

Browse the Canola Meal Feed Guide by chapter:
Canadian solvent-extracted canola meal is derived from a blend of Brassica napus, Brassica rapa and Brassica juncea seed. The majority (> 95%) of the seed produced in Canada is Brassica napus. As with any crop, there is some variability in the nutrient composition of canola meal due to variation in environmental conditions during the growing season of the crop, harvest conditions, and to a minor extent, by cultivar and processing of the seed and meal. The basic nutrient composition of canola meal is shown in Table 1. These results are based on an extensive survey of 13 processing sites, conducted over a seven-year period.
Table 1. Composition of solvent extracted canola meal as determined from a 7-year survey of 13 Canadian processing plants1.
| wdt_ID | Component | 12% Moisture Basis | Dry Matter Basis |
|---|---|---|---|
| 2 | Moisture, % | 12.00 | 0.00 |
| 3 | Crude protein (N*6.25), % | 36.90 | 42.00 |
| 4 | Rumen escape protein, % of protein (NRC method)2 | 43.50 | 43.50 |
| 5 | Rumen escape protein, % of protein (CNCPS method)3 | 53.00 | 53.00 |
| 6 | Ether extract, % | 2.81 | 3.20 |
| 7 | Oleic acid, % | 1.74 | 1.98 |
| 8 | Linoleic acid, % | 0.56 | 0.64 |
| 9 | Linolenic acid, % | 0.24 | 0.27 |
| 10 | Ash, % | 6.42 | 7.30 |
| 11 | Calcium, % | 0.67 | 0.76 |
| 12 | Phosphorus, % | 1.03 | 1.17 |
| 13 | Total dietary fibre % | 33.60 | 38.20 |
| 14 | Acid detergent fibre, % | 16.30 | 18.60 |
| 15 | Neutral detergent fibre, % | 25.50 | 29.00 |
| 16 | Sinapine, % | 0.88 | 1.00 |
| 17 | Phytic acid, % | 2.02 | 2.30 |
| 18 | Glucosinolates, uMol/g | 3.14 | 3.57 |
1 Radfar et al., 2017; 2 Broderick et al., 2016; 3 Ross et al., 2013.
For trading purposes, the minimum crude protein value of solvent extracted canola meal is 36%, on a 12% moisture basis. While the minimum crude protein guarantee for Canadian canola meal is 36% (12% moisture basis), the actual protein content usually ranges between 37 and 40%. The minimum allows for yearly variation in canola seed composition due to growing conditions. The influence of weather and soil conditions on the protein content of Canadian canola meal from 2000 to 2023 is shown in Figure 1. As the chart indicates, the protein content of canola meal varies from about 37–42% when calculated on an oil-free, 12% moisture basis.
