Clubroot is a serious soil-borne disease of cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More plants, such as canola. In canola, it causes swellings or galls to form on the roots, which can ultimately cause premature death of the plant. It is caused by a fungus-like protist called Plasmodiophora brassicae. Currently, there are no economical control measures that can remove this pathogenA disease-causing organism (such as a fungus or bacteria). More from a field once it has become infested. However, it is possible to curtail the spread of the pathogenA disease-causing organism (such as a fungus or bacteria). More and reduce the incidence and severity of the disease. Management of infested fields through the combination of minimizing the risk of soil movement and using a sanitation plan, diligently scouting and record keeping, controlling host weeds and volunteer canola, utilizing clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More (and properly rotating resistance genes), practicing an effective crop rotation, and using patch management techniques for infested areas are the most effective methods of controlling this disease.
Overview
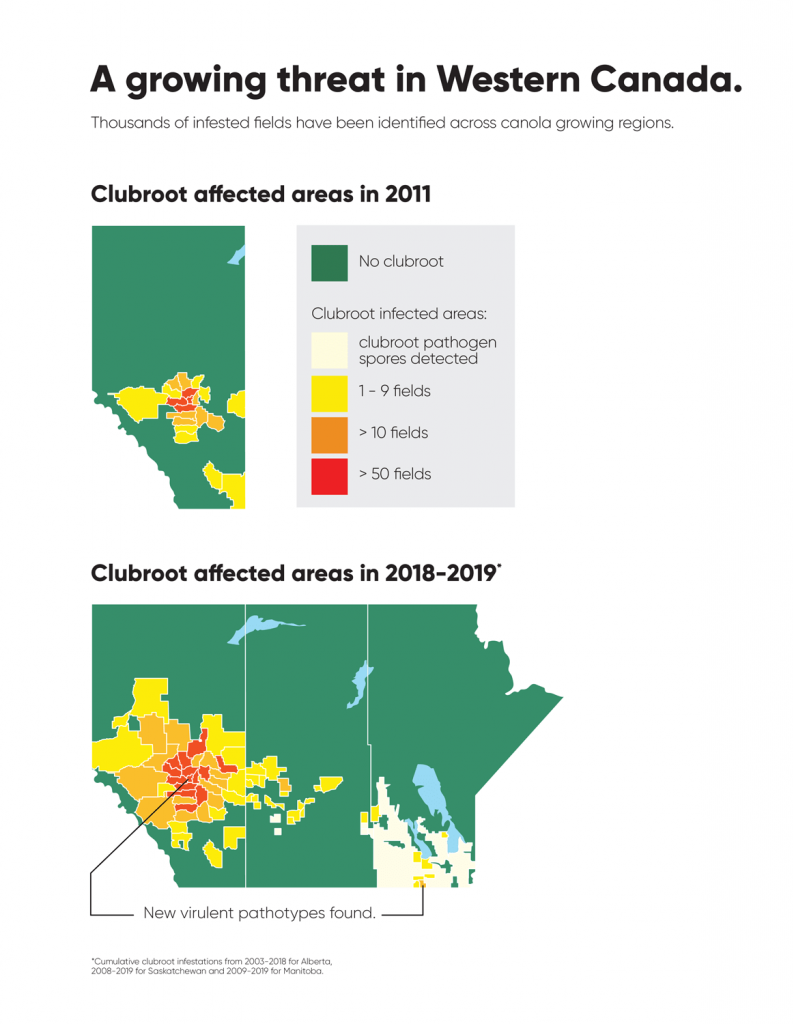
Clubroot had been seen in cole (cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More) crops across Canada before canola; it has been recognized as a problem in Brassica vegetable crops (e.g. broccoli, cabbage, radish, and rutabaga) in Ontario, Quebec, British Columbia, and Atlantic Canada for several years. However, the first report of this disease in a commercial canola field in western Canada occurred near Edmonton, Alberta in 2003. Since then, thousands of infested fields have been identified across canola growing regions in Canada1.
For a thorough overview of clubroot, review the disease cycle and environmental factors impacting it, and then investigate clubroot identification and clubroot testing information. To check out the current distribution, see the affect regions and then learn about how to control and manage the disease, as well as the regulations around the disease and the research on various clubroot topics. Additional contact and resource information is also provided below.
Disease cycle
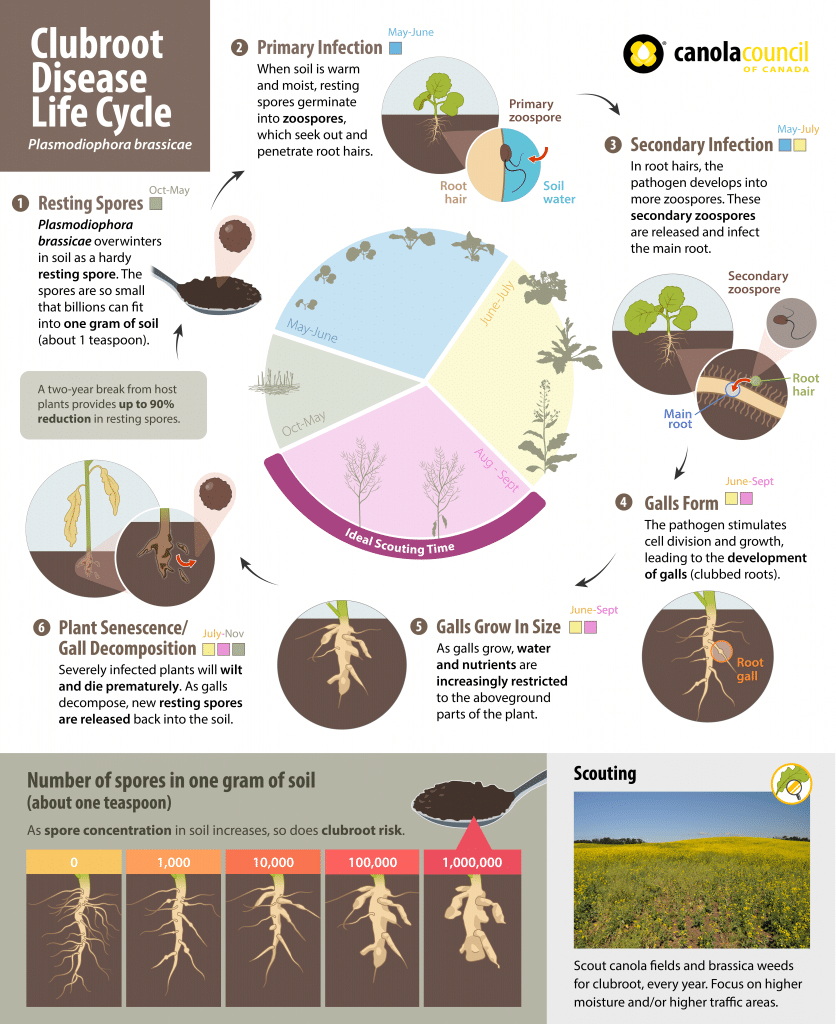
Plasmodiophora brassicae is regarded as a protist, which means it is an organism with plant, animal and fungal characteristics. P. brassicae is an obligate parasite, which means the pathogenA disease-causing organism (such as a fungus or bacteria). More can’t grow and multiply without a living host such as canola or other susceptible crops and weeds.
The pathogenA disease-causing organism (such as a fungus or bacteria). More overwinters in the soil as very hardy resting spores. In the spring, the frequency of resting spore germination is stimulated by secretions from the roots of various plants. Resting spores then germinate and transform into zoospores, which are capable of swimming short distances in water or water-films in the soil and seek out root hairs to infect.
The mobility of these zoospores makes clubroot a very different disease from others in canola. This mobility actually allows the zoosporeSpores which disperse short distances in response to external stimuli. For example, zoospores from the Pythium species (which causes root rots in canola) swim in soil water towards canola seeds and roots, attracted by root exudates. More to seek out potential host plants instead of solely relying on independent distribution methods like wind or rain (as is the case with sclerotinia, blackleg, etc.). The swimming zoosporeSpores which disperse short distances in response to external stimuli. For example, zoospores from the Pythium species (which causes root rots in canola) swim in soil water towards canola seeds and roots, attracted by root exudates. More phase is short-lived.
After initial infection through root hairs or wounds, the zoosporeSpores which disperse short distances in response to external stimuli. For example, zoospores from the Pythium species (which causes root rots in canola) swim in soil water towards canola seeds and roots, attracted by root exudates. More forms an amoeba-like cell. This abnormal cell multiplies and joins with others to form a plasmodium (a naked mass of protoplasm with many nuclei). The plasmodium eventually divides to form many secondary zoospores that are released into the soil after the host root begins to decompose.
These second generation zoospores re-infect the roots of the initial host or nearby plants and are able to invade the cortex (interior) of the root. This is where genetic resistance operates, preventing the invasion from secondary zoospores.
Once in the cortex, the amoeba-like cells multiply or join with others to form a secondary plasmodium. As this plasmodium develops, plant hormones are altered, which causes the infected cortical cells to swell. Clusters of these enlarged cells form clubs or galls.
Some amoeba-like cells are able to move up and down roots through vascular tissue (which transports nutrients and water) of the plant. After secondary plasmodia mature, they divide into many resting spores. When the galls (rapidly) decay by soil microbes, millions of long-living resting spores are left in the soil.
The longevity of these resting spores is a key reason why clubroot is known as a serious disease. The spores can survive in the soil for up to 15-20 years.
However, many of the resting spores appear to become inactive or non-viable after a two-year break from a host crop, indicating the importance of crop rotation, weed control and sanitation to help reduce spore loads in the soil. While this represents a significant decline in inoculum in the field, it is dependent on the number spores present. The more spores that start the infection, the more that will remain over time. This is why it is so important to prevent major infestations from becoming established in a field by using a crop rotation that includes at least a two-year break from canola (and longer under heavy disease pressure). Under severe infestations, which can occur quickly when a susceptible cultivar is grown on infested land, much longer periods without susceptible hosts will be required to reduce inoculum to manageable levels via natural population decline1.
Environmental factors
Warm soil (20-24 degrees Celsius), high soil moisture and acid soil (pH less than 6.5) all favour conditions for infection and increased disease severity (if the pathogenA disease-causing organism (such as a fungus or bacteria). More and a host plant are both present). Soils with a pH over 7.2 tend to inhibit spore germination and disease development, although the disease can still develop. High pH soils will not prevent P. brassicae from arriving in a field, nor prevent the disease from infecting susceptible plants, and nor will it prevent yield loss from occurring. Liming fields to a pH greater than seven will be most successful under low levels of infestation and become less effective under high spore loads in the soil. So liming proactively or when soil infestations are discovered early will help reduce the severity and impact of the disease.
Areas of a field with more soil moisture typically see the most severe infestations (as moisture is needed for the zoospores to swim to root hairs that they can infect). These wet areas are found in depressions, spots with higher clay content, or with subsoil horizons that result in poor water infiltration (such as Gray Wooded or Solonetzic soils).
Symptoms in the field will be dependent on timing1.
Yield loss
Yield loss is dependent on many factors including time of infection, soil moisture and temperature, spore load, soil pH, soil texture, host genotype, pathogenA disease-causing organism (such as a fungus or bacteria). More pathotype, amongst others. An early infection with favorable conditions and moderate to high spore loads can lead to 100 per cent loss, while low spore loads with less favorable conditions may result in little or no yield loss1.
