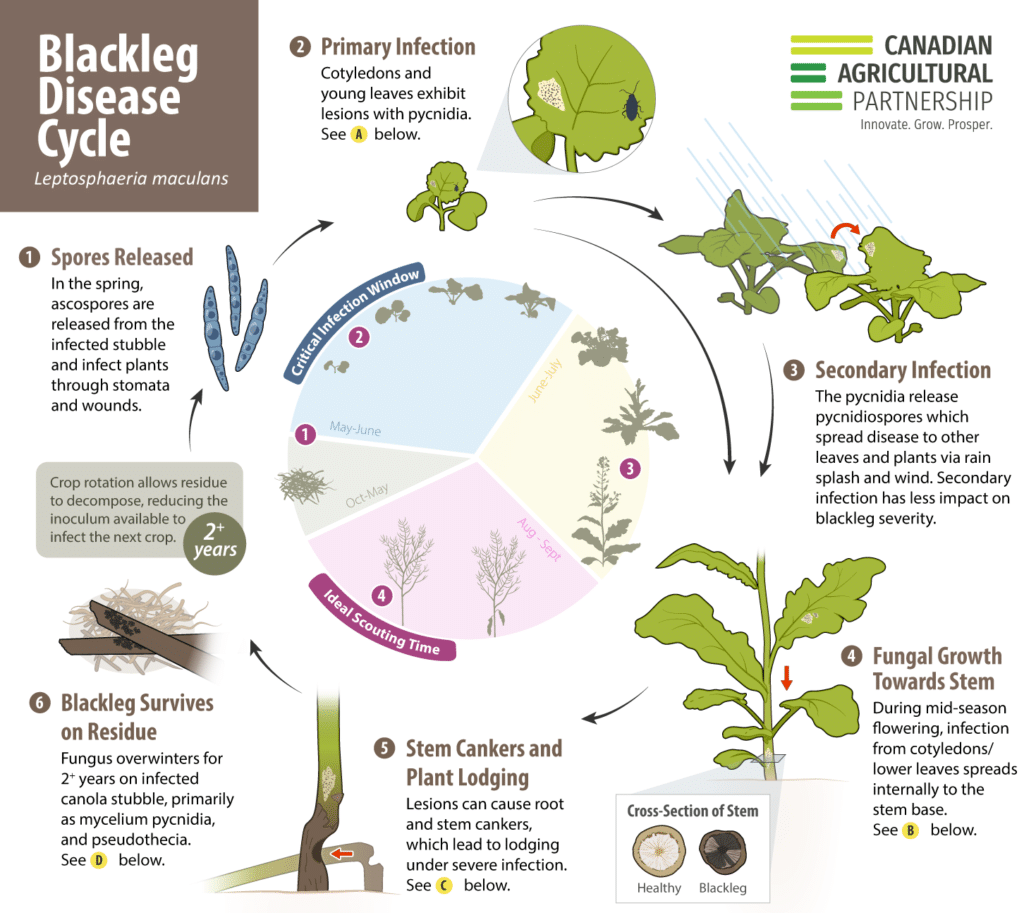
Although scouting for blackleg (and assessment of potential damage) usually occurs near the end of the growing season, the most critical stage of infection is actually at the cotyledon stage. Reviewing disease cycle details like this helps illuminate the timing and reasoning behind necessary steps to outsmart the fungus that causes this disease. Using available videos, graphics and explanations to clarify the key blackleg resistance concepts can help empower proactive decision-making for maximum disease management impact. Browsing a few research findings can also upgrade your knowledge of the interesting host-pathogen dynamic.
Blackleg disease cycle refresher
The fungus that primarily causes blackleg disease (Leptosphaeria maculans) has multiple interactions with canola plants throughout the season. Even before canola seed is planted, the fungus may already be in a field – since it can overwinter for two or more years on infected canola stubble (hence the benefit of crop rotation allowing time for host plants and infected residue to decompose and the amount of fungus available to infect the field to decrease). It can also overwinter on volunteer canola and related Brassica weeds and that it may have previously infected, so controlling these host weeds also helps reduce the amount of blackleg-causing fungus in the field.