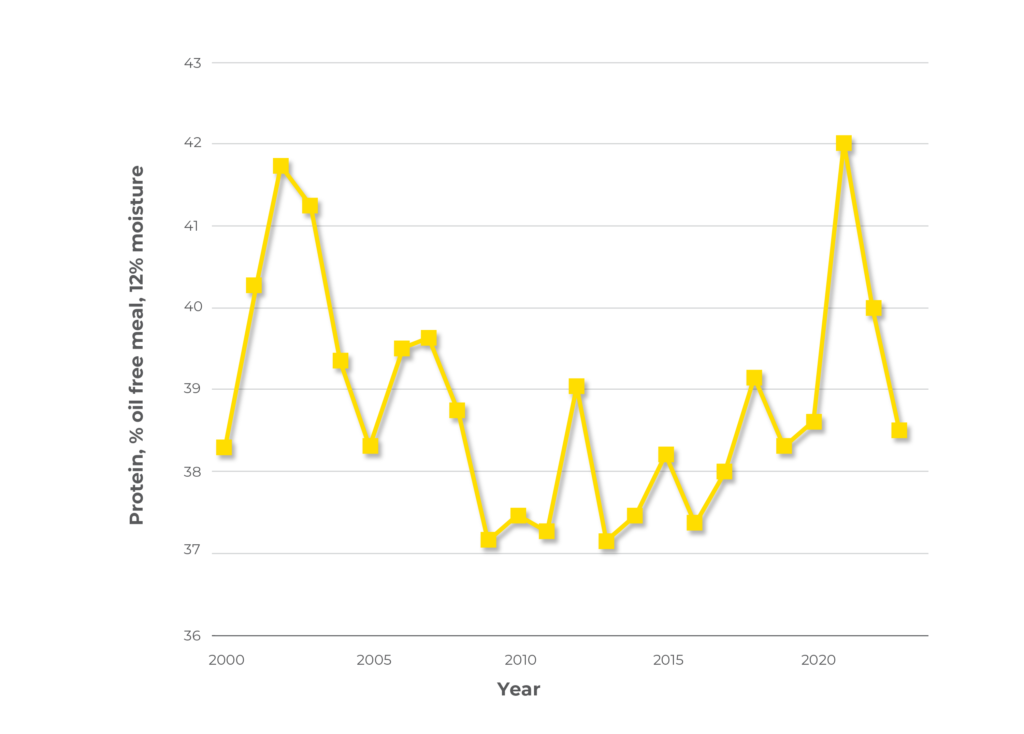
Figure 1. Protein content of canola meal from 2000 to 2023. Protein values calculated on an oil-free, 12% moisture basis (Canadian Grains commission, https://grainscanada.gc.ca/en/grain-research/grain-harvest-export-quality/canola/2023/
The amino acid profile of canola meal is well suited for animal feeding (Table 2). Like many vegetable protein sources, canola meal is limiting in lysine, but the meal is noted for having high levels of methionine and cysteine. The amino acid profile values in Table 2 were corrected to a 36% protein basis, and are therefore likely lower than actual. Amino acid content varies with protein content and can be calculated by multiplying the crude protein content of the meal by the proportion of amino acid as a percentage of protein, as shown.
Table 2. Amino acid composition of canola meal on a 36% as-fed protein basis1,2.
| wdt_ID | Amino Acid | % of Meal | % of Crude Protein |
|---|---|---|---|
| 2 | Alanine | 1.58 | 4.38 |
| 3 | Arginine | 2.19 | 6.08 |
| 4 | Aspartate + Asparagine | 2.49 | 6.92 |
| 5 | Glutamate + Glutamine | 6.22 | 17.28 |
| 6 | Glycine | 1.73 | 4.81 |
| 7 | Histidine | 1.08 | 3.00 |
| 8 | Isoleucine | 1.38 | 3.84 |
| 9 | Leucine | 2.38 | 6.60 |
| 10 | Lysine | 2.04 | 5.66 |
| 11 | Methionine | 0.69 | 1.93 |
| 12 | Methionine + cysteine | 1.33 | 3.69 |
| 13 | Phenylalanine | 1.34 | 3.71 |
| 14 | Proline | 2.49 | 6.92 |
| 15 | Serine | 1.32 | 3.66 |
| 16 | Threonine | 1.43 | 3.97 |
| 17 | Tryptophan (2) | 0.48 | 1.33 |
| 18 | Tyrosine | 0.90 | 2.51 |
| 19 | Valine | 1.61 | 4.46 |
1 Radfar et al., 2017; 2 Broderick et al., 2016; 3 Ross et al., 2013.
The ether extract content of Canadian canola meal tends to be relatively high at 3.2% (Table 1) compared to 1–2% in canola and rapeseed meals produced in most other countries. In Canada, it is general practice to include canola glycolipids and phospholipids back with the meal during the refining of the oil. Likewise, canola meal may further contain 1–2% of the free fatty acids that are derived from canola oil refining. These components increase the energy value of the meal and help to reduce dustiness.