Identify clubroot disease
Symptoms and scouting

The roots of infected canola plants become malformed due to increased cell division and growth, which leads to the development of galls. Clubroot spores then occupy the plant cells and restrict the flow of water and nutrients within the plant, so that severely infected roots can’t transport adequate water and nutrients to aboveground plant tissues. This can result in wilting, reduced seed production, stunting and possibly premature death of the plant.
Since the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More and primary disease symptoms all occur underground, it is crucial to scout fields throughout the season and pull up roots to look for symptoms. Aboveground symptoms may not always be present or may only be present when pathogenA disease-causing organism (such as a fungus or bacteria). More levels are very high. As a result, it is important to always pull plants (carefully, to ensure the root system remains intact) in high clubroot risk areas of the field and look for below ground symptoms.
Clubroot is spread by the movement of soil containing soil-borne resting spores. Soil transport occurs mainly on farm machinery and can occur in both canola and non-canola cropping years. The clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More can be introduced to a field by soil movement in non-canola years and be present at low levels. Minimizing the movement of soil into and out of the field is the best way to prevent the introductions and spread of clubroot.
Clubroot surveys in Alberta have found that many new infestations begin near the field access, which indicates that contaminated equipment is the predominant spread mechanism. Other areas that are at a higher risk of hosting the disease are low spots which often have excess moisture, water runs, high traffic areas, and exploration activity areas or areas that are accessed by outside equipment. In addition, Manitoba fields show that areas where pooling water occurs are at a higher risk for infestation. When monitoring for clubroot, it is important to monitor all high clubroot risk areas in the field. If a field is located in an area where clubroot is not yet known to occur, focusing scouting near the field entrances is a good approach as clubroot infested soil will most likely be introduced via equipment in these areas.

Depending on local conditions and timing of infection, clubroot-infected canola may look very similar to canola suffering from other diseases or environmental stresses. Patches of prematurely ripening canola due to clubroot infection could be confused with other diseases such as sclerotinia, blackleg or fusarium wilt, or moisture stress (e.g. drought or waterlogging) if only viewed from a distance (rather than pulling up and examining the root).
Symptoms in the field will vary depending on the growth stage of the crop when infection occurs, the severity of infection and environmental conditions after the infection takes place. Early infection (at the seedling stage) can later result in wilting, stunting, yellowing and even death of canola plants at the late rosette to early podding stage.
Infection that occurs at later crop stages may not show plant wilting, stunting or yellowing. However, infected plants may ripen prematurely, resulting in shriveled seeds and negatively impacting both yield and quality (e.g. oil content). Lack of moisture, high temperatures late in the season and other canola diseases can also produce similar symptoms (hence the importance of examining the plant below ground as well as aboveground).
Typically, it takes six to eight weeks from initial infection to gall formation, but this depends on temperature as well as when the field receives rain, and can happen as quickly as three weeks after emergence. The best time to scout for clubroot symptoms on roots is late in the season, approximately two weeks before swathing, since root galls should be easy to identify at this time.

Another option is to identify patches of concern while swathing and sample afterwards. Since the entire field is traversed during swathing, this will give the most detailed indication of the incidence in the field. If suspicious plants are not sampled until several weeks after swathing, the root galls may have decayed already, and typical whitish galls will no longer be present. Instead, the decayed galls give roots a brown, peaty appearance rather than the healthy white colour associated with normal roots. If clubroot infection is severe, the plants may have very little healthy root tissue which can result in them being easily pulled from the field. In these areas plants may be up rooted during swathing. If this occurs, it is important to stop and examine the plants in the area from clubroot symptoms.
Clubroot infects all species of plants in the Crucifer (Brassicaecea) family. Weeds such as Shepherd’s purse, stinkweed, flixweed, wild mustard and many others in this family will carry and increase this disease.
Hybridization nodules on canola roots, although rare, could be confused with clubroot galls although these appear as small, round nodules located at root nodes. The interior texture of a clubroot gall is spongy or marbled, while hybridization nodules are uniformly dense inside, like healthy roots. Also, hybridization nodules will not decay rapidly to a peaty or saw dust-like appearance as clubroot galls will1.

Testing
The most reliable way to confirm clubroot is by pulling plants and looking for galls. Soil and plant testing for clubroot DNA is conducted by laboratories, however, understanding the variability in soil is important when interpreting soil sample results. Many labs offer a detection test (PCR) and a quantitative detection test (qPCR). The PCR test will confirm whether or not the pathogenA disease-causing organism (such as a fungus or bacteria). More DNA has been detected in the sample or not, while the qPCR test will provide the number of spores per gram of soil. The extreme variability of clubroot in the soil, the sample size, location(s) of the sample, the number of cores taken for the sample, and handling the sample can all influence the results. Although the quantitative results can be useful information, especially if tracking the increase or decrease of spore loads in a specific spots or getting a feel for the level of infestation, the results need to be understood and it would be reasonable to assume there is extreme spore load variability across the area being tested1.
Soil and plant samples can be tested for the presence of clubroot DNA via PCR methodology. The labs in Canada that currently provide this service are listed below:
20/20 Seed Labs
507 11th Ave
Nisku, AB T9E 7N5
(780) 955-3435
Element
7217 Roper Road NW
Edmonton, AB T6B 3J4
(780) 438-5522
A&L Canada Laboratories Inc.
2136 Jetstream Road
London, ON N5V 3P5
(519) 457-2575
Discovery Seed Labs
450 Melville Street
Saskatoon, SK S7J 4M2
(306) 249-4484
Quantum Genetix
Site 501, Comp 11
RR 5 Station Main
Saskatoon, SK S7K 3J8
(306) 956-2071
Pest Surveillance Initiative (PSI)
5A 1325 Markham Road
Winnipeg, MB R3T 4J6
(204) 813-2171
SGS BioVision
Sherwood Park Laboratory
#310 – 280 Portage Close
Sherwood Park, AB T8H 2R6
1-800-952-5407
Grande Prairie Laboratory
Unit 106, 10136 128 Ave
Grande Prairie, AB, T8V 1E9
1-877-532-8889
Soil sampling (for a clubroot test)
Obtain approximately two to three cups of soil from the top five to 10 centimetres (A horizon), excluding as much surface organic matter as possible. This could either be a composite sample or a location sample. A composite sample is taken in a “W” pattern near the major approach or entrance to the field, in the plot area or other high clubroot risk areas in the field and is typically used for detection purposes (PCR). A location sample is taken from a marked spot in the field (one spot only, identified with GPS or flagged) and not combined with other areas. This test can help with monitoring if spore concentrations are increasing or declining within the location sampled (qPCR)1.
Plant sampling (for a clubroot test)
Obtain roots from suspected plants and place in a plastic, sealable bag (e.g. Ziploc®). These can be fresh or dry roots. If fresh roots are collected, it is important to ship or send samples quickly to avoid rotting of the plant tissue. If available, root tissue samples are preferable to soil samples to test for the presence of the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More.
The PCR test is a reliable method for molecular detection of the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More and will give either a positive or negative result for the presence of clubroot in the sample. However, there is currently no mechanism in place to correlate this test result to the potential for infection in the field or the expected level of yield loss. Research is underway in this area.
If a sample tests positive for the presence of the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More, use caution when extrapolating the results obtained for a particular sample to an entire field.
When sending soil or plant samples to a laboratory for testing, it is important to ensure that they are packages securely to prevent cross-contamination between samples and/or escape of the clubroot infested tissue or soil during transport1.
Testing in Alberta
Producers finding suspicious roots can contact and send samples to one of four commercial labs (20/20 Seed Labs, SGS Biovision, Discovery Seed Labs or Element in Edmonton) for testing.
Testing in Manitoba
As the founding supporter of the Pest Surveillance Initiative (PSI), Manitoba Canola Growers are offering free clubroot testing for their members. Agronomists are welcome to submit tests on a farmer’s behalf. See more details from the Manitoba Canola Growers.
Pest Surveillance Initiative (PSI) Lab
5A-1325 Markham Road
Winnipeg, Manitoba
Canada, R3T 4J6
Testing in Saskatchewan
For visual diagnosis of plants, submit suspicious samples to the Crop Protection Laboratory. To submit, send a complete sample (the entire plant, preferably more than one) along with the proper form with as much information filled out as possible to the Crop Protection Laboratory. This form is available directly from the laboratory, through Saskatchewan Agriculture, or can be found online. Note: when downloading and using the Internet version of the form, four copies of the fully completed form are required, along with the sample, for submission to the laboratory. For more information or to submit a complete sample:
Crop Protection Laboratory
346 McDonald Street
Regina, Saskatchewan
S4N 6P6
(306) 787-8130
For PCR diagnosis, farmers finding suspicious roots can contact and send samples to one of the commercial labs listed above.
Note that information in this section has also be contributed to by the Clubroot Steering Group (which the CCC chairs).
Affected regions
Clubroot is a disease that affects crucifers worldwide. In Canada, clubroot has been detected in vegetable growing regions of British Columbia, Alberta, Manitoba, Quebec, Ontario, and the Atlantic provinces. The first report of clubroot in a commercial canola field came in 2003, and since then clubroot has been found in most canola growing regions of Canada.
