

Canola meal makes ration balancing simple. The superior amino acid profile, along with a high percentage of rumen bypass protein has repeatedly been shown to support milk production with lower amounts of protein, resulting in lower levels of milk urea nitrogen (MUN) and less waste.
Canola meal is a well-researched feed ingredient for dairy cows that consistently provides value for producers, as shown in recent academic studies. Field trials conducted in Wisconsin have shown that canola meal can be an alternative for animal proteins and can also be used to replace the high bypass protein soybean meal products that are often needed in rations balanced for amino acids.
When picking protein sources for dairy rations, the stress has historically been on the crude-protein levels of protein sources. More protein is always better, right? Not necessarily. Cows need the right protein, in the right place. When these requirements are met, the cow uses protein in the most efficient way possible, meaning less protein ends up in the manure pile. Fortunately, canola meal provides the right protein, in the right place, at the right time.
Canola meal has more rumen undegraded protein, or bypass protein, than many other frequently used protein sources. When protein enters a cow’s rumen, rumen microbes break down, or degrade, a fraction of the available protein. The remaining protein bypasses the cow’s rumen and becomes available to satisfy the cow’s amino acid requirements. This means that while it may not always beat other protein sources in crude protein levels, a high portion of canola meal’s protein makes it through the cow’s rumen, putting the protein right where the cow needs it to produce milk. See how canola meal stacks up against soybean meal when it comes to bypass protein in the table below. Feeding canola meal means better feeding efficiency and higher milk production for dairy operations.
Pound for pound, canola meal provides as much digestible RUP as soybean meal, even with its lower crude protein levels. The RUP in canola meal provides more available methionine and histidine than soybean meal, and about 90% as much available lysine, as shown in the table below. Although high protein distiller’s grains supplies more digestible RUP than either soybean meal or canola meal, it fails to supply the amino acids most critical for milk production, and much more would be needed in the diet to supply the amino acids.
1Maxin, G., Ouellet, D.R. and Lapierre, H., 2013.
2Ross, D.A., Gutierrez-Botero, M. and Van Amburgh, M.E. 2013.
Another way canola meal provides value is through its amino acid profile. A high level of bypass protein combined with canola meal’s unique amino acid profile not only helps cows optimize milk production, but also supports the production of high-quality milk.
Canola meal’s amino acid profile helps explain the consistent milk yield response when canola meal is included in rations. In a summary of 24 research trials with canola meal included in dairy diets, the average milk production response was an additional 2.2 pounds of milk per day, per cow. With results like that, canola meal is a must-try for all dairy producers looking to increase profits and efficiencies in their operation. Canola’s amino acid pattern matches that of milk more closely than other vegetable protein sources (Table below) and therefore is utilized more efficiently.
Ingredient and rumen microbe amino acid composition as compared milk essential amino acid (EAA) composition (The first limiting amino acid is highlighted for each source)
| Milk¹ EAA | Rumen² Microbes | Corn DDGS | Soybean Meal % of Amount in Milk | Corn Gluten | Cottonseed Meal | Canola Meal | |
| Methionine | 2.50 | 88 | 79 | 53 | 95 | 56 | 84 |
| Lysine | 8.00 | 101 | 35 | 76 | 21 | 50 | 68 |
| Arginine | 3.38 | 140 | 127 | 216 | 93 | 342 | 191 |
| Threonine | 4.31 | 124 | 86 | 90 | 77 | 71 | 97 |
| Leucine | 9.56 | 82 | 121 | 78 | 169 | 57 | 63 |
| Isoleucine | 5.12 | 112 | 71 | 89 | 78 | 59 | 66 |
| Histidine | 2.73 | 69 | 97 | 96 | 74 | 100 | 132 |
| Phenylalanine | 4.94 | 108 | 99 | 102 | 125 | 107 | 81 |
| Tryprophan | 0.90 | 131 | 89 | 148 | 60 | 136 | 143 |
| Valine | 5.60 | 102 | 87 | 84 | 81 | 75 | 91 |
¹ Lee C., Giallongo, F., Hristov, A.N., Lapierre, H., Cassidy, T.W., Heyler, K.S., Varga, G.A. and Parys, C., 2015
² Sok, M., Ouellet, D.R., Firkins, J.L., Pellerin, D. and Lappiere, H., 2017
Another way we can look at this is through canola meal’s milk protein score. Feed ingredients are sometimes given milk protein scores as another way to measure their value. The milk protein score takes the amino acid profile of protein sources and compares it to the amino acid composition of milk itself. Canola meal has the highest milk protein score among vegetable-based protein sources, including soybean meal. A high milk protein score equals high-quality milk.