Table 3. Fatty acid composition of canola oil1.
| wdt_ID | Fatty Acid | % of Total Fatty Acids |
|---|---|---|
| 2 | C16:0 Palmitic acid | 4.5 |
| 3 | C16:1 Palmitoleic acid | 0.2 |
| 4 | C18:0 Stearic acid | 2.4 |
| 5 | C18:1 Oleic acid | 64.5 |
| 6 | C18:2 Linoleic acid (omega 6) | 17.7 |
| 7 | C18:3 Linolenic acid (omega 3) | 8.6 |
| 8 | C22:1 Erucic acid | < 0.1 |
| 9 | Total saturated | 7.8 |
| 10 | Total monounsaturated | 65.4 |
| 11 | Total polyunsaturated | 26.3 |
1 Ghazani and Marangoni, 2013.
Table 3 provides the complete fatty acid analysis for canola oil. As the table shows, this oil contains only a small amount of saturated fatty acids, and a high concentration of oleic acid. Canola meal provides a 2:1 ratio of omega-6 to omega-3 fatty acids and is a good source of omega-3 fatty acids. Canola oil is sometimes used in diets to enrich the fatty acid profile of milk, meat or eggs (Gallardo, et al., 2012; Gül, et al., 2012; Chelikani, et al., 2004).
The carbohydrate matrix of canola meal is quite complex (Table 4). The fibre content is higher than for some vegetable proteins, as the hull cannot be readily removed from the seed. Much of the fibre is in the form of acid detergent fibre (ADF), with neutral detergent fibre (NDF) levels about 10% higher than ADF. The non-fibre component is rich in sugar, which is mostly provided as sucrose (Table 4).
Table 4. Carbohydrate and dietary fibre components of canola meal1,2,3
| wdt_ID | CARBOHYDRATE FRACTIONS | 12% MOISTURE BASIS | DRY MATTER BASIS |
|---|---|---|---|
| 1 | Monosaccharides (Fructose and Glucose), % | 1.55 | 1.76 |
| 2 | Disaccharides (sucrose), % | 5.58 | 6.34 |
| 3 | Oligosaccharides, % | 2.23 | 2.53 |
| 4 | Starch, % | 0.43 | 0.49 |
| 5 | Acid detergent fibre, % | 16.32 | 18.55 |
| 6 | Neutral detergent fibre, % | 25.51 | 28.99 |
| 7 | Total dietary fibre, % | 34.53 | 39.24 |
| 8 | Non-Starch polysaccharides, % | 20.15 | 22.90 |
| 9 | Cellulose, % | 7.65 | 8.69 |
| 10 | Non-cellulosic polysaccharides, % | 12.50 | 14.21 |
| 11 | Glycoprotein(NDF insoluble crude protein), % | 4.30 | 4.89 |
| 12 | Lignin and polyphenols, % | 8.68 | 9.86 |
| 13 | Lignin, % | 5.82 | 6.61 |
1 Adewole et al., 2016; 2 Broderick et al., 2016; 3 Slominski and Rogiewicz, unpublished.
Most references on the mineral content of canola meal use the values derived by Bell and Keith (1991), which were reconfirmed in a survey by Bell et al. (1999), and again by the current survey (Broderick et al., 2016; Adewole et al., 2016). The data show that canola meal is a relatively good source of essential minerals (Table 5) compared to other oilseed meals. Canola meal is an especially good source of selenium and phosphorus. Like other vegetable protein sources of phosphorus, a portion of the total is in the form of phytate.
