Key Result
This study has developed a reliable, pheromone-monitoring system that can be used to monitor populations of canola flower midge. It has also determined that both buds/flowers and occasionally pods can be infested by this insect. Although short-lived, the midge emergence is prolonged over the summer when canola plants are susceptible to infestations. Continued monitoring and vigilance for this species is recommended, as its full pest potential is yet to be determined.
Project Summary
Background
The canola flower midge, Contarinia brassicola, is a recently discovered insect pest of canola on the Prairies. Damage results when larvae feed on developing flower buds which prevents the flowers from opening and forming pods. The population density of canola flower midge is difficult to evaluate as densities are variable across the species range and damage is discreet and can be easily overlooked except under outbreak conditions. Midge control using traditional insecticides is complicated by the cryptic feeding nature of larvae. Larvae feed within developing buds and flowers, which minimizes the time and life stages that are susceptible to control.
To develop an integrated pest management program for the canola flower midge, reliable monitoring and forecasting tools are needed to assess midge population densities prior to damage occurring in the field. Synthetic versions of adult produced sex pheromones can be excellent population monitoring tools. This is especially true for species where the adult stages occur before damage in the field, as with canola flower midge. Thus, monitoring adult canola flower midge populations will allow the population density to be determined before damage occurs. Recent work identified the female-produced canola flower midge pheromone and optimized the pheromone blend and dose to create a potent male attractant.

Purpose
Pest monitoring tools are needed across the Prairies to help producers evaluate production risk and increase agricultural and economic sustainability. This project will determine the relationships between the number of canola flower midges captured on pheromone-baited traps and larval population density and damage in the field, creating a tool that producers and agronomists can use to monitor midge populations. This project will also help to determine at what densities midge populations cause economic damage. It will also increase our knowledge of the abiotic factors that affect midge population density. Ultimately, this research, led by Dr. Mori, the 2020-2025 Natural Sciences and Engineering Research Council Industrial Research Chair in Agricultural Entomology, will help to determine the pest status of the canola flower midge, assist in promoting plant health and increase and protect the sustainability of the canola industry in Canada.
Objectives
The goals of this project are to develop a pheromone-monitoring tool for the canola flower midge and to enhance the knowledge of the factors that contribute to its pest status.
The specific objectives are to:
1) Determine the factors effecting pheromone-baited trap capture of male canola flower midge.
2) Determine the relationship between the number of midges captured on pheromone traps, larval density and damage in the field.
3) Determine the abiotic conditions that effect midge emergence and population densities.
4) Investigate the emergence pattern, longevity and oviposition behavior of the canola flower midge.
Results
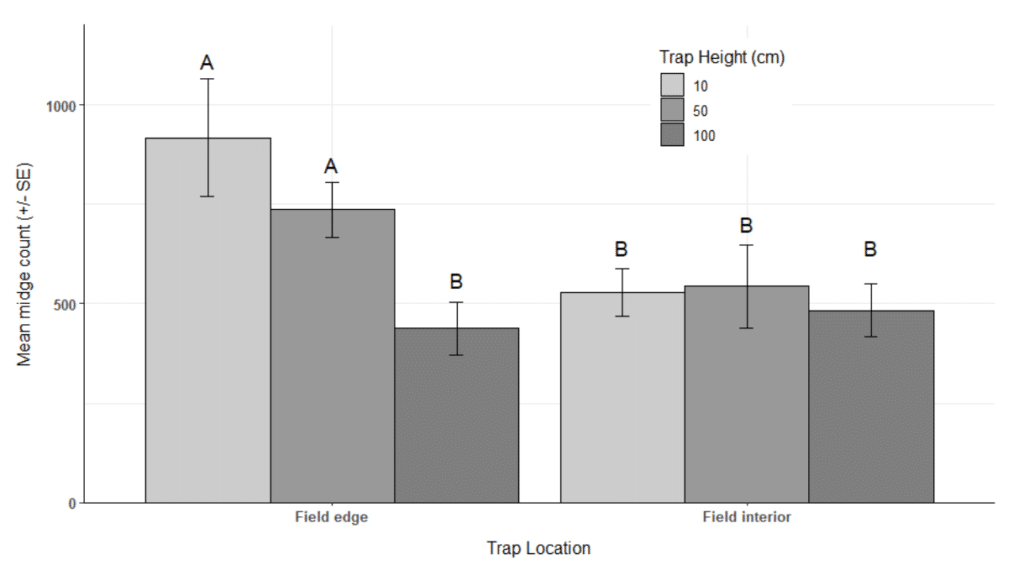
Researchers identified the pheromone-baited Jackson trap, deployed 50 cm above the soil surface along the crop edge, as the optimal combination of trap type and position to effectively monitor the canola flower midge. Male midge activity peaked twice during the day – morning and late-afternoon – which could be used to increase the precision of monitoring strategies or potential treatment applications.