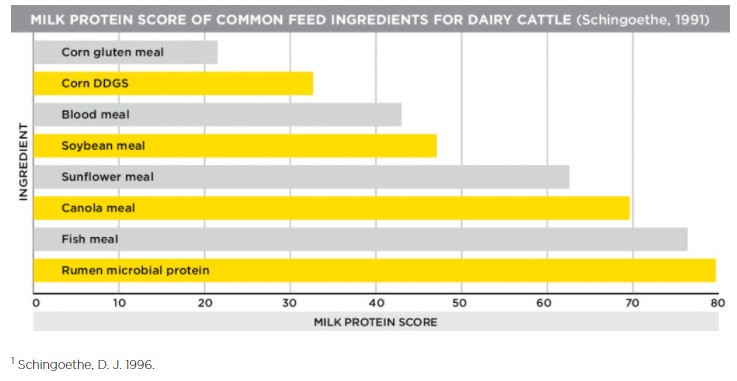
Canola meal’s bypass protein levels and great milk protein score combine to support high levels of components in milk. Milk components, or the fat and protein portions of milk, are important because they are indicative of high-quality milk, and could mean more milk check premiums for dairy producers.
As the table below illustrates, daily protein and fat yields increased when canola meal replaced either soybean meal or cottonseed meal in diets for high-producing dairy cows.
| Added Protein Source | Soybean Meal | Cottonseed Meal | Canola Meal |
| Measurement | |||
| Percent of diet dry matter | 12.1 | 14.1 | 16.5 |
| Dry matter intake, lbs/day | 54.3 | 54.5 | 54.9 |
| Milk yield, lbs/day | 88.2 | 89.3 | 90.6 |
| Protein yield, lbs/day | 2.71 | 2.60 | 2.80 |
| Fat yield, lbs/day | 2.69 | 2.60 | 2.84 |
*U.S. Dairy Forage Research Center, Madison, WI (Brito, et al., J. Dairy Sci., 90:1828-1841, 2007)
| 15% Crude Protein | 15% Crude Protein | 17% Crude Protein | 17% Crude Protein | |
| Soybean Meal | Canola Meal | Soybean Meal | Canola Meal | |
| Measurement | ||||
| Dry matter intake, lbs | 54.6 | 55.6 | 55.4 | 56.1 |
| Milk yield, lbs | 86.9 | 88.4 | 87.8 | 90.4 |
| Protein yield, lbs | 2.62 | 2.66 | 2.66 | 2.73 |
| Fat yield, lbs | 3.43 | 3.50 | 3.52 | 3.63 |
*U.S. Dairy Forage Research Center, Madison, WI (Broderick, et al., J. Dairy Sci., 98:5672-5687, 2015)
These results are consistent with newer research that now recognizes that the soluble fraction of protein meals is not all degraded in the rumen, and some of the soluble protein from both soybean meal and canola meal will bypass the rumen. For more information, you can check out recent research abstracts and visit the Dairy Blog.
Canola meal adds persistent milk yield throughout lactation – June 2023, Progressive Dairy
A 10-week trial shows new results for canola meal and lactation persistency.
Formulating dairy diets to reduce greenhouse gas – February 2022, Progressive Dairy
Currently available feed ingredients like monensin, fat and canola meal are effective ways to reduce methane emissions from livestock.
Commitment to accuracy: the 2019 canola meal dairy feed guide – February 1, 2020 Industry Voice
Canola meal is a preferred source of protein and amino acids in dairy diets. But what do we know about its nutrient profile? A seven-year study brings clarity.
