type and level of basal fat and level of fish oil supplementation on yolk fat composition and n-3 fatty acids deposition efficiency in laying hens. Animal Feed Science and Technology, 141(1-2), pp.104-114.
Chen, X., Parr, C., Utterback, P. and Parsons, C.M., 2015. Nutritional evaluation of canola meals produced from new varieties of canola seeds for poultry. Poultry Science, 94(5), pp.984-991.
Cortes Cuevas, A., Cedillo Monrroy, M.S., Gómez Verduzco, G., Balderas Gonzalez, A. and Avila González, E., 2016. Performance and egg quality in semi-free range hens fed diets with different levels of canola meal. Revista Mexicana de Ciencias Pecuarias, 7(2), pp.173-184.
Czerwiski, J., Smulikowska, S., Mieczkowska, A., Konieczka, P., Piotrowska, A. and Bartkowiak-Broda, I., 2012. The nutritive value and phosphorus availability of yellow-and dark-seeded rapeseed cakes and the effects of phytase supplementation in broilers. Journal of Animal and Feed Sciences, 21(4), pp.677-695.
David, L.S., Abdollahi, M.R., Bedford, M.R. and Ravindran, V., 2021. True ileal calcium digestibility in soybean meal and canola meal, and true ileal phosphorous digestibility in maize-soybean meal and maize-canola meal diets, without and with microbial phytase, for broiler growers and finishers. British Poultry Science, 62(2), pp.293-303.
Elnesr, S.S., Alagawany, M., Elwan, H.A., Fathi, M.A. and Farag, M.R., 2020. Effect of sodium butyrate on intestinal health of poultry–a review. Annals of Animal Science, 20(1), pp.29-41.
Fu, Z., Su, G., Yang, H., Sun, Q., Zhong, T. and Wang, Z., 2021. Effects of dietary rapeseed meal on growth performance, carcass traits, serum parameters, and intestinal development of geese. Animals, 11(6), p.1488.
Gallardo, C., Dadalt, J.C., Kiarie, E. and Trindade Neto, M.A., 2017. Effects of multi-carbohydrase and phytase on standardized ileal digestibility of amino acids and apparent metabolizable energy in canola meal fed to broiler chicks. Poultry Science, 96(9), pp.3305-3313.
Goldberg, E.M., Ryland, D., Aliani, M. and House, J.D., 2016. Interactions between canola meal and flaxseed oil in the diets of White Lohmann hens on fatty acid profile and sensory characteristics of table eggs. Poultry Science, 95(8), pp.1805-1812.
Gonzalez-Esquerra, R. and Leeson, S., 2001. Alternatives for enrichment of eggs and chicken meat with omega-3 fatty acids. Canadian Jjournal of Animal Science, 81(3), pp.295-305.
Gopinger, E., Xavier, E.G., Elias, M.C., Catalan, A.A.S., Castro, M.L.S., Nunes, A.P. and Roll, V.F.B., 2014. The effect of different dietary levels of canola meal on growth performance, nutrient digestibility, and gut morphology of broiler chickens. Poultry Science, 93(5), pp.1130-1136.
Gorski, M.F., 2015. Nutritional evaluation of canola meal produced from a new variety of canola seeds in broiler chickens and laying hens. Ph.D. Thesis, University of Illinois.
Gorski, M., Foran, C., Utterback, P. and Parsons, C.M., 2017. Nutritional evaluation of conventional and increased-protein, reduced-fiber canola meal fed to broiler chickens. Poultry Science, 96(7), pp.2159-2167.
Goudarzi, S.M., Saki, A., Mohammadi, N. and Zamani, P., 2017. The effect of substitution of soybean with canola meal in laying hen diets formulated on the basis of total or digestible amino acids on performance and blood parameters. Journal of Livestock Technology, 5, pp.1-8.
Inglis, G.D., Wright, B.D., Sheppard, S.A., Abbott, D.W., Oryschak, M.A. and Montina, T., 2021. Expeller-Pressed Canola (Brassica napus) Meal Modulates the Structure and Function of the Cecal Microbiota, and Alters the Metabolome of the Pancreas, Liver, and Breast Muscle of Broiler Chickens. Animals, 11(2), p.577.
Jamroz, D., Wiliczkiewicz, A. and Skorupinska, J., 1992. The effect of diets containing different levels of structural substances on morphological changes in the intestinal walls and the digestibility of the crude fiber fractions in geese (Part III). Journal of Animal Feed Science, 1(3), pp.37-50.
Jayaraman, B., MacIsaac, J. and Anderson, D., 2016. Effects of derived meals from juncea (Brassica juncea), yellow and black seeded canola (Brassica napus) and multicarbohydrase enzymes supplementation on apparent metabolizable energy in broiler chickens. Animal Nutrition, 2(3), pp.154-159.