Alberta
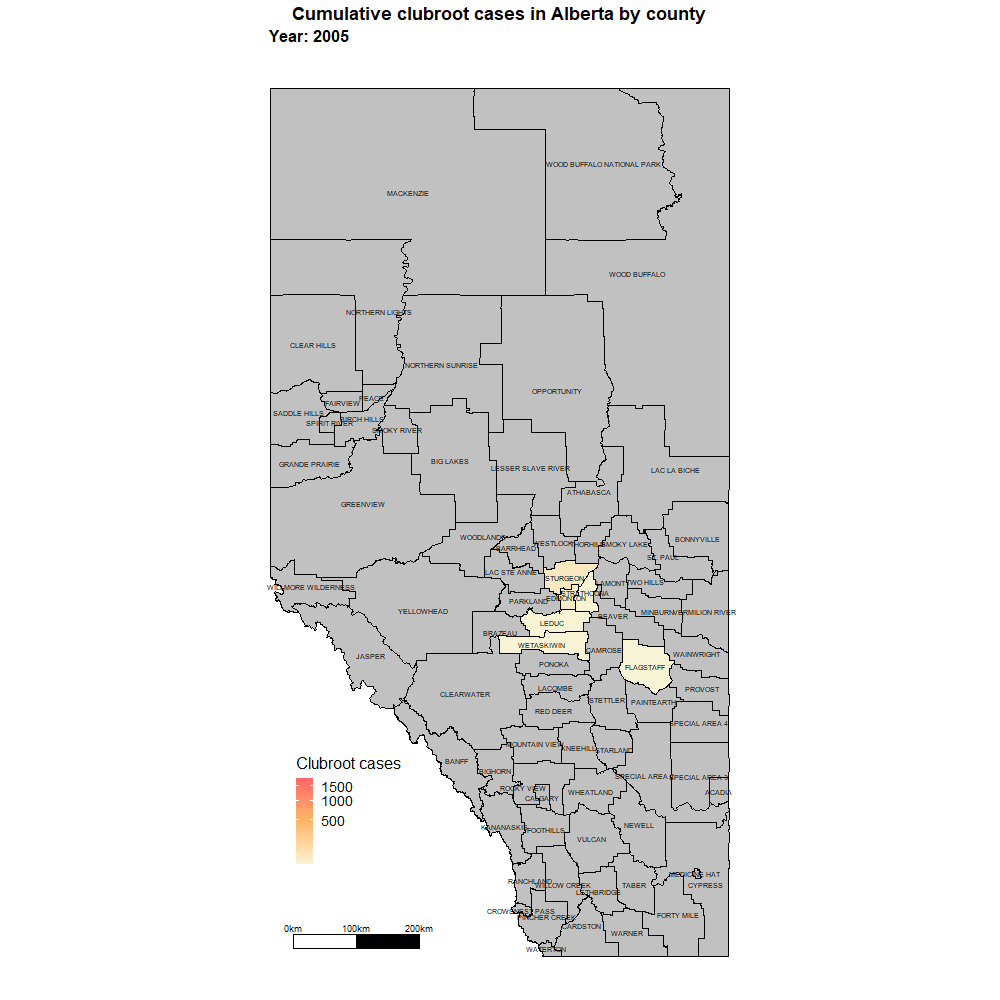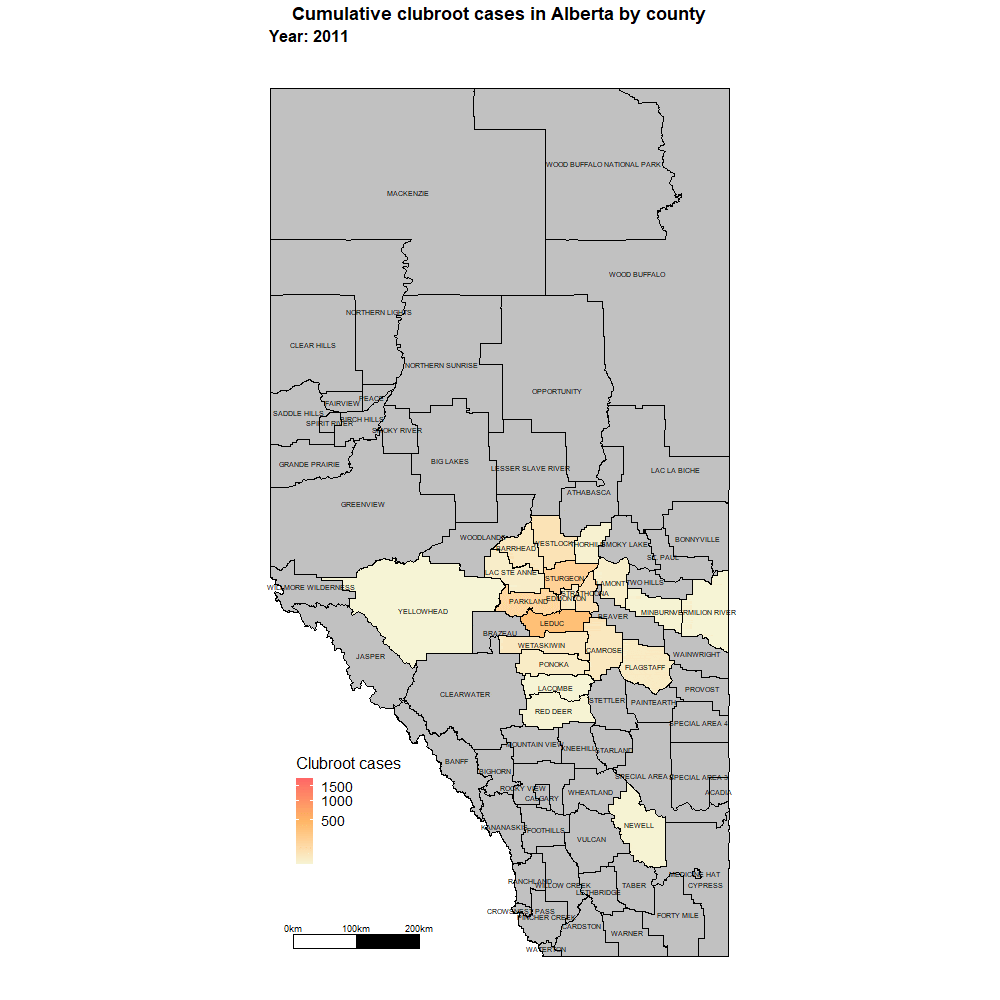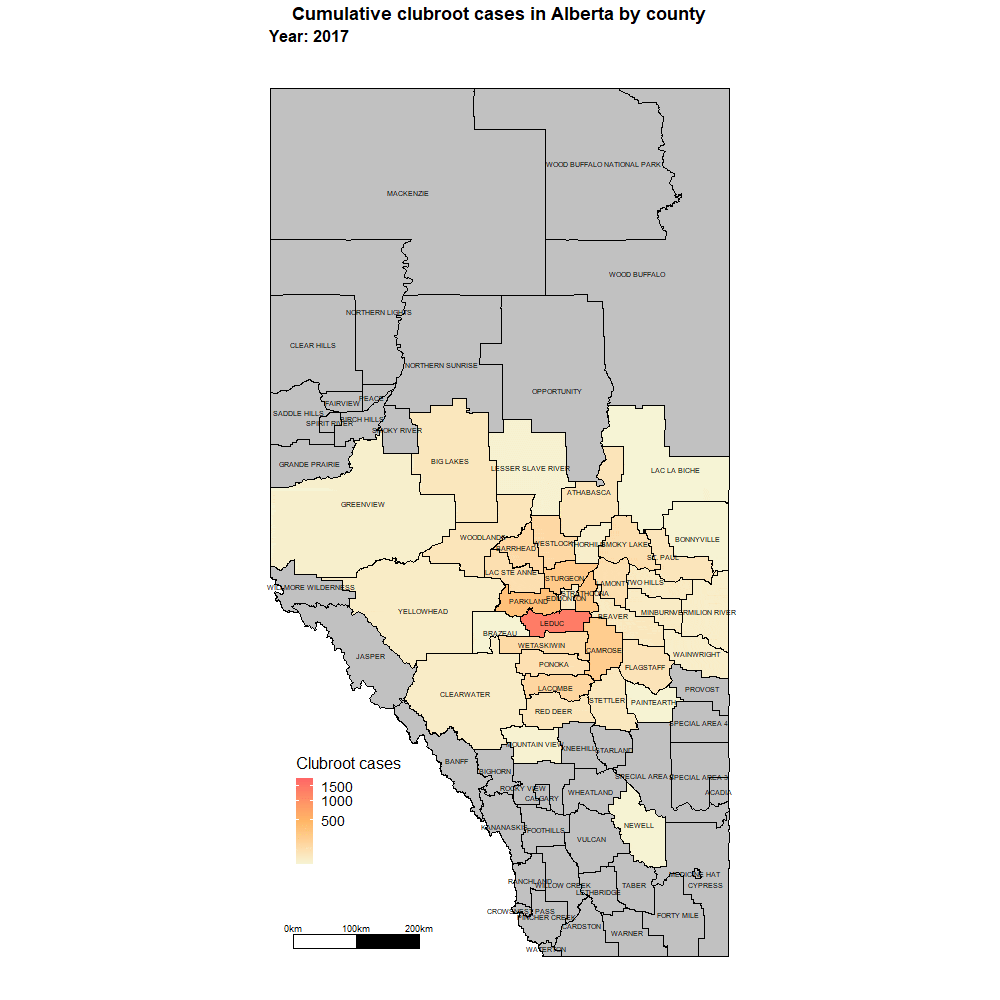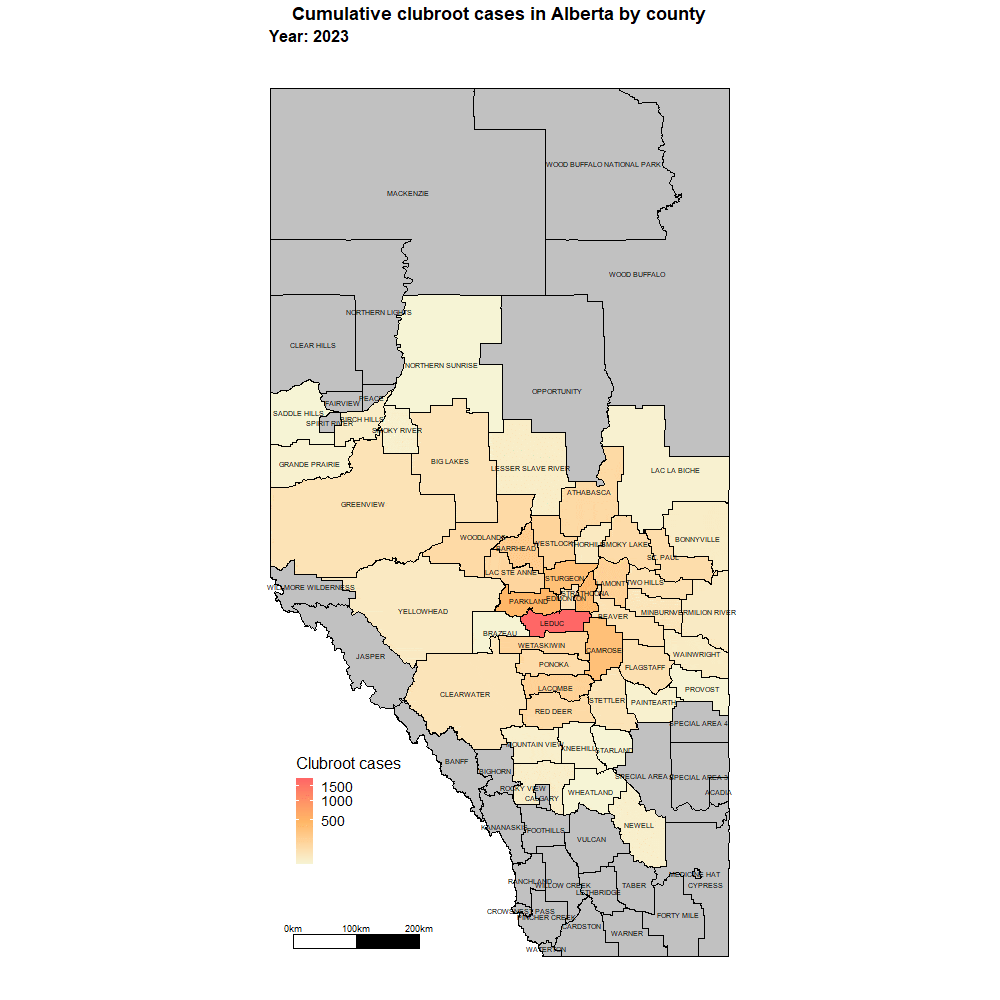
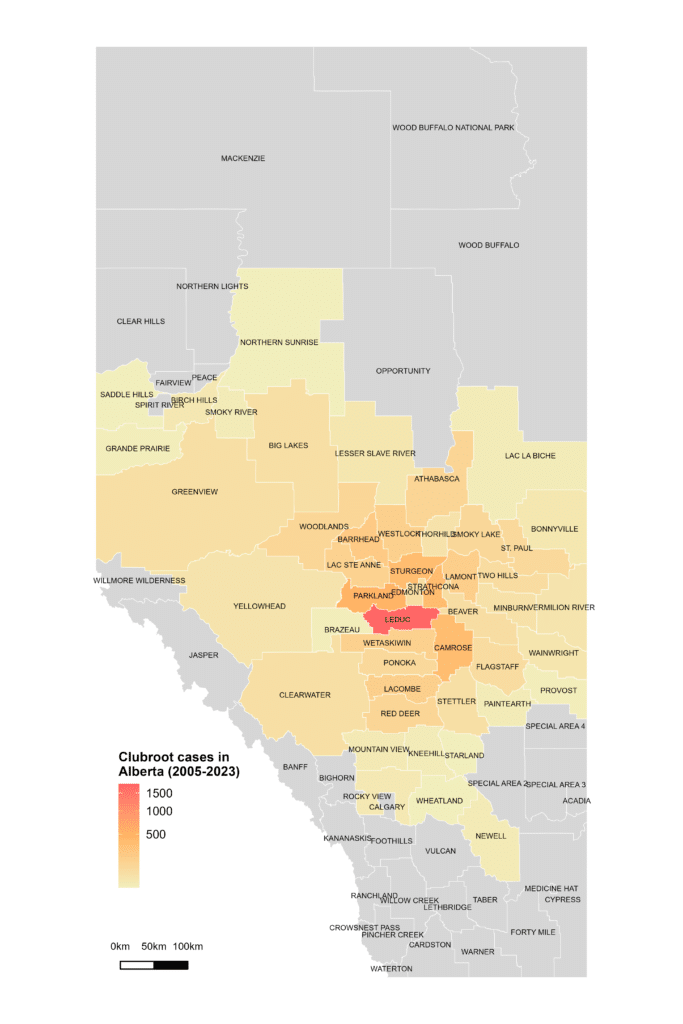
Alberta named the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More (P. brassicae) as a declared pest in the Alberta Pests Act in 2007 and released the first Alberta Clubroot Management Plan in 2008 (revised in 2010 and 2014 and 2019). The November 2018 map of cumulative clubroot infestations in Alberta, which was released by the University of Alberta and Alberta Agriculture and Forestry showed the huge distribution of the disease within the province. In 2020 clubroot had been found in over 3,000 fields across Alberta, with incidence levels ranging from below 30 per cent (low) to above 60 per cent (high). The static map (pictured) shows the cumulative cases of clubroot in Alberta as of January 2024. The disease has been diagnosed in 5,532 canola crops in the province from 2005 to 2023, representing nearly 4,000 individual fields.