Canola meal in dairy rations: – July 24, 2020 By Treena Hein
New research explains why use of this high-protein ingredient has increased in many countries. Canola meal is continuing to make inroads into dairy cow rations in many countries, including Canada, the USA, South Africa, Mexico, China and several other Asian countries. This high-protein ingredient is also fed to other livestock, including aquaculture species. In Australia for example, canola meal is now fed to fish, dairy cattle, poultry and to beef cattle in feedlots.
Canola Meal: Too Often Overlooked in Dairy Cattle Nutrition Programs – November 2, 2019 Les Berghorn and Essi Evans
It is not uncommon for published nutrient values for canola meal to be inconsistent with values required for current feed formulation models. Some of the common errors can result in overlooking a valuable commodity.
Dairy cows on canola meal diets may outperform those on soybean meal – May 29, 2019 by Aerin Einstein-Curtis
Dropping prices for protein meals are prompting a comparison of canola and soybean meal, but a feeding trial shows that canola meal may provide more support for high performing dairy cow production and body condition than soybean meal, says researcher.
Canola meal boosts milk performance in dairy cattle trials – November 20, 2018
The trial ration is competitive or even cheaper than the current diet.
New Dairy Trial Shows Value of Canola Meal – October 17, 2018
After participating in a recent field trial where canola meal was fed at high inclusion rates, Shiloh Dairy Manager Gordon Speirs said “Canola contributes to cost-efficient production. We no longer have to source and store expeller soybean meal”
6 reasons to consider canola meal – September 2018 by Essi Evans
When compared against soybean meal, canola meal not only holds its own but often produces measurable advantages in feed efficiency and milk production.
Best practices for feeding canola meal to dairy cows – July 5, 2018 No Particular Author
Research survey evaluates the effects of forage and starch on lactating dairy cow diets with canola meal as the main protein supplement.
The True Value of Feeding Canola Meal – 2018 Timothy Mutsvangwa
Canola meal (CM) is a highly palatable feed ingredient for dairy cows, and it can be included in dairy cow diets up to 20% of dietary dry matter. Cows fed CM as a protein source produce, on average, 1.4 kg/day more milk compared with cows fed other protein sources, and 0.7 kg/d more milk compared with cows fed soybean meal (SBM). Newer research shows that CM has a greater content of ruminally undegradable protein (RUP; bypass protein) than has been previously reported, and CM RUP is at least equal to, if not greater, than that of SBM. Compared to other protein sources like SBM, CM is an excellent source of essential amino acids like methionine and histidine. Although CM contains greater amounts of fibre compared with other protein sources, recent research shows that the energy value of CM is higher than previously thought because the fiber is more digestible and can provide more energy for milk production.
Canola Meal Research Shows an Increase in Milk Production and Feed Cost Savings – December, 2017
At the recent Penn State Dairy Nutrition Workshop, Kenneth Kalscheur, a prominent dairy nutrition researcher at the U.S. Dairy Forage Research Center in Madison, Wisconsin, provided findings on the subject of “Canola Meal vs. Soybean Meal in Dairy Cow Diets.”
Comparison of Feed Proteins for Dairy Cows takes Careful Thought – June 5, 2017 Glen Broderick, Essi Evans and Brittany Dyck
Nutritionists struggle to estimate the value of protein ingredients, and selecting the wrong metric can make a considerable difference to the bottom line.
Canola Meal in Cows’ diet beats the competition – March 23, 2017 By Barb Glen
Tim Mutsvangwa, a professor of ruminant nutrition at the University of Saskatchewan, said cows produce more milk when canola meal is in their diet and it also contains essential amino acids and higher amounts of fibre than other protein sources.
Canola meal edges soybean meal as dairy cow protein source – February, 2016 Glen Broderick
During the past few years, we conducted several feeding trials at the U.S. Dairy Forage Research Center in Madison, Wisconsin, comparing the yields of milk and milk components from lactating dairy cows supplemented with either canola meal or soybean meal.