Jia, W., Mikulski, D., Rogiewicz, A., Zdunczyk, Z., Jankowski, J. and Slominski, B.A., 2012. Low-fiber canola. Part 2. Nutritive value of the meal. Journal of agricultural and food chemistry, 60(50), pp.12231-12237.
Kaminska, B.Z., 2003. Substitution of soyabean meal with” 00″ rapeseed meal or its high-protein fraction in the nutrition of hens laying brown-shelled eggs. Animal and Feed Sciences, 12(1), pp.111-120.
Kanakri, K., Carragher, J., Hughes, R., Muhlhausler, B. and Gibson, R., 2018. The effect of different dietary fats on the fatty acid composition of several tissues in broiler chickens. European. Journal of Lipid Science and Technology, 120(1), p.1700237.
Khajali, F. and Slominski, B.A., 2012. Factors that affect the nutritive value of canola meal for poultry. Poultry Science, 91(10), pp.2564-2575.
Khalil, M.M., Abdollahi, M.R., Zaefarian, F. and Ravindran, V., 2021. Influence of feed form on the apparent metabolisable energy of feed ingredients for broiler chickens. Animal Feed Science and Technology, 271, p.114754.
Kiiskinen, T., 1989. Effect of long-term use of rapeseed meal on egg production. Annals Agriculture Fenniae. 28, pp.385-396.
Kim, E.J., Utterback, P.L. and Parsons, C.M., 2012. Comparison of amino acid digestibility coefficients for soybean meal, canola meal, fish meal, and meat and bone meal among 3 different bioassays. Poultry Science, 91(6), pp.1350- 1355.
Kluth, H. and Rodehutscord, M., 2006. Comparison of amino acid digestibility in broiler chickens, turkeys, and Pekin ducks. Poultry Science, 85(11), pp.1953-1960.
Kong, C. and Adeola, O., 2013. Comparative amino acid digestibility for broiler chickens and White Pekin ducks. Poultry Science, 92(9), pp.2367- 2374.
Kong, C. and Adeola, O., 2016. Determination of ileal digestible and apparent metabolizable energy contents of expeller-extracted and solvent extracted canola meals for broiler chickens by the regression method. Springer Plus, 5(1), p.693.
Kozłowski, K., Helmbrecht, A., Lemme, A., Jankowski, J. and Jeroch, H., 2011. Standardized ileal digestibility of amino acids from high-protein feedstuffs for growing turkeys–a preliminary study. Archiv Geflügelk, 75, pp.185-190.
Kozlowski, K., Mikulski, D., Rogiewicz, A., Zdunczyk, Z., Rad-Spice, M., Jeroch, H., Jankowski, J. and Slominski, B.A., 2018. Yellow-seeded B. napus and B. juncea canola. Part 2. Nutritive value of the meal for turkeys. Animal Feed Science and Technology, 240, pp.102-116.
Meng, X. and Slominski, B.A., 2005. Nutritive values of corn, soybean meal, canola meal, and peas for broiler chickens as affected by a multicarbohydrase preparation of cell wall degrading enzymes. Poultry Science, 84(8), pp.1242-1251.
Meng, X., Slominski, B.A., Nyachoti, C.M., Campbell, L.D. and Guenter, W., 2005. Degradation of cell wall polysaccharides by combinations of carbohydrase enzymes and their effect on nutrient utilization and broiler chicken performance. Poultry Science, 84(1), pp.37-47.
Mandal, A.B., Elangovan, A.V., Tyagi, P.K., Tyagi, P.K., Johri, A.K. and Kaur, S., 2005. Effect of enzyme supplementation on the metabolisable energy content of solvent-extracted rapeseed and sunflower seed meals for chicken, guinea fowl and quail. British Poultry Science, 46(1), pp.75-79.
Mnisi, C.M. and Mlambo, V., 2018. Growth performance, haematology, serum biochemistry and meat quality characteristics of Japanese quail (Coturnix coturnix japonica) fed canola meal-based diets. Animal Nutrition, 4(1), pp.37-43.
Moss, A.F., Chrystal, P.V., Dersjant-Li, Y., Selle, P.H. and Liu, S.Y., 2018. Responses in digestibilities of macro-minerals, trace minerals and amino acids generated by exogenous phytase and xylanase in canola meal diets offered to broiler chickens. Animal Feed Science and Technology, 240, pp.22-30.
Munoz, J.A., Hanna, C.D., Utterback, P.L. and Parsons, C.M., 2018. Phosphorus retention in corn, spray dried plasma protein, soybean meal, meat and bone meal, and canola meal using a precision-fed rooster assay. Poultry Science, 97(12), pp.4324-4329.