Over the two years of this study, the number of male midges captured on pheromone-baited traps was not predictive of damage in the field. Average air temperature and weekly cumulative rainfall, at the scale measured in this study, did not have an impact on the number of midges captured in either year.
Midge larvae were identified from pods in commercial canola fields and experiments also confirmed canola flower midge larvae can develop in pods, but no alternative host plants were identified.
Although individual midge are short-lived, the midge emergence is prolonged over the summer when canola plants are susceptible to infestations. Researchers urge continued monitoring and vigilance for this species as its full pest potential is yet to be determined.
Conclusions
Overall, this study has developed a reliable pheromone-monitoring tool for C. brassicola and has furthered our understanding of this species’ life history, which is critical as we continue to understand its potential as an agricultural pest.
More specifically:
- A reliable, pheromone-monitoring system that can be used to monitor populations of C. brassicola (pheromone-baited Jackson traps, deployed 50 cm above the soil surface along the crop edge) was developed. Two daily peaks in male midge activity, during the morning and late-afternoon, were observed and could be used to increase the precision of monitoring strategies or potential treatment applications.
- The number of male C. brassicola captured on pheromone-baited traps was not predictive of damage in the field, but in 1 of 2 years there was a significant relationship between the total number of midges captured and the total number of galls sampled over the season, according to data in this project.
- While there wasn’t a large disparity between the sex of emerging adult C. brassicola midges, a higher proportion of those able to live for more than one day are female rather than male. The laboratory midges live for five days or less.
- Meteorological conditions measured in this study did not have an impact on the number of midges captured.
- The environmental conditions and plant host characteristics optimal for C. brassicola oviposition are still not well understood.
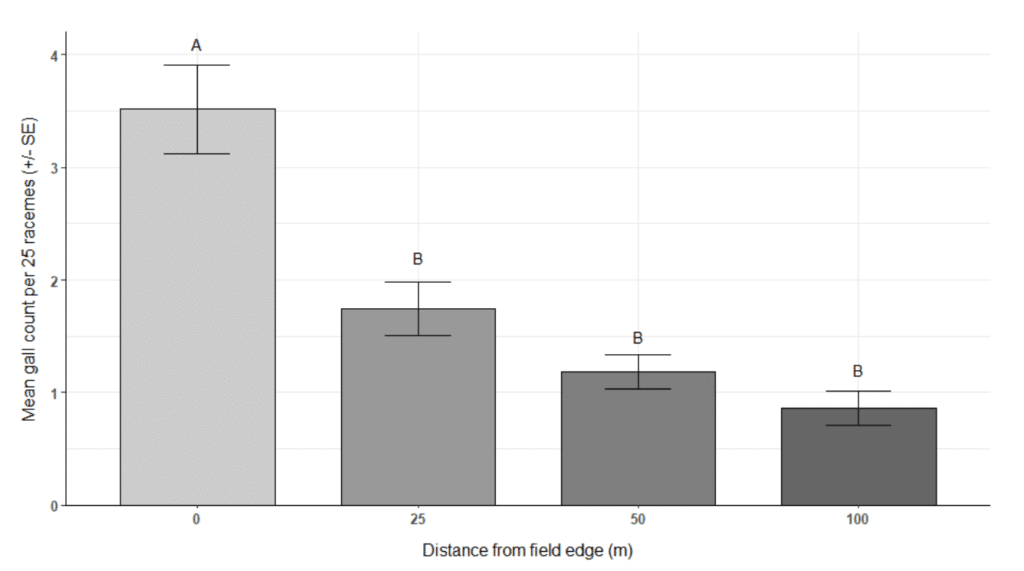
- There was a significant damage edge effect, which will be useful information when designing future management strategies and, if warranted, management could potentially focus on the field edges.
- No alternative hosts were infested over the course of the study; however, under certain conditions C. brassicola are capable of ovipositing and larvae develop in pods.
- Continued monitoring and vigilance for this species is highly recommended, as its full pest potential is yet to be determined.
Future research
Dr. Mori is planning to collaborate with researchers in other parts of Canada and the world to determine if C. brassicola is a native or invasive species. Pheromone traps deployed in southern Sweden in summer 2022 did not capture C. brassicola, but further studies are needed. A controlled insecticide trial to determine the yield loss caused by C. brassicola is also warranted.