Seven Ways to Ensure Efficient Use of Protein by Dairy Cows – Sept 11, 2015 Brittany Dyck, Carson Callum and Essi Evans
With a better understanding of RUP, ingredient selections can be made to reduce protein overages and reduce nitrogen lost to the environment
Avoid Too Much of a Good Thing: Protein – May 2015 Brittany Dyck and Essi Evans
Dairy producers and nutritionists understand the importance of providing adequate levels of nutrients in dairy cow rations. However, it is possible to provide too much of a good thing, which can lead to some undesirable consequences.
Meeting methionine requirements across livestock species with Canola Meal – January 5, 2015 Industry Voice
Canola meal is a good source of methionine, but feed formulators are often surprised to find the amino acid profile of canola meal is well suited to the needs of poultry and swine
Canola Meal and High-Forage Diets Go Hand and Hand – August 2014 Essi Evans
Higher grain prices in recent years have encouraged dairy producers to consider increasing the amount of forage and byproduct ingredients in rations for lactating dairy cows. Can canola meal be useful with higher forage diets?
Canola Meal and Distillers Grains: A Match Made in Heaven – October 31, 2013 Essi Evans
Distillers grains are often less costly per ton than canola meal. In spite of that fact, many nutritionists find that they must limit the level of inclusion in the diet or else sacrifice milk production. Trials at the University of Saskatchewan and University of California show that canola meal can shore up the value of distillers’ grains
Evaluating Dairy Ration Proteins: When Do They Pay? – Brittany Dyck and Essi Evans
To what extent does that protein ingredient result in production advantages, and do those production advantages outweigh the higher cost of an ingredient?
Review of research in canola meal for dairy cows including meta analyses
https://www.canolacouncil.org/canolamazing/wordpress/wp-content/uploads/2023/09/canola-meal-as-a-feed-ingredient-for-lactating-dairy-cows-a-review.pdf
Canola Meal as a Feed Ingredient for Lactating Dairy Cows.
Evans, E. and Wood, B., 2023. Journal of Advanced Dairy Research, 11, p.625.
Determining the optimum level of inclusion of canola meal in diets for dairy cows
https://www.proquest.com/openview/a253056954faf12231f1f1f5f75e7c96/1?pq-origsite=gscholar&cbl=18750&diss=y
Production and Physiological Effects of Including Soybean Meal or Canola Meal in Dairy Cow Diets from the Close-Up Dry Period to Mid-Lactation.
Kuehnl, J.M., 2022. The University of Wisconsin-Madison.
https://researchoutput.csu.edu.au/ws/portalfiles/portal/266789683/PhD_Final_Lynch.pdf
Effects of varying inclusion levels of canola meal for grass-fed cattle.
Lynch, E., 2022.
Understanding the nutritive contribution of canola meal for dairy cows
https://www.ajol.info/index.php/sajas/article/view/264686
Replacement of soybean with canola improves short-term milk yield and nitrogen-use efficiency in high-producing, early-lactation Holstein cows.
Mohammadi, F., Firouzabadi, M.S.S., Savari, M., Kachoie, M.A., Rayshan, A.R., Sivapriya, T. and Abdollahzadeh, F., 2023. South African Journal of Animal Science, 53(5), pp.649-657.
https://www.tandfonline.com/doi/abs/10.1080/10408398.2022.2033686
Research progress and future study on physicochemical, nutritional, and structural characteristics of canola and rapeseed feedstocks and co-products from bio-oil processing and nutrient modeling evaluation methods.
Oliveira, A.M.D. and Yu, P., 2023. Critical Reviews in Food Science and Nutrition, 63(23), pp.6484-6490.
https://www.proquest.com/openview/a253056954faf12231f1f1f5f75e7c96/1?pq-origsite=gscholar&cbl=18750&diss=y
Production and Physiological Effects of Including Soybean Meal or Canola Meal in Dairy Cow Diets from the Close-Up Dry Period to Mid-Lactation.