Mutucumarana, R.K., Ravindran, V., Ravindran, G. and Cowieson, A.J., 2015. Measurement of true ileal phosphorus digestibility in maize and soybean meal for broiler chickens: Comparison of two methodologies. Animal Feed Science and Technology, 206, pp.76-86.
Naseem, M.Z., Khan, S.H. and Yousaf, M., 2006. Effect of feeding various levels of canola meal on the performance of broiler chicks. Journal of Animal and Plant Sciences, 16, pp.3-4.
Nassar, A.R., Goeger, M.P. and Arscott, G.H., 1985. Effect of canola meal in laying hen diets. Nutrition reports international 31, pp.1349-1355.
Newkirk, R., 2011. Meal nutrient composition. In Canola (pp. 229-244). AOCS Press.
Newkirk, R.W. and Classen, H.L., 2002. The effects of toasting canola meal on body weight, feed conversion efficiency, and mortality in broiler chickens. Poultry Science, 81(6), pp.815-825.
Novak, C., Yakout, H. and Scheideler, S., 2004. The combined effects of dietary lysine and total sulfur amino acid level on egg production parameters and egg components in Dekalb Delta laying hens. Poultry Science, 83(6), pp.977-984.
Noll, S.L., Sernyk, J.L. and Hickling, D. 2017. University of Minnesota Research Update: Canola Meal Use in Market Turkey Diets. Proceedings of the Minnesota Nutrition Conference.
Niu, Y., Rogiewicz, A., Shi, L., Patterson, R. and Slominski, B.A., 2022. The effect of multi-carbohydrase preparations on non-starch polysaccharides degradation and growth performance of broiler chickens fed diets containing high inclusion level of canola meal. Animal Feed Science and Technology, 293, p.115450.
Oryschak, M.A. and Beltranena, E., 2013. Further Processing for Better Utilization of Co-products in Monogastrics. In Proceedings of the 34th Western Nutrition Conference pp. 48-61.
Oryschak, M.A., Smit, M.N. and Beltranena, E., 2020. Brassica napus and Brassica juncea extruded-expelled cake and solvent-extracted meal as feedstuffs for laying hens: Lay performance, egg quality, and nutrient digestibility. Poultry Science, 99(1), pp.350-363.
Osho, S.O., Babatunde, O.O. and Adeola, O., 2019. Additivity of apparent and standardized ileal digestibility of amino acids in wheat, canola meal, and sorghum distillers dried grains with solubles in mixed diets fed to broiler chickens. Poultry Science, 98(3), pp.1333-1340.
Park, C.S., Ragland, D., Helmbrecht, A., Htoo, J.K. and Adeola, O., 2019. Digestibility of amino acid in full-fat canola seeds, canola meal, and canola expellers fed to broiler chickens and pigs. Journal of Animal Science, 97(2), pp.803-812.
Ramesh, K.R., Devegowda, G. and Khosravinia, H., 2006. Effects of enzyme addition to broiler diets containing varying levels of double zero rapeseed meal. Asian-Australian Journal of Animal Sciences, 19(9), p.1354
Rad-Spice, M., Rogiewicz, A., Jankowski, J. and Slominski, B.A., 2018. Yellow-seeded B. napus and B. juncea canola. Part 1. Nutritive value of the meal for broiler chickens. Animal Feed Science and Technology, 240, pp.66-77.
Rogiewicz, A., Dyck, B. and Slominski, B.A., 2015. High inclusion levels of canola meal in laying hen diets. In 14th International Rapeseed Congress, Saskatoon, Canada (p. 139).
Ross, M.L., Bryan, D.D., Abbott, D.A. and Classen, H.L., 2019. Effect of protein sources on performance characteristics of turkeys in the first three weeks of life. Animal Nutrition, 5(4), pp.396-406.
Rowghani, E., Arab, M., Nazifi, S. and Bakhtiari, Z., 2007. Effect of canola oil on cholesterol and fatty acid composition of egg-yolk of laying hens. International journal of Poultry Science, 6(2), pp.111-114.
Sarıcicek, B.Z., Kılıc, U. and Garipolu, A.V., 2005. Replacing soybean meal (SBM) by canola meal (CM): The effects of multi-enzyme and phytase supplementation on the performance of growing and laying quails. Asianaustralasian journal of animal sciences, 18(10), pp.1457-1463
Savary, R.K., MacIsaac, J.L., Rathgeber, B.M., McLean, N.L. and Anderson, D.M., 2017. Evaluating Brassica napus and Brassica juncea meals with supplemental enzymes for use in laying hen diets: production performance and egg quality factors. Canadian Journal of Animal Science, 97(3), pp.476-487.