Table 5. Mineral content of canola meal1,2,3
| wdt_ID | Mineral | 12% Moisture Basis | Dry Matter Basis |
|---|---|---|---|
| 1 | Calcium, % | 0.65 | 0.74 |
| 2 | Phosphorus, % | 0.99 | 1.13 |
| 3 | Phytate phosphorus, % | 0.64 | 0.73 |
| 4 | Non-phytate phosphorus, % | 0.35 | 0.40 |
| 5 | Sodium, % | 0.07 | 0.08 |
| 6 | Chlorine, % | 0.10 | 0.11 |
| 7 | Potassium, % | 1.13 | 1.28 |
| 8 | Sulfur, % | 0.63 | 0.72 |
| 9 | Magnesium, % | 0.54 | 0.61 |
| 10 | Copper, mg/kg | 4.70 | 5.30 |
| 11 | Iron, mg/kg | 162.00 | 184.00 |
| 12 | Manganese, mg/kg | 58.00 | 66.00 |
| 13 | Molybdenum, mg/kg | 1.40 | 1.60 |
| 14 | Zinc, mg/kg | 47.00 | 53.00 |
| 15 | Selenium, mg/kg | 1.10 | 1.30 |
1 Adewole et al., 2016; 2 Sauvant et al., 2002; 3 Dairy One (www.dairyone.com).
Information on the vitamin content of canola meal is very limited and the values provided in Table 6 were averaged from four sources (Wickramasuriya et al., 2015). Canola meal is noted as rich in choline, biotin, folic acid, niacin, riboflavin and thiamine (NRC, 2012). As is recommended with most natural sources of vitamins in animal feeds, users should not place too much reliance on these values and use supplemental vitamin premixes instead.
Table 6. Vitamin content of canola meal1
| wdt_ID | Vitamin | 12% Moisture Basis | Dry Matter Basis |
|---|---|---|---|
| 1 | Biotin, mg/kg | 1.08 | 1.22 |
| 2 | Choline, g/kg | 6.70 | 7.6 |
| 3 | Folic acid, mg/kg | 1.55 | 1.76 |
| 4 | Niacin, mg/kg | 160.00 | 182 |
| 5 | Pantothenic acid, mg/kg | 9.40 | 10.6 |
| 6 | Pyridoxine, mg/kg | 7.10 | 8.10 |
| 7 | Riboflavin, mg/kg | 5.80 | 6.5 |
| 8 | Thiamine, mg/kg | 5.20 | 5.9 |
| 9 | Vitamin E, mg/kg | 18.50 | 21.0 |
1 Wickramasuriya et al., 2015.
Rapeseed meal, the parent of canola meal, is recognized as an ingredient that may need to be limited in diets for livestock and fish due to certain anti-nutritional factors, primarily glucosinolates. These factors have been reduced in Canadian canola meal to levels that do not pose threats to performance and feeding for most species.
Glucosinolates are a large group of secondary plant metabolites common to all cruciferous plants. While nontoxic on their own, breakdown products of glucosinolates can adversely affect animal performance. Canola glucosinolates are composed of two main types, aliphatic and indolyl (or indole) glucosinolates. Aliphatic glucosinolates make up approximately 85% of the glucosinolates present in canola meal, while indolyl glucosinolates account for the other 15% (Adewole et al., 2016). The low glucosinolate content of canola, compared to previous cultivars of rapeseed, constitutes the major improvement in meal quality achieved by plant breeders.
The average total glucosinolate content of Canadian canola meal, based on seven years of data, is 3.6 μmol/g (Slominski and Rogiewicz, unpublished). By comparison, traditional rapeseed meal contains levels as high as 120 μmol/g of total glucosinolates. The reason that glucosinolates are expressed on a molecular (μmol/g) basis rather than on a weight (mg/kg) basis is that glucosinolates have significantly different molecular weights, depending on the size of their aliphatic side chain. Since the negative effect on the animal is at the molecular level, the most accurate estimate of this effect must be gauged by expressing glucosinolate concentration on a molecular basis.
According to the most recent data provided by The Canadian Grains Commission (2023) (https://www.grainscanada.gc.ca/en/grain-research/export-quality/oilseeds/canola/2021/08-glucosinolate.html) the content of glucosinolate compounds in canola seed is low and has not changed noticeably since 2000. The level of glucosinolates in Canadian canola seed prior to processing has averages around 10 μmol/g. Glucosinolate content is then concentrated in the meal; after that, the glucosinolates are reduced during processing to values averaging 3.6 μmol/g.
Consumption of this fatty acid has been associated with myocardial lesions. However, Canadian plant breeders successfully reduced the amount of erucic acid in canola oil to very near zero levels (Figure 2). Erucic acid is no longer considered a problem for either the meal or the oil.
Tannins are present in canola meal at a range of 1.5–3.0%, with brown-seeded varieties having higher levels than yellow-seeded varieties. The tannins in canola meal are associated with the hull and are primarily insoluble. These tannins do not appear to have the same negative effects on palatability and protein digestibility that they do in other edible plants (Khajali and Slominski, 2012).