In the spring, tiny spores, called ascospores and pycnidiospores, are released into the air from the infested stubble and can infect young canola cotyledons, and to less extent, lower true leaves.
As the infected plant grows and develops the fungus grows downward through the stem. This can lead to cankering at the base of the root, where the fungus restricts the moisture and nutrient supply to the plant, causing yield loss. See the Blackleg disease cycle (pictured below) for more details and use the blackleg yield loss calculator to estimate the impact of the disease.
Apply this on your farm
- Check out the blackleg disease cycle
- Use the Blackleg management guide to map out an effective blackleg strategy
- Recalling blackleg race diversity, submit samples from each field for blackleg testing
- Interpret results, save records and consult canola trait labels before booking seed for each field
- Remember to use the helpful resources available to you
The ideal time to scout, identify blackleg and assess the damage it has caused is when the crop is ripening until just after harvest. However, canola stem samples can still be submitted to labs all through the fall and even in early spring, as noted in the Blackleg testing podcast (provided above).

The lab will determine if the blackleg-causing fungus is present, the predominant race of blackleg found within the samples collected and specific information on the blackleg race. This knowledge can then inform which resistance genes are needed in the canola cultivar that is planted next time (in the rotation) in that canola is grown on the field that was tested.
For a clear, calm description of this test and how to interpret it, see this ‘How to interpret race tests for blackleg resistance selection‘ video (provided above).
When the annual seed purchasing decision time is approaching (for the fields that will have canola, which could be a couple years after the tests were completed), revisit the results before checking the Current canola cultivar trait labels table to determine which cultivars contain the blackleg resistance groups needed. Then use the Blackleg Management Guide and to effectively integrate a range of tools into a cohesive blackleg management strategy.
Prairie blackleg race survey
The reason blackleg race identification tests and the blackleg resistance group information is needed to optimize blackleg management is because the fungus that causes the disease has a lot of diversity.
Building on his earlier Analysis and monitoring of Leptosphaeria maculans race dynamics in western Canada for effective blackleg resistant management strategies project, Dr. Gary Peng recently completed Monitoring the race dynamics of Leptosphaeria maculans for effective deployment and rotation of resistance genes for sustainable management of blackleg of canola in western Canada.
This project analyzed annual L. maculans population on the Prairies (between 2017-2021) and identified 83 different races in the L. maculans population. Of these races identified, the top 10 races accounted for 72.3%, 93.7% and 57.1% of the populations in Alberta, Saskatchewan and Manitoba.
Results
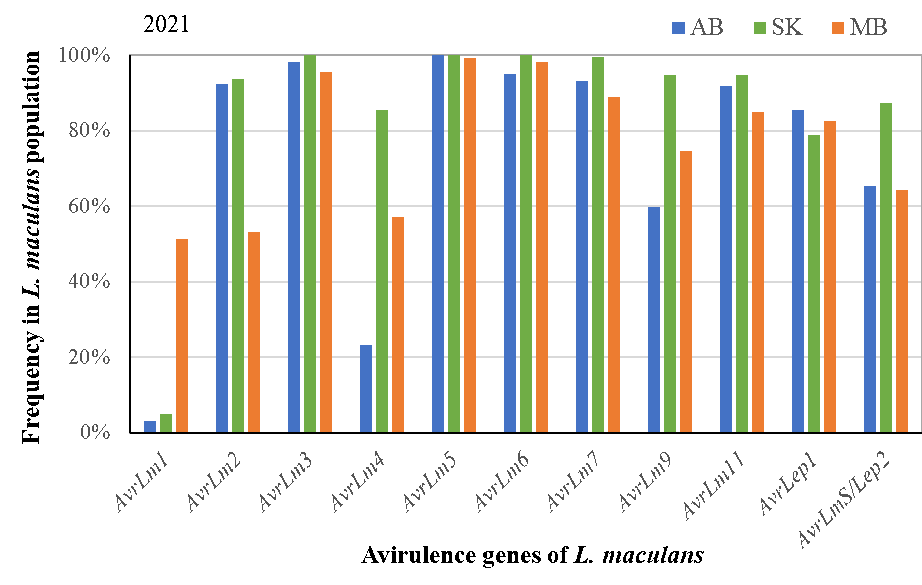
These results provide important guidance to the selection of effective genes for blackleg resistance breeding, as well as the recommendation of canola cultivars carrying proper/effective resistance (R) genes.