For more information on clubroot in specific counties, utilize the ‘Contact information for clubroot specifics in each county’ table.
Contact information for clubroot specifics in each county
Saskatchewan
According the Saskatchewan Ministry of Agriculture, in 2008, the clubroot pathogen’s DNA was first detected in a soil sample collected from one of 30 randomly-selected fields in the clubroot survey. While the pathogenA disease-causing organism (such as a fungus or bacteria). More appeared to be present, the clubroot disease itself was absent, since no infected canola plants were identified at that time. But a subsequent bioassay did confirm that the resting spore concentration was sufficient enough to produce clubroot symptoms in plants. In subsequent clubroot surveys in 2009 and 2010, no further evidence of this disease was found.
In 2011, clubroot was identified in canola plants growing in two fields in the rural municipalities (RMs) of St. Louis and Aberdeen in north central Saskatchewan. Soil tests around these sited confirmed the presence of the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More in the soil. These fields were associated with canola research trials. One more clubroot positive field was identified from the 2012 canola disease survey in the rural municipality of Biggar. This field was randomly selected and a soil sample tested positive for the presence of the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More. A subsequent bioassay further confirmed that resting spore concentrations were high enough to produce symptoms in brassica plants.
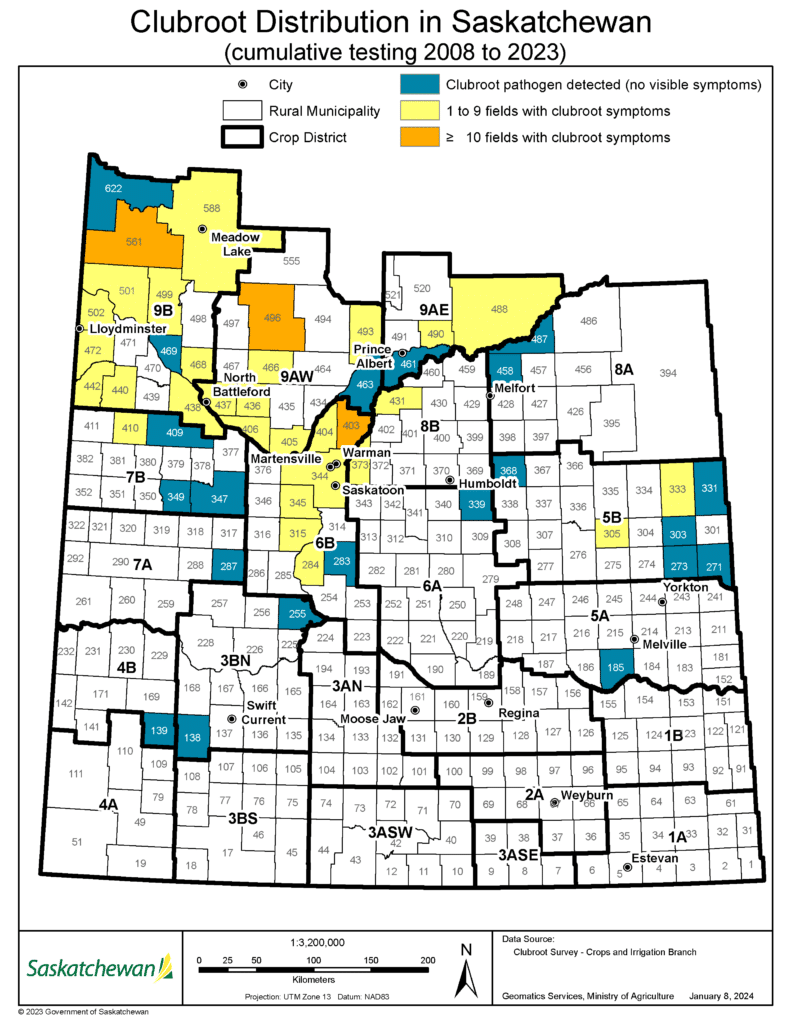
In 2017, clubroot symptoms were identified and clubroot was confirmed in crop districts 9A and 9B, and since has been discovered in other regions of the Province. At the end of 2020, clubroot had been found – either the pathogenA disease-causing organism (such as a fungus or bacteria). More has been detected or visible symptoms have been observed – in 40 municipalities in Saskatchewan.
In 2023, over 500 fields were examined with producers’ permission. One component of the clubroot monitoring program is the soil testing bags available free to producers and industry agronomists. SaskCanola, select RMs, and the Saskatchewan Association of Rural Municipalities (SARM) helped distribute the soil testing bags, with SaskCanola paying for the tests.
No new visible clubroot symptoms were recorded through the clubroot monitoring program in 2023, while the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More was detected in one new field through DNA-based testing. Adding these results to previous years, the total number of commercial fields with visible clubroot symptoms remains at 82. However, the number of commercial fields identified to have the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More through DNA testing rises from 42 to 43.
The Saskatchewan clubroot distribution map2 illustrates the rural municipalities (RMs) where clubroot has been identified since the province started it’s clubroot survey in 2018.
The distribution of the clubroot disease and the clubroot-causing pathogenA disease-causing organism (such as a fungus or bacteria). More in the province and can be used as an estimation of regional clubroot risk. All detections of clubroot and the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More are included in this map, as the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More is long-lived and cannot be eradicated. As a result, areas where clubroot and/or the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More were first identified in Saskatchewan are considered to still have an increased risk and are therefore included in this map illustrating cumulative findings. In addition to illustrating the distribution of fields with visible clubroot symptoms, the map provides information on the number of fields in each RM confirmed to have clubroot visible symptoms.
The information in this map is organized into four main categories that are illustrated by four different colours:
- Blue: The clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More was detected (in soil samples from at least one field in the RM), but there were no visible symptoms.
- When only the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More is detected it means that the pathogenA disease-causing organism (such as a fungus or bacteria). More is present at levels lower than those required to cause disease symptoms under field conditions. When this occurs, producers are encouraged to implement proactive management strategies to keep the pathogenA disease-causing organism (such as a fungus or bacteria). More levels low to prevent symptom development and potential yield losses.
- Yellow: Visible symptoms of clubroot were identified in one to nine fields in the RM.
- Orange: Visible symptoms of clubroot were identified in 10 or more fields in the RM.
- White: The clubroot survey area
The map of Saskatchewan RM with and without Clubroot Bylaws is available here.
Further Saskatchewan clubroot reading is available from the Ministry of Saskatchewan.
Manitoba
According to information provided by Manitoba Agriculture, the presence of the pathogen (Plasmodiophora brassicae) responsible for clubroot has been reported in Manitoba on symptomatic cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More vegetables. The first note dated back to an unconfirmed report on rutabaga in 1925. In the 1980’s, clubroot was found on market garden cruciferousPlants belonging to the family Brassicaceae or (previously referred to as) Cruciferae. More vegetables.
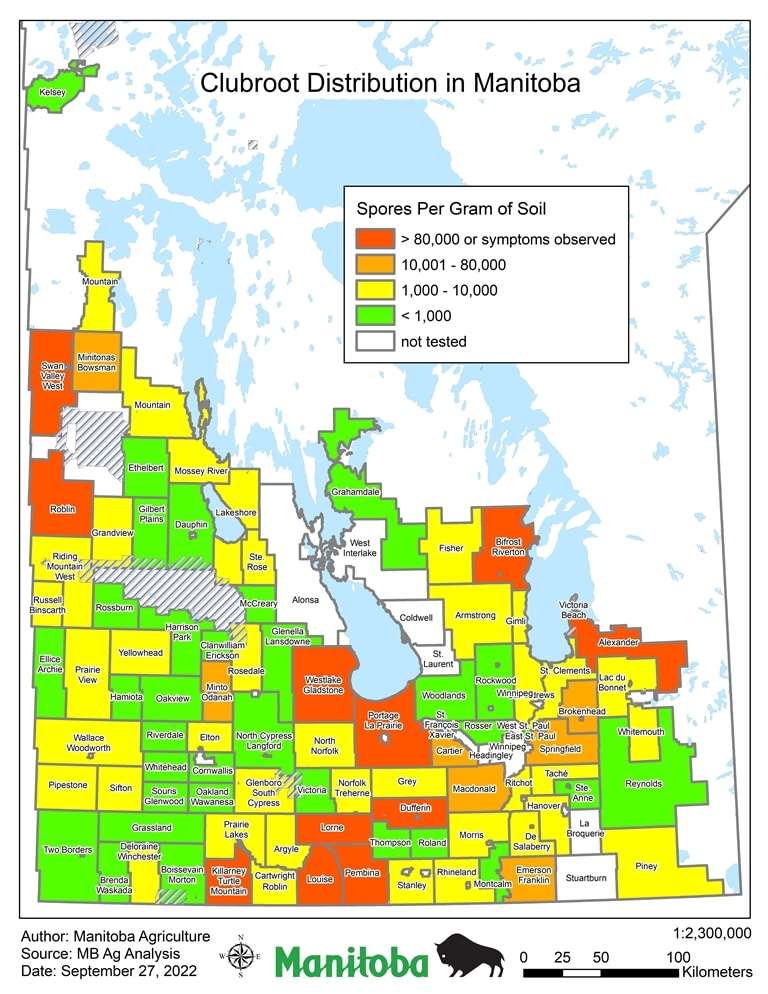
In 2011 and 2012 the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More was detected in soil samples at very low concentrations3. Since then the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More has been detected in more fields and several fields have been discovered showing clubroot symptoms on canola plants4. Manitoba has not declared clubroot a pest under the Manitoba Pest Act. Information on clubroot testing in Manitoba is available from the Pest Surveillance Initiative.