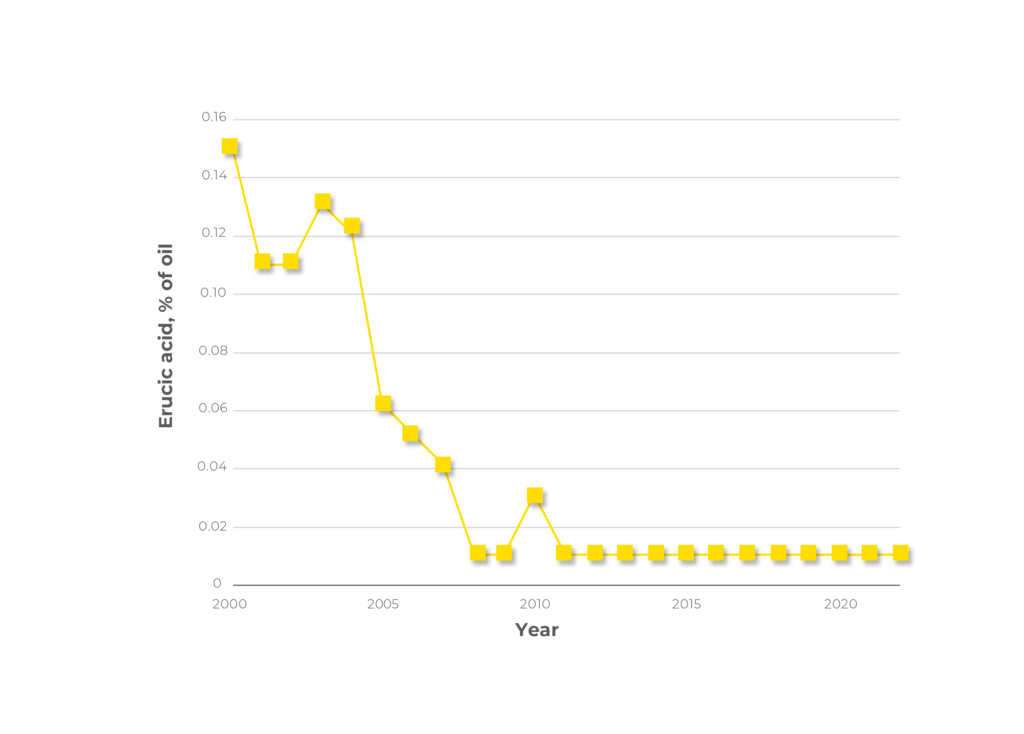
Figure 2. Erucic acid levels in canola oil from 2000 to 2022. (https://www.grainscanada.gc.ca/en/grain-research/export-quality/oilseeds/canola/2021/10-fatty-acid-composition.html).
Several terms are used interchangeably to differentiate solvent extracted versus expeller-extracted meals. Terms commonly used to describe the meal include expeller meal, double-press meal and presscake. Currently in Canada, a small percentage of seed is processed using the expeller method. Smaller oilseed plants as well as those associated with some biodiesel plants use double-press expeller processing rather than solvent extraction. Since the oil is extracted simply by mechanical means, the resulting meal contains significantly more oil than that of standard solvent-extracted canola meal.
The nutritional profile of the meal is like that of canola meal, except that it contains 8–12% fat and therefore has much higher energy values. The nutritional composition of expeller meal is provided in Table 7. Fat content can vary widely, so it is important that the expeller meal is analyzed for fat, and the energy value adjusted accordingly. High levels of fat will also dilute other nutrients in the resultant meal, relative to solvent-extracted canola meal.