Kuehnl, J.M., 2022. The University of Wisconsin-Madison.
https://www.mdpi.com/2076-2615/13/13/2192
Impact of partially substituting canola meal with solvent-extracted distillers grain with solubles as a protein source on milk production in a commercial Holstein dairy herd.
Edwards, R.J., Ledgerwood, D.N., Ferreira, F.C. and Rossow, H.A., 2023. Animals, 13(13), p.2192.
https://www.canolacouncil.org/canolamazing/wordpress/wp-content/uploads/2023/09/canola-meal-as-a-feed-ingredient-for-lactating-dairy-cows-a-review.pdf
Canola Meal as a Feed Ingredient for Lactating Dairy Cows.
Evans, E. and Wood, B., 2023. Journal of Advanced Dairy Research, 11, p.625.
https://www.sciencedirect.com/science/article/pii/S0022030223004101
Production effects of extruded soybean meal replacing canola meal in the diet of lactating dairy cows.
Cueva, S.F., Räisänen, S.E., Wasson, D.E., Lage, C.F.A., Silvestre, T., Kniffen, D.M., Fabin, R.A. and Hristov, A.N., 2023. Journal of dairy science, 106(9), pp.6198-6215.
https://www.sciencedirect.com/science/article/abs/pii/S0022030221006925
Diet supplementation with canola meal improves milk production, reduces enteric methane emissions, and shifts nitrogen excretion from urine to feces in dairy cows.
Benchaar, C., Hassanat, F., Beauchemin, K.A., Gislon, G. and Ouellet, D.R., 2021. Journal of Dairy Science.
https://www.mdpi.com/2076-2615/11/6/1636
Canola Meal versus Soybean Meal as Protein Supplements in the Diets of Lactating Dairy Cows Affects the Greenhouse Gas Intensity of Milk.
Holtshausen, L., Benchaar, C., Kröbel, R. and Beauchemin, K.A., 2021. Animals, 11(6), p.1636.
www.sciencedirect.com/science/article/abs/pii/S0022030221000138
Lactational performance, enteric gas emissions, and plasma amino acid profile of dairy cows fed diets with soybean or canola meals included on an equal protein basis.
Lage, C.F.A., Räisänen, S.E., Stefenoni, H., Melgar, A., Chen, X., Oh, J.O.O.N.P.Y.O., Fetter, M.E., Kniffen, D.M., Fabin, R.A. and Hristov, A.N., 2021.
https://www.sciencedirect.com/science/article/abs/pii/S002203022100271X
Canola meal or soybean meal as protein source and the effect of microencapsulated sodium butyrate supplementation in calf starter mixture. I. Performance, digestibility, and selected blood variables.
Burakowska, K., Penner, G.B., Flaga, J., Kowalski, Z.M. and Górka, P., 2021. Journal of dairy science, 104(6), pp.6646-6662.
https://www.sciencedirect.com/science/article/abs/pii/S0022030221005403
Effects of canola meal inclusion rate in starter mixtures for Holstein heifer calves on dry matter intake, average daily gain, ruminal fermentation, plasma metabolites, and total-tract digestibility.
Burakowska, K., Górka, P. and Penner, G.B., 2021. Journal of Dairy Science, 104(8) pp 8736-8745.
https://www.sciencedirect.com/science/article/pii/S00220302193107683
Effects of replacing soybean meal with canola meal for lactating dairy cows fed 3 different ratios of alfalfa to corn silage.
Paula, E.M., Broderick, G.A. and Faciola, A.P., 2020. Journal of Dairy Science, 103(2), pp.1463-1471.
www.sciencedirect.com/science/article/abs/pii/S0022030220305026
Effect of heat-treated canola meal and glycerol inclusion on performance and gastrointestinal development of Holstein calves.
Burakowska, K., Górka, P., Kent-Dennis, C., Kowalski, Z.M., Laarveld, B. and Penner, G.B., 2020. Journal of Dairy Science, 103(9), pp.7998-8019.
https://li01.tci-thaijo.org/index.php/anres/article/view/248673
Utilisation of canola meal as protein source in dairy cow diets: a review.