The Manitoba clubroot distribution map (pictured) shows positive clubroot findings by RM, discovered through visual field symptoms and/or presence of clubroot spores in soil through laboratory testing for DNA. Testing completed from 2009 to September 2022 is still considered limited. Positive findings have been at low concentrations, usually below 5,000 spores/gram soil and throughout the province. As more fields are sampled, the map will be updated.
RMs have been highlighted to show the highest concentration of clubroot spores found. This does not mean all fields in the specific RM have these concentrations. As clubroot spore concentrations can be highly variable within a field, the RM classification indicates only the maximum spore concentration found in any field in the RM. Further explanation on the concentration categories are as follows:
Red: Symptoms Observed – Clubroot symptoms have been observed in fields in this RM. Management to prevent continued build-up or spread of this pest should be implemented on farms.
Orange: 10,001 to 80,000 spores per gram soil – Clubroot spores have been found in fields in this RM at concentrations approaching levels known to cause field symptoms. Without management, concentrations may increase to where symptoms are visible in the field within one crop cycle.
Yellow: 1,001 to 10,000 spores per gram soil – Clubroot spores have been found in this RM at low to intermediate levels. With proper management, spore concentrations can decline over time.
Green: 0 to 1,000 spores per gram soil – Clubroot spores have not been detected, or detected at very low levels. At this concentration, clubroot is not a major concern, but fields should be monitored over time. Re-testing is recommended within the next five years to determine if concentrations are increasing.
It is generally reported that 100,000 spores per gram of soil are required to see symptoms under field conditions, but under favourable conditions, lower concentration can cause field symptoms of clubroot.
Annual surveys for clubroot started in 2009, testing 80 – 100 fields annually. Since 2015, soil sampling and laboratory testing increased, as part of expanded efforts to better understand clubroot distribution in Manitoba. From the cumulative survey efforts, as of September 13, 2019, there have been 296 individual fields were found positive for clubroot through soil analysis and 35 fields were confirmed to have symptoms.
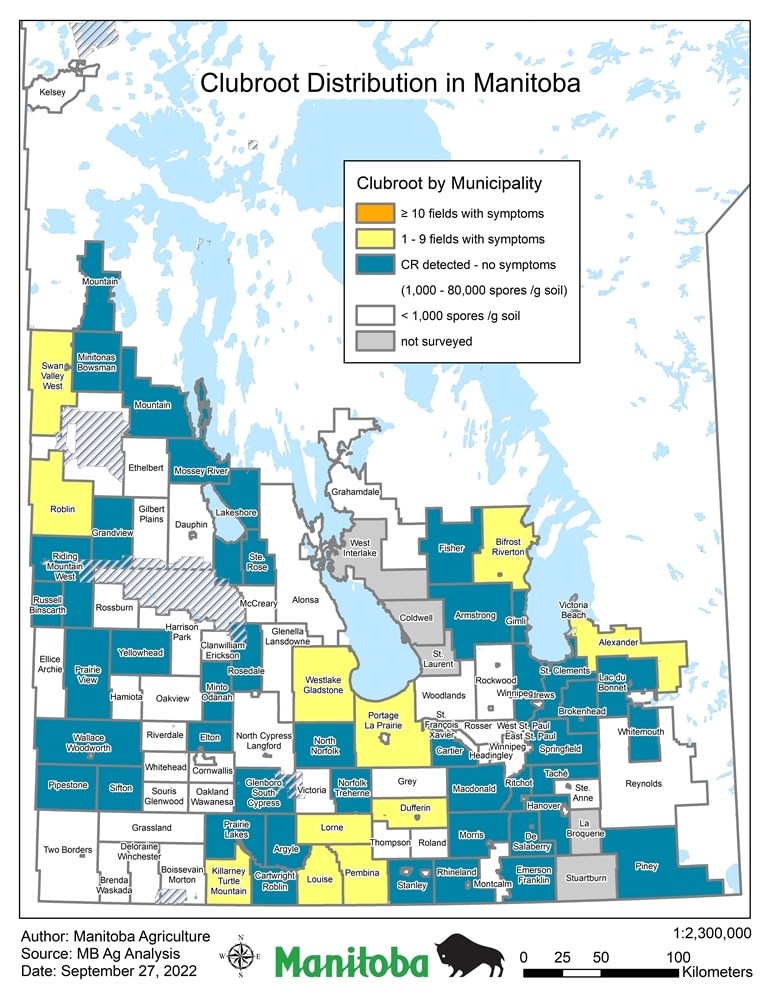
In September 2022 Manitoba Agriculture also released an alternative depiction of clubroot distribution situation in Manitoba. It is based on the same data as the map above, but primarily differs in the number of categories. Another difference is that the alternative map identifies how many fields within a Rural Municipality have had documented symptomatic cases (galls on canola roots), rather than the number of clubroot spores detected in the soil. Thus far, none of the RM’s in Manitoba has had 10 or more fields identified.
This map is intended for those who farm and/or advise growers on either side of the Manitoba-Saskatchewan border. Vigilance and awareness of the clubroot risk in all geographic regions is vital.5
Biosecurity Implications
As less than 10 per cent of farms in Manitoba have been sampled, it is recommended that all fields be tested to determine if clubroot spores are present, regardless of RM classification.
Clubroot Soil Sampling on the Prairies (PDF 254 KB)
Interpreting a Clubroot Soil Test Result (PDF 46 KB)
Clubroot on the Prairies
Credit: Corteva Agriscience
Control Clubroot disease
Prevent clubroot disease
The best way to prevent clubroot in canola is to prevent the introduction of contaminated soil, if at all possible. The pathogenA disease-causing organism (such as a fungus or bacteria). More has several stages in its lifecycle including hardy resting spores, which are released into the soil from infected plant root material, and due to their tiny size, can easily move with soil particles. Therefore, minimizing the risk of soil movement is critical to preventing the introduction of clubroot to a new area (ex. a new field or farm). Any individual who contacts agricultural soil should consider the risks of moving P. brassicae. As an added benefit, equipment sanitation will also help reduce the spread of other diseases, weeds and insects too.
Any vector that moves soil could move this disease, and generally the vectors that move the greatest amount of soil are the greatest risk. Farm equipment, machinery and vehicle traffic can move clubroot resting spores when they move soil, as can wind and water erosion. So by ensuring all incoming traffic is free of soil and ensuring farm operations minimize the movement of soil (ex. by minimizing tillage), the risk of clubroot will be reduced. Talk with stakeholders about sanitation expectations and limitations and agree on a sanitation plan that best manages risk, cost and logistics. Review the Managing Clubroot: Equipment Sanitation Guide for detailed information on sanitation options. It explains the three steps to successful sanitation, including:
- Remove all large, visible chunks of soil (with the help of tools such as a hand scraper, wire brush, etc.).
- Remove smaller soil clumps with high pressure water or air.
- Disinfect by applying a recommended disinfectant product, which includes a two per cent solution of sodium hypochlorite bleach or other recommended products shown to have good efficacy6 (and allow sprayed areas to remain saturated for 20 minutes).
Note that a two per cent concentration of sodium hypochlorite bleach can be made by mixing a 50/50 blend of regular four per cent household bleach and water. Do not buy bleach unless the jug specifies the sodium hypochlorite concentration. For more details on this, see how to make a two per cent sodium hypochlorite solution disinfectant7.
However, just like any tool, it also has its disadvantages. Bleach is corrosive to metals, causes rubber to harden, stains and damages clothing and footwear. Bleach also degrades fairly quickly, so purchase quantities you can use quickly. Discard bleach after the bottle has been open for three months. Check best before dates for unopened bottles.
Two per cent sodium hypochlorite bleach solution can also be used in a boot bath and for misting bottles to sanitize tools. Prior to this disinfection stage, you may want to use other scouting kit tools such as wire brushes, scrapers and disinfectant wet wipes to help clean gear between fields.
Sanitation alternatives to bleach were investigated a recent Alberta Agriculture study. It evaluated 24 different disinfectants and found that while sodium hypochlorite (bleach) was able to kill nearly 100 per cent of the resting spores at concentrations above 1.7 per cent, there were several other disinfectants which were able to kill greater than 90 per cent of the clubroot spores6,((Alberta Agriculture and Forestry (Government of Alberta). 2015. Clubroot Disease of Canola and Mustard. Agri-Facts, Agdex 140/638-1 (Revised September 2015).)). Take a look at the summary of results and products tested here
Since soil can also be moved by means of exploration/construction equipment, recreational vehicles, livestock, manure (clubroot spores are reported to survive through the digestive tracts of livestock), hay or straw bales, seed potatoes (that have soil on them), common (uncleaned and untreated) seed, and even human footwear, these methods may also move this pathogenA disease-causing organism (such as a fungus or bacteria). More.
The amount of soil required to initiate infection in a new field depends on the number of spores in the soil being moved. Heavily infested soil requires significantly less soil to initiate infection than lightly infested soil (and therefore will require the greatest amount of sanitation). As a result, any soil transfer from an infested field should be viewed as a risk.
Effective clubroot prevention can also be proactive. If you are a grower or landowner, ask those coming onto your land about the sanitation measures they use to prevent the spread of clubroot.
Additional prevention and management strategies
In addition to preventing the movement of soil which could contain clubroot resting spores, these strategies may also help prevent or manage clubroot.
- Planting clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More on fields before clubroot gets established is a key management strategy. Although this won’t prevent clubroot from arriving to your field, this strategy relies on the genetic resistance to greatly reduce disease development/establishment compared to susceptible cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More if clubroot is inadvertently introduced to the field.
- Choosing direct seedingA seeding method where the crop is sown into the field without any previous spring tillage operations. More and other soil conservation practices to reduce erosion. Resting spores move readily in soil transported by wind or water erosion and overland flow.
- Utilizing crop rotations. Rotating canola with other crops is a valuable tool that can prevent clubroot resting spores from multiplying quickly once they are in a field. A minimum of a two-year break between clubroot hosts is needed to help reduce clubroot spore levels in soil. Again, longer breaks between susceptible hosts will not prevent clubroot from arriving at a field or new location, and it will not prevent this pathogenA disease-causing organism (such as a fungus or bacteria). More from being spread around a field or being moved to subsequent fields, but it will allow clubroot spores to die off and be more easily managed before another host is grown.
Growers in clubroot-infested areas should grow clubroot-resistant canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and have a break of AT LEAST TWO YEARS between canola crops, in order to prevent clubroot resting spore numbers from increasing in the soil if they arrive1.
Field entry strategy
Crop pests (ex. weed seeds, insects and pathogens causing plant diseases) can easily be transferred from field to field and cause significant consequences if no precautions are taken. Clubroot, for example, is a very serious soil-borne disease that can affect many agricultural stakeholders (ex. farmers, landowners and agronomists), oil and gas industry members, and anyone in rural landscapes if they contact contaminated soil or plant material (and potentially contaminated , runoff water). Therefore measures should be taken to prevent the unintended transfer of any crop pests into or out of any fields by an individual, their tools, implements or vehicles.