Table 7. Typical composition of expeller canola meal1.
| Component | Component | 12% Moisture Basis | Dry Matter Basis |
|---|---|---|---|
| 1 | Moisture (as measured), % | 4.02 | |
| 2 | Crude protein (N*6.25), % | 34.28 | 38.95 |
| 3 | Rumen escape protein, % of protein (NRC method) (2) | 48.50 | 48.50 |
| 4 | Rumen escape protein, % of protein (CNCPS method) (3) | 59.10 | 59.10 |
| 5 | Ether extract, % | 10.96 | 12.44 |
| 6 | Oleic acid, % | 6.85 | 7.75 |
| 7 | Linoleic acid, % | 2.20 | 2.50 |
| 8 | Linolenic acid, % | 0.91 | 1.03 |
| 9 | Ash, % | 6.96 | 7.90 |
| 10 | Calcium, % | 0.62 | 0.71 |
| 11 | Phosphorus, % | 0.96 | 1.09 |
| 12 | Total dietary fibre | 37.07 | 42.12 |
| 13 | Acid detergent fibre, % | 16.72 | 19.00 |
| 14 | Neutral detergent fibre, % | 26.83 | 30.49 |
| 15 | Glucosinolates, μmol/g | 8.85 | 10.06 |
| 16 | Methionine, % of crude protein | 1.93 | 1.93 |
| 17 | Lysine, % of crude protein | 5.93 | 5.93 |
| 18 | Threonine, % of crude protein | 3.69 | 3.69 |
1 Adewole et al., 2016; 2 Broderick et al., 2016; 3 Ross et al., 2013.
The key nutrient values for canola seed are shown in Table 8. These values were obtained from recent publications (Assadi et al., 2011; Leterme et al., 2008). Most nutrient values for canola seed can be calculated from the nutrient values in canola meal and oil, considering that approximately 56% of the seed is meal and 44% is oil. The exception is energy content, because the energy value of canola seed cannot be estimated reliably from the addition of the energy values for canola oil and meal. For swine and poultry, the seed has less energy than the sum of its oil and meal components. This is likely because whole canola seed is not processed to the same degree as canola oil and meal; and therefore, not as well digested. Heat treatment and particle size reduction of canola seed by micronization, extrusion or expansion is often used to increase its energy digestibility.
Table 8. Reported chemical composition of canola seed (12% moisture basis).
| wdt_ID | Components | Feedipedia, 2018 | Assadi et al., 2011 | Montoya and Leterme, 2008 | DairyOne, 2023 |
|---|---|---|---|---|---|
| 2 | Moisture, % | 6.8 | 5.0 | 5.7 | 5.8 |
| 3 | Crude protein, % | 18.4 | 20.0 | 20.7 | 21.5 |
| 4 | Ether extract,% | 40.5 | 43.8 | 38.6 | 34.5 |
| 5 | Linoleic acid, % | 8.3 | 8.5 | 7.9 | – |
| 6 | Linolenic acid, % | 4.1 | 4.2 | 3.9 | – |
| 7 | Ash, % | 3.8 | 3.7 | 4.1 | 4.7 |
| 8 | Crude fibre, % | 8.9 | – | – | 8.9 |
| 9 | ADF, % | 12.7 | – | 10.6 | 15.9 |
| 10 | NDF, % | 17.9 | 16.6 | 12.9 | 22.5 |
| 11 | Calcium, % | 0.43 | – | – | 0.39 |
| 12 | Phosphorus, % | 0.64 | – | – | 0.65 |
Adewole, D.I., Rogiewicz, A., Dyck, B. and Slominski, B.A., 2016. Chemical and nutritive characteristics of canola meal from Canadian processing facilities. Animal Feed Science and Technology, 222, pp.17-30.
Assadi, E., Janmohammadi, H., Taghizadeh, A. and Alijani, S., 2011. Nutrient composition of different varieties of full-fat canola seed and nitrogen-corrected true metabolizable energy of full-fat canola seed with or without enzyme addition and thermal processing. Journal of Applied Poultry Research, 20(1), pp.95-101.
Bell, J.M. and Keith, M.O., 1991. A survey of variation in the chemical composition of commercial canola meal produced in Western Canadian crushing plants. Canadian Journal of Animal Science, 71(2), pp.469-480.
Bell, J.M., Rakow, G. and Downey, R.K., 1999. Mineral composition of oil-free seeds of Brassica napus, B. rapa and B. juncea as affected by location and year. Canadian Journal of Animal Science, 79(3), pp.405-408.