Heim, R. and Krebs, G., 2020. Agriculture and Natural Resources, 54(6), pp.623-632.
http://vetjournal.ankara.edu.tr/en/download/article-file/1032037
and canola meal as protein sources in starter diets for calves: current knowledge and directions of future studies.
GÓRKA, P. and Penner, G.B., 2020. Ankara Üniversitesi Veteriner Fakültesi Dergisi, 67(3), pp.313-321.
https://www.sciencedirect.com/science/article/pii/S0022030220301156
Production and nitrogen metabolism in lactating dairy cows fed finely ground field pea plus soybean meal or canola meal with or without rumen-protected methionine supplementation.
Pereira, A.B.D., Moura, D.C., Whitehouse, N.L. and Brito, A.F., 2020. Journal of Dairy Science, 103(4), pp.3161-3176.
https://www.sciencedirect.com/science/article/abs/pii/S0022030220305026
Effect of heat-treated canola meal and glycerol inclusion on performance and gastrointestinal development of Holstein calves.
Burakowska, K., Górka, P., Kent-Dennis, C., Kowalski, Z.M., Laarveld, B. and Penner, G.B., 2020. Journal of Dairy Science, 103(9), pp.7998-8019.
https://onlinelibrary.wiley.com/doi/abs/10.1002/jsfa.10572
The effect of pistachio by‐product extracts treatment in protecting soybean meal and canola meal protein from rumen microbial degradation.
Shakeri, P., Reiasi, A. and Tahmasbi, R., 2020. Journal of the Science of Food and Agriculture.
https://www.mdpi.com/2076-2615/10/10/1735
Replacement of Soybean Meal with Heat-Treated Canola Meal in Finishing Diets of Meatmaster Lambs: Physiological and Meat Quality Responses.
Sekali, M., Mlambo, V., Marume, U. and Mathuthu, M., 2020. Animals, 10(10), p.1735.
https://www.mdpi.com/2076-2615/9/10/704
Feeding canola, camelina, and carinata meals to ruminants.
Paula, E.M., da Silva, L.G., Brandao, V.L.N., Dai, X. and Faciola, A.P., 2019. Animals, 9(10), p.704.
https://www.sciencedirect.com/science/article/pii/S0022030219302887
Does blending canola meal with other protein sources improve production responses in lactating dairy cows? A multilevel mixed-effects meta-analysis.
Martineau, R., Ouellet, D.R. and Lapierre, H., 2019. Journal of dairy science, 102(6), pp.5066-5078.
https://www.sciencedirect.com/science/article/pii/S0022030219305831
Performance of dairy cows fed diets formulated at 2 starch concentrations with either canola meal or soybean meal as the protein supplement.
Sánchez-Duarte, J.I., Kalscheur, K.F., Casper, D.P. and García, A.D., 2019. Journal of dairy science, 102(9), pp.7970-7979.
https://www.sciencedirect.com/science/article/pii/S0377840118312938
Impacts of incremental substitution of soybean meal for canola meal in lactating dairy cow diets containing a constant base level of corn derived dried distillers’ grains with solubles.
Gauthier, H., Swanepoel, N. and Robinson, P.H., 2019. Animal feed science and technology, 252, pp.51-63.
https://www.publish.csiro.au/AN/AN17346
Milk production of cows grazing pasture supplemented by a partial mixed ration with or without canola meal.
Auldist, M.J., Wright, M.M., Marett, L.C., Hannah, M.C., Kennedy, E., Jacobs, J.L. and Wales, W.J., 2019. Animal Production Science, 59(4), pp.778-786.
https://www.sciencedirect.com/science/article/pii/S0377840120301620
Impacts of substitution of canola meal with soybean meal, with and without ruminally protected methionine, on production, reproduction and health of early lactation multiparous Holstein cows through 160 days in milk.
Swanepoel, N., Robinson, P.H. and Conley, A., 2020. Animal Feed Science and Technology, p.114494.
https://www.sciencedirect.com/science/article/pii/S0377840117312191
Impacts of increasing levels of canola meal in diets of high producing Holstein cows on their productive performance.