The document below outlines the proper stewardship measures that should be followed by individuals who are requested or required to enter fields, in order to minimize the transmission of crop pests into or out of a specific field.
Manage clubroot disease
Managing clubroot after plant establishment in a canola field is difficult. Many commercial cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with clubroot resistance are available and provide high levels of clubroot control when used in a proper rotation. It is important to note, however, that various strains of the pathogenA disease-causing organism (such as a fungus or bacteria). More exist in field populations, so clubroot resistance may become less effective with continued use, especially when spore loads are high. Therefore, canola growers are encouraged to use multiple tools such as rotation, sanitation, biosecurity planning (including the use of separate field entrances and exits), patch management, etc., to manage clubroot, as this will give the best long-term control.
Clubroot management factsheets
These three factsheets (L-R: manage existing clubroot areas, clubroot prevention, and general clubroot management messaging) distill the key practices to mitigate clubroot and maintain profitability, including growing clubroot-resistant (CR) cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More responsibly on all canola acres.
When combined, like ingredients in a recipe, the following practices will help growers to limit clubroot damage in canola, allowing for the continued success and profitability of canola production across the Prairies.
Recipe for clubroot management
- Vigilantly scout all canola fields (early) for symptoms, even if growing a clubroot-resistant cultivar. Symptoms of the disease are most noticeable late in the season, and can still be seen during and after harvest on plant roots. Producers are strongly encouraged to familiarize themselves with clubroot symptoms and start scouting now before the galls degrade. If galls are degrading they will look brown and peaty and may fall off when plants are pulled from the ground. Scout heavy traffic areas (entrances, exploration, gas wells, etc.), low areas/water runs and areas where dust/snow tends to drift and settle. It is good practice to pull plants and examine roots whenever you are looking at later-stage canola as the disease could show up anywhere.
- Keep a minimum two-year break between canola crops. This crop rotation is crucial in the stewardship of genetic resistance. With a two-year break between clubroot hosts, we see a rapid decline in living resting spores. Longer breaks may be required when spore loads are high.
- Seed clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More and understand if/when to deploy different sources of clubroot resistance. Planting clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More before the disease arrives and gets established will give you a better chance of keeping the resting spore load at a manageable level and maintaining effective resistance. Rotation of resistance genes could also be important to maintain resistance efficacy. With repeated use of cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with the same resistant traits under high spore loads, virulentA form which causes infection (and can result in a disease) More races can multiply and effectiveness of resistant will be seriously compromised.
- Limit activities that can introduce foreign soil or cause erosion. Minimum tillage and equipment sanitation (as simple as knocking off visible dirt before leaving a field) will greatly reduce the risk of moving infested soil around. Note that wet soil conditions increase the amount of soil that clings to equipment.
- Control host weeds. Common weeds that can host clubroot include stinkweed, shepherd’s purse, flixweed, all mustards and volunteer canola. They need to be controlled within three weeks of emergence to prevent a new batch of spores being produced.
- Isolate field entrances and hot spotsAreas within bulk (seed) that have a higher temperature than the surrounding material (ex. in stored canola). More. Use patch management strategies to reduce spore loads, such as grassing the affected area, which will also limit soil movement. Patches that are visibly worse than the remainder of your field often have billions more spores per gram of soil than elsewhere and are often the first place where clubroot resistance breaks down. Removing these hot spotsAreas within bulk (seed) that have a higher temperature than the surrounding material (ex. in stored canola). More from cultivation for a few extra years significantly reduces spread and risk of resistance breakdown. As part of this, having separate field entrances and exits could reduce the amount of infested soil leaving the field on machinery.
Researchers continue to investigate new management tools and management strategies, such as liming and bait crops and patch management strategies.
Terminology of genetic resistance and loss of resistance
Plant disease resistance is a term used to describe the reduction of pathogenA disease-causing organism (such as a fungus or bacteria). More growth on plants to minimize damage from the pathogenA disease-causing organism (such as a fungus or bacteria). More and potential yield loss. Qualitative resistance comes from a major gene or genes and provides a clear benefit when it matches the pathogenA disease-causing organism (such as a fungus or bacteria). More raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More present. Quantitative resistance can come from many genes and is often considered a baseline of protection from major losses. Disease can still occur with resistance but resistance traits should mean the plant has lower disease severity and yield loss than plants that are susceptible.
So how can we lose the effectiveness of a resistance trait? For example, how does a clubroot-resistant cultivar work one year and not the next?
Resistance breakdown occurs because of:
Natural selection. One clubroot gall produces millions to billions of resting spores and that population of spores can contain multiple Plasmodiophora brassicae pathotypes. However, one or two pathotypes tend to be dominant across a field, with other pathotypes present only at low levels. First generation clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More are resistant to the most common pathotypes across the Prairies. Natural selection can occur with repeated use of the same type of resistance, which will keep the common pathotypes from increasing but opens the door for rapid increases of some less common pathotypes that were present in the field at low levels. Over time, these less common pathotypes could become the new dominant pathotypes in the population. This is the selection pressure that makes natural selection work on clubroot. Through repeated use of the same resistance sources (especially in short rotations), you select for the pathotypes that can overcome the resistance used.
Mutation. This refers to a permanent change in the DNA (nucleotide sequence) of an organism, which can be passed on to its progeny. Mutations can occur randomly, and while most are not advantageous (and are often harmful), occasionally a random mutation confers some advantage to the organism in question. For example, a mutation in a P. brassicae could result in a brand new pathotype developing which can cause infection in a resistant canola cultivar. Mutations are one way that diversity can be introduced into a pathogenA disease-causing organism (such as a fungus or bacteria). More population, but mutations like this are rare and slow.
Recombination. This refers to the exchange of genes during sexual reproduction (chromosomal crossover) that leads to the offspring having different gene combinations from the parents. This can also add diversity to the pathogenA disease-causing organism (such as a fungus or bacteria). More population and result in the emergence of new strains. While recombination is possible with other diseases, it is still uncertain whether or not it occurs with P. brassicae.
Other common terms used in a discussion on pathogens and resistance:
RaceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More/strain/pathotype. These terms are often used interchangeably in common language and in general refer to subgroups of a pathogenA disease-causing organism (such as a fungus or bacteria). More that can be distinguished based on virulence, symptom expression or host range, but not by morphological features. Usually “raceA specific term that describes pathotypes or strains which differ in their impact on specific resistance genes of a host. It is more specific than strain or pathotype. More” is the most precise of these definitions, and often specifically implies the presence or absence of specific avirulence genes in the pathogenA disease-causing organism (such as a fungus or bacteria). More that interact with matching resistance genes in the host (so they’re often based on the clearly defined genetics of the interaction). “Pathotype” is a somewhat looser term, since these are often distinguished based on their virulence (ability to cause disease) and/or the type of symptoms they cause, while the molecular or genetic basis of the interaction may not be known. Finally, “strain” is the loosest of the terms, implying groups that are different in some way (e.g. virulence or some other parameter) but which are not always defined by any agreed upon system (for example: we recognized that there were multiple strains of P. brassicae, but needed to develop the Canadian clubroot differential (CCD) set to classify them into pathotypes).
Isolate. This is a subpopulation of a pathogenA disease-causing organism (such as a fungus or bacteria). More or other microorganism that is separated from its parent population (for example, by collecting a single-spore or single-gall) and maintained under controlled conditions.
Genotype and phenotype. The genotype is the set of genes responsible for particular trait – the genetics present. Phenotype is the physical expression of a trait – what it looks like in the field this year.
PathogenA disease-causing organism (such as a fungus or bacteria). More. This refers to the agent that causes a disease. Sometimes use of the terms “pathogenA disease-causing organism (such as a fungus or bacteria). More” and “disease” are confused. The pathogenA disease-causing organism (such as a fungus or bacteria). More causes the disease. The disease is the result of the pathogenA disease-causing organism (such as a fungus or bacteria). More, host and conducive environment coming together. For example, P. brassicae is the pathogenA disease-causing organism (such as a fungus or bacteria). More that causes clubroot. So that means that P. brassicae is what infects canola. Clubroot is the name of the disease that results from infection1.
Utilizing genetic resistance
Each time a susceptible host is grown in the presence of the disease, clubroot spores multiply, making management more difficult. Pathotype 3H, the predominant pathotype on the Prairies, is very well controlled by the genetics available in all current cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Screening of field clubroot populations show that there are many other pathotypes present as well and this diversity in pathotypes allows the pathogenA disease-causing organism (such as a fungus or bacteria). More to persist by favouring development of those pathotypes not controlled by resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More. Managing new genetic tools properly through appropriate crop rotations and agronomic practices will be necessary to preserve their effectiveness against clubroot, please see your seed rep or agronomist for further information.
Growers using clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More in clubroot-infested fields may experience some infected plants, which can be attributed to susceptible volunteers and off-types. Volunteer canola seed can germinate many years after it was last grown, and if this comes from a susceptible canola crop, then all the volunteers will be susceptible, while only a percentage of volunteers from resistant seed will carry resistance. Off-types are a normal part of hybrid canola production – no canola hybrid is 100 per cent pure, so there may be a small amount of the seed that didn’t receive the resistant gene during breeding (one to 20 per cent).
When scouting for clubroot in a field of a clubroot-resistant cultivar, look at the distribution of the infected plants. If there are patches or clusters of infected plants, this is more likely caused by the presence of pathotypes that have overcome the resistance. A random scattering of infected plants would more likely be a result of off-types or volunteers1.

Understanding clubroot resistance
Clubroot resistant canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More are key tools used to delay clubroot establishment and manage clubroot disease on the farm. However, to prevent rapid genetic shifts in clubroot populations and subsequent loss of effective resistance in clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, this valuable resource must be used judiciously in an integrated management approach, which includes diligent scouting, practicing a diverse crop rotation with at least a two-year break between canola crops, effectively managing weeds, sanitizing equipment and minimizing soil movement.
Resistance classification system
Clubroot resistance in a cultivar should be substantiated through standard testing procedures outlined in the Western Canada Canola/Rapeseed Recommending Committee (WCC/RRC) guidelines and protocols. CultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More are compared to the susceptible check cultivar for clubroot infection and are assigned either resistant (R), intermediate (I) or susceptible (S) ratings.
Clubroot resistance classifications
Clubroot resistance classifications
Clubroot pathotypes
A base resistance (R) label requires that the cultivar is resistant to the predominant clubroot strains or pathotypes in Western Canada. Additional ratings can be appended to the base (R) label to describe resistance to specific uncommon or new pathotypes.
As clubroot populations in infested fields become more diverse over time, and more clubroot-resistant resistance genes are bred into canola cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More, the usefulness of rotating clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More with different resistance will increase. Currently there are no tests commercially available for growers to distinguish or detect new virulentA form which causes infection (and can result in a disease) More strains in their infested fields, but genetic markers (which will become another management tool) are being worked on for quick identification of these pathotypes.
Careful scouting to detect early infestations to alert growers and deploy resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More is of utmost importance. Waiting to use resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More until significant infestations have developed will only create high soil spore loads and increase the probability for pathogenA disease-causing organism (such as a fungus or bacteria). More shifts which can rapidly defeat cultivar resistance (and the use of resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More before clubroot arrives won’t reduce the effectiveness of the resistance if it does arrive).