Broderick, G.A., Colombini, S., Costa, S., Karsli, M.A. and Faciola, A.P., 2016. Chemical and ruminal in vitro evaluation of Canadian canola meals produced over 4 years. Journal of Dairy Science, 99(10), pp.7956-7970.
Canadian Grains Commission. 2023. https://www.grainscanada.gc.ca/en/grain-research/export-quality/oilseeds/canola/2022/
Chelikani, P.K., Bell, J.A. and Kennelly, J.J., 2004. Effects of feeding or abomasal infusion of canola oil in Holstein cows 1. Nutrient digestion and milk composition. Journal of Dairy Research, 71(3), pp.279-287.
DairyOne, Feed Composition Library. 2023. https://dairyone.com/services/forage-laboratory-services/feed-composition-library/
Evonik, AminoDat 6.2.2023. https://animal-nutrition.evonik.com/en/products-and-solutions/feed-quality/aminodat-62-132013.html
Feedipedia.org. 2018. https://www.feedipedia.org/
Gallardo, M.A., Pérez, D.D. and Leighton, F.M., 2012. Modification of fatty acid composition in broiler chickens fed canola oil. Biological Research, 45(2), pp.149-161.
Ghazani, S.M. and Marangoni, A.G., 2013. Minor components in canola oil and effects of refining on these constituents: A review. Journal of the American Oil Chemists’ Society, 90, pp.923-932.
Grieve, S.M. 1978. Rapeseed gums for lactating dairy cows. 57th Annual Feeders’ Day Report, University of Alberta, p. 66.
Gül, M., Yörük, M.A., Aksu, T., Kaya, A. and Kaynar, Ö., 2012. The effect of different levels of canola oil on performance, egg shell quality and fatty acid composition of laying hens. International Journal of Poultry Science, 11(12), p.769.
Khajali, F. and Slominski, B.A., 2012. Factors that affect the nutritive value of canola meal for poultry. Poultry Science, 91(10), pp.2564-2575.
Leterme, P., Kish, P. and Beaulieu, A.D., 2008. Digestibility energy determination of canola meal and full-fat canola seeds in pigs: limitations of the substitution method. Journal of Animal Science, 86 (Suppl. 2) pp. 186.
Mathison, G.W., 1978. Rapeseed gum in finishing diets for steers. Canadian Journal of Animal Science, 58(2), pp.139-142.
McCuaig, L.W. and Bell, J.M., 1981. Effects of rapeseed gums on the feeding value of diets for growing-finishing pigs. Canadian Journal of Animal Science, 61(2), pp.463-467.
Montoya, C.A. and Leterme, P., 2010. Validation of the net energy content of canola meal and full-fat canola seeds in growing pigs. Canadian Journal of Animal Science, 90(2), pp.213-219.
NRC. 2012. Nutrient Requirements of Swine. National Academy of Sciences, Washington, D.C.
Qiao, H. and Classen, H.L., 2003. Nutritional and physiological effects of rapeseed meal sinapine in broiler chickens and its metabolism in the digestive tract. Journal of the Science of Food and Agriculture, 83(14), pp.1430-1438.
Sauvant, D., Perez, J.M. and Tran, G, 2004. Tables of composition and nutritional value of feed materials. Wageningen Academic Publishers, INRA Editions.
Summers, J.D., Leeson, S. and Slinger, S.J., 1978. Performance of egg-strain birds during their commercial life cycle when continuously fed diets containing Tower rapeseed gums. Canadian Journal of Animal Science, 58(2), pp.183-189.
Radfar, M., Rogiewicz, A. and Slominski, B.A., 2017. Chemical composition and nutritive value of canola-quality Brassica juncea meal for poultry and the effect of enzyme supplementation. Animal Feed Science and Technology, 225, pp.97-108.
Wickramasuriya, S.S., Yi, Y.J., Yoo, J., Kang, N.K. and Heo, J.M., 2015. A review of canola meal as an alternative feed ingredient for ducks. Journal of Animal Science and Technology, 57(1), pp.1-9.