The diversity in the pathogen also shows the potential for resistance erosion as the project reported that virulent races (the races that can infect resistance cultivars) are present for almost all resistance genes, except for one (Rlm10). To manage selection pressure/resistance breakdown, judicious uses of resistance genes will be important.
The research also reported that canola cultivars carrying any of the resistance genes Rlm5, Rlm6, Rlm7, Rlm10, Rlm11 and LepR1 are likely still resistant to blackleg in western Canada. This is due to the common presence of corresponding avirulence (Avr) genes in the pathogen population. However, the resistance genes Rlm3 or Rlm9 may be ineffective due to the ‘masking effect’ of AvrLm7 to AvrLm3 and AvrLm9. Rlm4 may be less effective in Alberta due to low levels of AvrLm4.
Conclusions and applications
The results of this study have prompted the deployment of new resistance genes (including Rlm2, Rlm4 and Rlm7) in canola cultivars, which is very helpful. Growers and agronomists can also use this information to better select and rotate canola cultivars with effective resistance genes for management of blackleg on a regional basis.
As well, judicious deployment of resistance genes and rotation of resistant cultivars (in addition to crop rotation) is important for genetic control of blackleg. If a resistance gene is used repeatedly, there will be selection pressure for virulent races that can eventually break down the resistance.
Implementing improved resistance management

Blackleg requires an integrated management strategy that utilizes a range of tools to minimize the risk to canola. Work through all four levels of the Blackleg Management Guide to clearly understand your risk of blackleg damage and how to manage it.
- For a better understanding of the blackleg disease cycle, watch this informative Blackleg disease video.
- To support on your monitoring efforts:
- Check out this concise Blackleg scouting video.
- Reference this blackleg disease severity scale.
- Use the online tool to calculate your blackleg yield loss.
- For help preparing and submitting samples for blackleg identification tests, watch this one-minute Blackleg testing video.
- An explanation of the blackleg resistance and related management tools is available in the Blackleg in canola video.
Ongoing blackleg research progress
Blackleg research has made good progress over the years, as noted in the Blackleg research through the years video (embedded below).
A couple successes along the way include these findings:
- Dr. Borhan and Dr. Lydiate produced a set of single blackleg-resistance gene B. napus lines in a common susceptible background in the Defining populations of the L. maculans pathogen in test sites used for canola blackleg resistance trials project. Though these lines are for blackleg research and monitoring purposes only, and will not be for commercial production, they are useful for the accurate pathotyping of L. maculans isolates as well as the in-field monitoring of L. maculans populations in Western Canada.
- The ‘Understanding the mechanisms for race-specific and non-specific resistance for effective use of cultivar resistance against blackleg of canola in Western Canada’ research demonstrated that quantitative resistance (also referred to as minor gene resistance or adult plant resistance) is of value in alleviating blackleg impact on canola without the direct involvement of major resistance genes. It also concluded that quantitative resistance traits are highly useful for blackleg management in Western Canada, even with warmer temperatures encountered during rosette to early flowering stages.
Researchers continue to investigate critical blackleg management topics, such as:
- Understanding the critical infection window that causes blackleg of canola in western Canada
- Overcoming blackleg disease in canola through establishment of quantitative resistance
- Improving management of blackleg on canola via better flea beetle control and effective fungicide seed treatment in Western Canada
- Identification and genetic mapping of novel genes for resistance to blackleg in Chinese and Canadian B. napus lines
- Fine-tuning of the blackleg yield loss model in canola
Published May 1, 2023