A list of current clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More (as of October 2023)
| BrettYoung |
| BY 5125CL |
| BY 6204TF |
| BY 6207TF* |
| BY 6214TF* |
| BY 6216TF* |
| BY 6217TF* |
| BY 7102LL* |
| Brevant (Corteva) |
| 1028 RR |
| 2028 CL |
| B1030N |
| B2030MN |
| B3010M* |
| B3011 |
| D3157C |
| D3158CM |
| B3012 |
| B3014 |
| B3016* |
| B3017N* |
| B4015* |
| Canterra |
| CS2600 CR-T* |
| CS2700 CL* |
| CS2800 CL* |
| CS3000 TF* |
| CS3100 TF* |
| CS4000 LL |
| CROPLAN (WinField United) |
| CP20R3C* |
| CP22T1C* |
| CP213C |
| DeKalb (Bayer) |
| DKLL 84 CRSC |
| DKTF 97 CRSC |
| DKTF 98 CR* |
| DKTFLL 22 CRSC |
| DK400TL |
| DK900TF |
| DK901TF* |
| DK902TF* |
| DK903TF |
| InVigor (BASF) |
| L234PC* |
| L241C |
| L255PC |
| L258HPC |
| L340PC |
| L343PC* |
| L345PC |
| L350PC |
| L356PC |
| L358HPC |
| L359HPC |
| LR354PC |
| Pioneer (Corteva) |
| 44H44 |
| 45H42 |
| 45CM39* |
| 45CS40 |
| P501L |
| P505MSL |
| P506ML |
| P509L |
| P514CL* |
| P612L* |
| Proven (Nutrien) |
| PV 581 GC |
| PV 660 LCM |
| PV 661 LCM* |
| PV 680 LC |
| PV 681 LC |
| PV 780 TC |
| PV 781 TCM* |
| PV 881 OCM* |
| PV 280 CLC* |
| Victory (Cargill) |
| V25-3T |
| V25-5T |
*Contains clubroot resistance trait different from or in addition to 1st generation/Mendel.
Consult seed representatives for more details.
Crop rotation
Crop rotation is a valuable tool to help manage clubroot spore loads. A minimum two-year break between canola crops (and other hosts) will help reduce clubroot spore levels in soil, with over 90 per cent of the spores not being viable after a two year break. A long rotation between susceptible hosts (crops and weeds) will not prevent a field from having clubroot resting spores introduced to a new field or location, but it will allow clubroot spore numbers to decline before the next host crop is grown. If clubroot is undetected for a period of time, spore loads may have increased to levels which a two year break won’t be enough1.
Control host weeds
Controlling host weeds in all crops is very important to prevent the further increase in spores. Weeds should be controlled within three weeks of emergence so they aren’t able to produce new resting spores. Known clubroot hosts include: canola volunteers, Brassica weeds (ex. flixweed, stinkweed, shepherds purse, wild mustard), tame mustard, camelina, and other Brassica crops and vegetables1.
Reduce tillage
There has been one report from Norway of lower clubroot severity under reduced tillage. Reduced tillage or direct seedingA seeding method where the crop is sown into the field without any previous spring tillage operations. More also may help combat a clubroot infestation in Canada. Fewer tillage operations will help prevent movement of contaminated soil within a field and between fields. In some instances, fields with the largest affected areas have been fields where intensive tillage has been used1.
Fungicides
Currently there are no registered fungicides for clubroot control or suppression in canola. Although there are fungicides registered for clubroot control in cole crops around the world, the relatively high cost and application method (transplant bed drench or broadcast incorporation) make them uneconomical for canola on a field scale. Calcium cyanamide, an old form of nitrogen fertilizer with fungicidal properties, has shown promise for reducing clubroot in cole crops. However, high application rates, significant cost, and limited availability make it a poor option for canola.
The effectiveness of seed treatments for managing traces of clubroot on seed surfaces is currently being explored.
Liming
Liming of fields and patches has shown significant promise, due to its impact on soil pH. Increasing soil pH in areas with clubroot can reduce clubroot severity. Brassica vegetable growers around the world have been using lime for decades for this purpose. The increase in pH (on acidic soils) could have the added bonus of increasing availability of key nutrients, although the amount of lime (and pH boost) required to provide the nutrient benefit could be quite a bit less than the amount required to reduce the clubroot risk.
New research projects will give farmers and agronomists more detail on best lime sources, appropriate application rates and timing, and possible limitations of lime to bring very low pH soils up to a low-risk level. On this last point, lime might not be able to push past a soil’s “buffer pH” unless applied rates are very high. As it is, it can take a few tonnes of lime per acre to move soil up just one pH point.
Nicole Fox, Master’s student in plant pathology at the University of Alberta, leads one of the lime research projects. She has reported that low pH, clubroot-infested soil treated with hydrated lime significantly reduced the number and size of galls on the susceptible canola roots. She has also noted that liming can reduce the selection pressure on the current resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More planted, as well as decreasing the amount of spores reproducing in susceptible volunteer canola and even crucifer weed species present in the field throughout the crop rotations. However, types of lime, application style and timing will still need more research.
Targeting small early-identified patches is preferred because trucking the required rates can be expensive. When considering a lime source, look for a high concentration of calcium carbonate (CaCO3) or hydrated lime (Ca(OH)2) per tonne and consider purity of the source. Large-grit lime could take years to be fully effective but it is much easier to handle and apply. Powdered lime acts fast but is hard to handle and spread in large volumes. On application style, lime is often roto-tilled into the soil in research plots, but this tillage will add to the spread of clubroot if you aren’t careful. Ultimately, growers could benefit from a reduced-tillage or surface-applied liming technique, when done properly.
Bait crops
A new management technique that is being investigated is the use of bait crops. In a bait crop situation, plants that are susceptible to clubroot are allowed to grow for approximately four to five weeks to stimulate germination of the clubroot resting spores. The susceptible crop is then ploughed down and/or sprayed (the best method is still being investigated) before the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More completes its lifecycle, which prevents the addition of more resting spores to the soil. This strategy helps draw down the population of resting spores in that field, which may shorten the time needed between plantings of a commercial host crop. Although recent research in Alberta indicated that bait crops are not useful in severe infestations (the bait crops had no effect on clubroot severity in two highly infested field sites in Alberta), more research is needed on their impact under lighter infestations. One of the risks with bait crops is the unpredictability of weather conditions when the bait crop needs to be sprayed (at the proper spray timing), in order to effectively terminate the host plants before they are able to propagate clubroot.
Patch management
Often clubroot appears in small patches and these patches increase in size with the movement of soil outward. If caught early, these patches can be isolated and treated separate from the field. Grassing these patches and even pulling infected plants from these areas will help diminish spore loads and keep soil from moving. Read more patch management ideas in this Canola Watch article.
Patch management with liming is another tool to consider. Further investigation continues, but liming to a pH of 7.2 or above has been shown to reduce gall formation in some cases8,((Campbell, R.N., and Grethead, A.S. 1989. Control of clubroot of brassicas by liming. In: A.W. Engelhard (ed): Soilborne pathogens: management of disease with macro- and microelements. APS Press, St. Paul, MN. 90-101.)).
Grassing field entrances and other higher-risk areas
There are many benefits to grassing in areas for clubroot management. Clubroot is often detected at the field entrance as it is: a high where traffic area, an area where equipment is often parked, where seeding and tillage operations begin, and where compaction (often resulting in lack of crop growth) causes excessive moisture. For all these reasons, this is a good spot to turn into a grassed area.
Seeding entrances to grass can result in these benefits:
- Providing an area to clean off equipment before leaving the field
- If 90 per cent of the soil can be removed from equipment, 90 per cent of the risk of moving clubroot has also been removed.
- Keeping the soil in place and reducing the chance of moving clubroot spores in the soil (if present)
- The tiny clubroot spores can be moved significant distances within a field from tillage, seeding equipment, tires, animals, and even by wind dispersal/erosion. Grass will keep the soil in place and by choosing grasses suitable for the environment, plant growth will reduce opportunities for excessive moisture.
- Speeding up the reduction of viable spores in the soil
- Research has shown that seeding clubroot-infested soils to certain grasses (including Fiesta Perennial ryegrass, common Smooth bromegrass and Fleet Meadow bromegrass) will significantly speed up the rate of spore decline in the soil. Some grasses appear to coax spores out of dormancy and have shown to increase the spore reduction by greater than 90 per cent within a year. The grasses that can’t provide this added benefit will still provide the other benefits listed above (ex. keeping soil in place).
If clubroot patches are found in other areas of the field (in addition to the entrances), consider grassing in these areas too. Clubroot found in other patches other than the entrance can also be seeded to grass, if necessary. This could include water-runs, low spots, or random patches within the field. Similar to entrances, water-runs, very low spots and compacted areas with excessive moisture are also high risk areas which don’t produce a great return on investment. Grassing such areas may help reduce the chances of spreading clubroot without much loss of production. These areas can be cultivated and cropped again once spore levels have declined to manageable levels.
Regulation
Alberta
Information adapted from the Alberta Clubroot Management Plan, May 2010 Revision:
Clubroot was added as a declared pest to Alberta’s Agricultural Pests Act (APA) in April 2007. The APA is the legislative authority for enforcement of control measures for declared pests in Alberta. The Minister of Alberta Agriculture and Food is responsible for this Act. Since county policies may continue to be modified and updated periodically, it is also recommended to contact your local Ag Service Board or Agricultural Fieldman to ensure you are aware of the content in the latest version of their policy.
However, enforcement of pest control measures is the responsibility of the municipal authority, and Agricultural Fieldmen are responsible for enforcing pest control measures in their municipalities. Pest inspectors have the power to enter land at a reasonable hour, without permission, to inspect for clubroot and collect samples. The owner or occupant of the land has responsibility for taking measures to prevent the establishment and spread of clubroot. Alberta’s Clubroot Management Plan outlines management and sanitation measures to prevent the spread of clubroot from infested areas. The control measures represent an acceptable standard that is to be applied in all municipalities across the province. Municipalities can enhance the standard within their own jurisdictions. Failure to meet these recommendations may result in action being taken by the Agricultural Fieldmen to ensure compliance. Further information on the regulatory status of clubroot in Alberta is available on the Government of Alberta website.
Saskatchewan
Information adapted from the Saskatchewan’s Clubroot Management Plan, July 2018:
Clubroot was declared a pest under The Pest Control Act in Saskatchewan in June 2009. Under The Pest Control Act, people who own, occupy or control land are required to take measures to destroy, control and prevent the spread of pests. The Saskatchewan Ministry of Agriculture and the Saskatchewan Clubroot Initiative continues to provide information for public awareness and contribute to disease surveillance in Saskatchewan. Rural municipalities have the authority under The Pest Control Act to undertake prevention and enforcement measures related to the spread and control of clubroot disease. For more information on clubroot management in Saskatchewan visit the Saskatchewan Clubroot Initiative’s Clubroot Management Plan.
Manitoba
Currently there is no provincial legislation that regulates clubroot. Manitoba Agriculture does provide some helpful information on crop biosecurity, which is useful for clubroot management. There is also additional clubroot information on the Manitoba Agriculture website.
Guidelines for Canola Researchers
The canola research industry, as represented by the WCC/RRC (Western Canadian Canola/Rapeseed Recommending Committee), initially adopted these Clubroot Management Guidelines to help ensure that canola research and cultivar trials would not contribute to the spread of clubroot on the Prairies.
Research
Significant grower, government and private dollars are being invested into clubroot research to work towards a solution for this major disease of canola.