Robinson, P.H. and Swanepoel, N., 2018. Animal Feed Science and Technology, 237, pp.175-185.
https://www.wageningenacademic.com/doi/abs/10.3920/978-90-8686-891-9_137
Amino acid digestibility of canola meal estimated with pulse-dose in dairy cows or in roosters.
Békri, K., Roussi, A., Lapierre, H., Pellerin, D. and Ouellet, D.R., 2019. In EAAP Scientific Series (pp. 1701-1714). Wageningen Academic Publishers.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6722309/
Protein molecular structure, degradation and availability of canola, rapeseed and soybean meals in dairy cattle diets.
Tian Y, Zhang X, Huang R, Yu P. Asian-Australasian journal of animal sciences. 2019 Sep;32(9):1381.
https://www.ars.usda.gov/research/publications/publication/?seqNo115=353470/
Canola Meal for Early Lactation Cows
Moore, S.A. and Kalscheur, K.F., 2018, June.. In Symposium Proceedings (pp. 149-151).
https://lst.uk.ac.ir/article_2056.html
Effects of replacing soybean meal with canola meal and decreasing crude protein on milk production and nutrient utilization of dairy cows in early lactation.
Toti, J., Ghasemi, E. and Khorvash, M., 2018. Journal of Livestock Science and Technologies, 6(1), pp.19-29.
https://search.proquest.com/openview/722540355643bca1e87713c0143c629d/1?pq-origsite=gscholar&cbl=49113
Canola meal in dairy cow diets during early lactation increases production compared with soybean meal.
Moore, S.A.E. and Kalscheur, K.F., 2016. Journal of Animal Science, 94, pp.731-731.
Moura et al., 2018. Meta-analysis of the use of canola meal in diets for dairycows.
https://www.adsa.org/Portals/0/SiteContent/Docs/Meetings/PastMeetings/Annual/2018/087.pdf (M285)
Moore, S.A.E. and Kalscheur, K.F., 2018. Evaluation of a limit feeding strategy with canola or soybean meals on dairy cow performance.
https://www.adsa.org/Portals/0/SiteContent/Docs/Meetings/PastMeetings/Annual/2018/290.pdf (T229)
Auldist et al, 2017. Inclusion of canola meal increases milk response in partial mixed rations and grain mixes fed to grazing cows. https://www.adsa.org/Portals/0/SiteContent/Docs/Meetings/PastMeetings/Annual/2017/299.pdf (T244)
Paula et al., 2016. Effects of replacing soybean meal with canola meal or treated canola meal on ruminal digestion, and omasal nutrient fow in lactating dairy cows.
https://www.adsa.org/Portals/0/SiteContent/Docs/Meetings/PastMeetings/Annual/2016/614.pdf (1404)
Paula et al., 2016. Effects of replacing soybean meal with canola meal or treated canola meal on nitrogen metabolism and total tract digestibility in lactating dairy cows.
https://www.adsa.org/Portals/0/SiteContent/Docs/Meetings/PastMeetings/Annual/2016/614.pdf (1584)
Abeysekara, S and T. Mutsvangwa. 2016. Effects of feeding canola meal or wheat dried distillers’ grains with solubles alone or in combination as the major protein sources on ruminal function and production in dairy cows.
https://www.adsa.org/Portals/0/SiteContent/Docs/Meetings/PastMeetings/Annual/2016/614.pdf (1580)
Canola Meal versus Soybean Meal in Dairy Cow Diets Kenneth F. Kalscheur, Spencer A. E. Moore
Canola Meal, a Proven Advantage in Various Diet Formulations Kenneth. F. Kalscheur, Spencer A. E. Moore
Canola Meal as a Protein Source for Lactating Dairy Cows) Antonio Faciola, Eduardo M. Paula
True Value of Feeding Canola Meal Essi Evans
Why All of the Hype About Feeding Canola Meal? Essi Evans, Brittany Dyck
Getting Canola Meal Values Right in Your Formulation Essi Evans, Brittany Dyck