Savary, R.K., MacIsaac, J.L., Rathgeber, B.M., McLean, N.L. and Anderson, D.M., 2019. Evaluating Brassica napus and Brassica juncea meals with supplemental enzymes for use in brown-egg laying hen diets: production performance and egg quality factors. Canadian Journal of Animal Science, 99(4), pp.820-832.
Semwogerere, F., Neethling, J., Muchenje, V. and Hoffman, L.C., 2019. Meat quality, fatty acid profile, and sensory attributes of spent laying hens fed expeller press canola meal or a conventional diet. Poultry Science, 98(9), pp.3557-3570.
Sessingnong, T., Omotosho, O, Rogiewicz, A, Niu, Y., Ferreira, J., Patterson, R, and Slominski, B.A. 2022. Research on Evaluation of Expeller/Cold-pressed Canola (EPC) as a Valuable Feed Ingredient for Poultry. Proceedings, Animal Nutrition Conference of Canada.
Summers, J.D. and Bedford, M., 1994. Canola meal and diet acid-base balance for broilers. Canadian Journal of Animal Science, 74(2), pp.335-339.
Toghyani, M., Rodgers, N., Barekatain, M.R., Iji, P.A. and Swick, R.A., 2014. Apparent metabolizable energy value of expeller-extracted canola meal subjected to different processing conditions for growing broiler chickens. Poultry Sscience, 93(9), pp.2227-2236.
Veluri, S. and Olukosi, O.A., 2020. Metabolizable energy of soybean meal and canola meal as influenced by the reference diet used and assay method. Animals, 10(11), p.2132.
Waibel, P.E., Noll, S.L., Hoffbeck, S., Vickers, Z.M. and Salmon, R.E., 1992. Canola meal in diets for market turkeys. Poultry Science, 71(6), pp.1059- 1066.
Wickramasuriya, S.S., Yi, Y.J., Yoo, J., Kang, N.K. and Heo, J.M., 2015. A review of canola meal as an alternative feed ingredient for ducks. Journal of Animal Science and Technology, 57(1), p.29.
Wise, T.L. and Adeola, O., 2022. Validation of a 3-point model for the determination of energy values using the regression method in broiler chickens. Poultry Science, p.102336.
Woyengo, T.A., Kiarie, E. and Nyachoti, C.M., 2010. Metabolizable energy and standardized ileal digestible amino acid contents of expeller-extracted canola meal fed to broiler chicks. Poultry Science, 89(6), pp.1182-1189.
Wu, S.B., Choct, M. and Pesti, G., 2020. Historical flaws in bioassays used to generate metabolizable energy values for poultry feed formulation: a critical review. Poultry Science, 99(1), pp.385-406.
Yadav, S., Teng, P.Y., Choi, J., Singh, A.K., Vaddu, S., Thippareddi, H. and Kim, W.K., 2022. Influence of rapeseed, canola meal and glucosinolate metabolite (AITC) as potential antimicrobials: effects on growth performance, and gut health in Salmonella Typhimurium challenged broiler chickens. Poultry Science, 101(1), p.101551.
Yadav, S., Teng, P.Y., Singh, A.K., Choi, J. and Kim, W.K., 2022a. Influence of Brassica spp. rapeseed and canola meal, and supplementation of bioactive compound (AITC) on growth performance, intestinal-permeability, oocyst shedding, lesion score, histomorphology, and gene expression of broilers challenged with E. maxima. Poultry Science, 101(2), p.101583.
Yoshida, Y., Nishimura, S., Tabata, S. and Kawabata, F., 2022. Chicken taste receptors and perception: recent advances in our understanding of poultry nutrient-sensing systems. World’s Poultry Science Journal, 78(1), pp.5-20.
Zhang, K.X., Zhang, K.Y., Applegate, T.J., Bai, S.P., Ding, X.M., Wang, J.P., Peng, H.W., Xuan, Y., Su, Z.W. and Zeng, Q.F., 2020. Evaluation of the standardized ileal digestibility of amino acids of rapeseed meals varying in protein solubility for Pekin ducks. Poultry Science, 99(2), pp.1001-1009.
Zhong, R. and Adeola, O., 2019. Energy values of solvent-extracted canola meal and expeller-derived canola meal for broiler chickens and growing pigs determined using the regression method. Journal of Animal Science, 97(8), pp.3415-3425.
Zhu, Y.W., Yang, W.C., Liu, W., Yin, X.H., Luo, X.B., Zhang, S.A., Wang, W.C. and Yang, L., 2019. Effects of dietary rapeseed meal inclusion levels on growth performance, organ weight, and serum biochemical parameters in Cherry Valley ducks. Poultry Science, 98(12), pp.6888-6896.