Clubroot research funded through the Canola Agronomic Research Program
Grower funds are directed through the Canola Agronomic Research Program and other funding agencies. These projects include:
- 2004-21: Characterization of the clubroot disease problem on canola, Lead: Dr. Stephen Strelkov, University of Alberta. Full Summary Report Science Snapshot
- 2008-14: Seedborne transmission of clubroot of canola: evaluation and significance and control, Lead: Dr. Stephen Strelkov, University of Alberta. Final project report (PDF)
- 2008-16: Survey and evaluation of soil micro-organisms for control of clubroot on canola, Lead: Dr. Gary Peng
- 2008-19: Effect of non-host crops and host management on clubroot of canola, Leads: Dr. Sheau-Fang Hwang and Dr. Stephen Strelkov, Alberta Agriculture and Rural Development and University of Alberta. Final project report (PDF).
- PCARP: Survey and evaluation of soil micro-organisms for control of clubroot on canola, Lead: Dr. Gary Peng, AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More.
- 2009-18: Evaluation physical and chemical methods for cleaning and disinfesting tools, machinery, equipment, and other hard surfaces contaminated with clubroot, Lead: Dr. Ron Howard, Alberta Research Council.
- 2010-03: Effects of clubroot-resistant canola lines on soils infested by Plasmodiophora brassicae, Lead: Dr. Sheau-Fang Hwang.
- 2010F133R: Studies on the Genetic and Molecular Basis for Clubroot Resistance in Canola, Lead: Dr. Stephen Strelkov.
- ADF 2010-0098: Genomics of clubroot disease development in canola and development of in planta RNAi to impart novel resistance, Lead: Dr. Peta Bonham-Smith.
- 2012-5: Biocontrol of Clubroot and Blackleg by the Endophytic Micro-organisms, Lead: Paul Holloway
- 2015-6: Identification of genetic mapping of Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More for resistance to pathotype 5x, Lead: Dr. Fengqun Yu
- 2015-14: Characterization of the new strains of the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More in Alberta, Lead: Dr. Stephen Strelkov and Dr. Sheau-Fang Hwang
- 2015-15: Using SNP markers to assess genetic variability of Plasmodiophora brassicae in Canada, Lead: Dr. Bruce Gossen
- 2017-5: Introgression of clubroot resistance from B. rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More into B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More canola and identification of molecular markers for resistance, and pyramiding of this resistance with other resistance genes, Lead: Dr. Habibur Rahman
- 2018-20: Development of a harmonized clubroot map, Lead: Dr. Stephen Strelkov
- 2019-6: Managing small patches of clubroot infestations in canola fields, Lead: Dr. Bruce Gossen
- 2019.9: Influence of pH on the clubroot pathogenA disease-causing organism (such as a fungus or bacteria). More: are there pH-insensitive strains, Lead: Dr. Stephen Strelkov
- 2019.28: From field to the genome. Application of 3rd generation sequencing to direct genotyping of canola pathogens, Lead Dr. Bruce Gossen
- 2020-7: A proteomics-based approach towards identifying host and pathogenA disease-causing organism (such as a fungus or bacteria). More proteins critical to clubroot establishment in canola, Lead: Dr. Christopher Todd
- 2020-16: Beyond R-genes: candidate gene search for new sources of broader resistance to clubroot in Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More, through omics approaches, Lead: Dr. Dr. Guanqun (Gavin) Chen
Clubroot research funded through federal government programs and initiatives
The federal government of Canada committed 3.6 million dollars of the Growing Forward fund to clubroot research in 2009. As a result the Clubroot Risk Mitigation Initiative was launched to bring researchers across the canola industry together to collaborate on the toughest clubroot issues. These projects are jointly administered by the Canola Council of Canada and Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More. Some of these projects are:
- Development of a Canadian clubroot differential set
- Clubroot resistance stewardship: disease surveillance and pathotype monitoring
- Mechanisms of resistance
- Study of Plasmodiophora brassicae inoculum dispersal in western Canada
- Establishment of a clubroot nursery
- Integrated control strategies to reduce the impact of clubroot on canola
- Evaluating methods for disinfesting tools, machinery and other hard surfaces contaminated with clubroot resting spores
- Molecular mapping of the clubroot resistance of Rutabaga (B. napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More var. rapifera)
- Mapping clubroot resistance genes in B. rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More and developing molecular markers closely linked to these genes
- Interaction of climate, soil and pathogenA disease-causing organism (such as a fungus or bacteria). More biology on management of clubroot of canola
- Screening brassica germplasm for resistance and developing canola lines against prairie races of Plasmodiophora brassicae
- Developing microbial technologies for biocontrol of clubroot
- Formulation development for biological control of clubroot on canola
- Public and private breeding programs are also working to develop more clubroot-resistant cultivarsCultivars are variants in a species developed through the intervention of humans (despite the term 'variety' often being incorrectly used to describe this). Cultivars can be open-pollinated type, hybrid, synthetic, composite, etc. More for western Canada.
- Final Report for the Clubroot Risk Mitigation Initiative (CRMI) is available here.
Clubroot research shared by the Clubroot Steering Committee
Clubroot Steering Committee meeting – April 30, 2020 (International research update)
- Clubroot Steering Committee meeting Agenda
- Clubroot Steering Committee meeting notes with Q&A
- Sweden clubroot update – Dr. Ann-Charlotte Wallenhammar, Swedish University of Agricultural Sciences
- Clubroot in Poland- What’s new: pyramiding of resistance and mining the soil microbiome – Dr. Małgorzata Jędryczka, Polish Academy of Sciences
- The changing clubroot threat and sustainable management options in the UK – Dr. Fiona Burnett, Scotland’s Rural College (SRUC)
- Clubroot situation in Germany – Dr. Elke Diederichsen, Freie Universität Berlin
- Survey/Management options in North Dakota, USA – Dr. Venkat Chapara, North Dakota State University
- Evaluation of various strategies for the integrated management of clubroot of canola – Brittany Hennig, University of Alberta
- Clubroot Pathotypes – Keisha Hollman, University of Alberta
- Development of harmonized clubroot maps – Dr. Yoann Aigu, University of Alberta
- Clubroot management update – Dr. Bruce Gossen, Agriculture and Agri-Food Canada Saskatoon and Dr. Mary Ruth McDonald, University of Guelph
- Using RNA interference as a biological pesticide for clubroot in canola – Dr. Laura Keffer-Wilkes, University of Lethbridge
- Resistant sources and resources for clubroot at AAFC, Saskatoon – Dr. Fengqun Yu, Agriculture and Agri-Food Canada Saskatoon
- In-field distribution of Plasmodiophora brassicae inoculum and its relationship to soil pH, Ca and B – Andrea Botero-Ramírez, University of Alberta
- Harnessing Plant Immunity for Durable Clubroot Resistance – Dr. Edel Pérez-López, University Laval
- Biocontrol potential for clubroot by Acremonium alternatum- chances and challenges – Dr. Jutta Ludwig-Müller and Dr. Susann Auer, Technische Universität Dresden
Clubroot workshops and summits
2018 International Clubroot Workshop

The Canola Council of Canada hosted the International Clubroot Workshop in Edmonton, Alberta on August 7-9, 2018. This unique three-day program highlighted the latest in clubroot research from across Canada and around the world.
Topics included epidemiology, host-pathogen interaction, genetics and breeding, expanding clubroot resistance and stewardship, and clubroot management.
2018 International Clubroot Workshop Agenda
2018 International Clubroot Workshop Abstracts
- 2018 International Clubroot Workshop Opening – Dr. Curtis Rempel, Canola Council of Canada
- Clubroot status in Colombia – Andrea Botero-Ramírez, Fabian Padilla-Huertas, Lina Tarazona, Camilo Gómez, Juan Sebastián Navas and Celsa García
- Developing spring type Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More lines containing single clubroot resistance genes – Dr. Fengqun Yu, Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More
- Evaluation of various soil amendments to manage Clubroot on Canola in field condition – Dr. Venkat Chapara, North Dakota State University
- Genome similarity of Plasmodiophora brassicae collections from Canada, China, and the USA – Afsaneh Sedaghatkish, University of Guelph
- Impact, management and control of clubroot disease in the UK – Professor Fiona Burnet and Dr. Julie Smith, Scotland Rural College
- Influence of nitrogen constraint on quantitative resistance to clubroot in Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More – Dr. Maria Manzanares-Dauleux, University of Rennes (Agrocampus Ouest)
- Lime & Limestone production, products and trends in industry – a mining company’s perspective – Alexander Nguyen and Brad Anderson, Graymont
- Management of Clubroot of Canola in Alberta, Canada – Dr. Sheau-Fang Hwang, Alberta Agriculture and Forestry, and Dr. Stephen Strelkov, University of Alberta
- Management of clubroot: An overview of the challenges – Dr. Mary-Ruth McDonald, University of Guelph, and Dr. Bruce Gossen, Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More
- Managing small areas of clubroot infestation – Dr. Bruce Gossen, Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More and Dr. Mary-Ruth McDonald, University of Guelph
- Mechanisms and generational durability of clubroot resistance associated with stacked CR genes – Dr. Gary Peng, Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More
- Ocurrence of Plasmodiophora brassicae in agricultural soils, pathotype variation and means of clubroot control in Poland – Małgorzata Jędryczka, Institute of Plant Genetics, Polish Academy of Sciences
- Overview of the clubroot incidence and variation in the pathotypes of Plasmodiophora brassicae population in Europe – N. Zamani-Noor, A.-C. Wallenhammar, G. Cordsen-Nielsen, G. Orgeur, V. Konradyová, F. Burnett, F. Dussart, J. Smith, M. Korbas and M. Jedryczka
- Resting spores of clubroot detection service by LAMP method in Japan – Kenji Wakayama, Ministry of Agriculture and Forestry Japan
- The Changing Face of Clubroot in Canada – S.E. Strelkov (University of Alberta), S.F. Hwang (Alberta Agriculture and Forestry), M.D. Holtz (Alberta Agriculture and Forestry), V.P. Manolii (University of Alberta), M.W. Harding (Alberta Agriculture and Forestry)
- The impact of clubroot resistant canola varieties on Plasmodiophora brassicae resting spore concentrations in the soil – T.W. Ernst, University of Alberta
- Transcriptome changes in Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More varieties upon interaction with Plasmodiophora brassicae pathotype 5X – L. Galindo-González (University of Alberta), S.F. Hwang (Alberta Agriculture and Forestry) and S.E. Strelkov (University of Alberta)
- Transferring clubroot resistance by intergeneric hybridizations between Brassica napusAlso referred to as Argentine canola, it is the species of canola currently commonly grown in Canada. More and Raphanus sativus – Elke Diederichsen, Freie Universität Berlin
- Using knowledge on plant hormone metabolism by Plasmodiophora brassicae – a possibility to control the clubroot pathogen – Jutta Ludwig-
2013 International Clubroot Workshop

On June 19 to 22, the 2013 International Clubroot Workshop was held in Edmonton, Alberta, which brought together clubroot researchers from around the world along with canola growers, agronomists, petroleum industry and government staff all together to discuss clubroot. This Workshop was also the culmination of the Clubroot Risk Mitigation Initiative in which all research projects results were shared. Below are copies of the presentations that were delivered over the course of the workshop.
- Clubroot – A Biological and Agricultural Challenge Worldwide – Dr. Geoffrey R. Dixon, University of Reading, United Kingdom
- Clubroot and the Importance of Canola in Canada – Dr. Curtis Rempel, Canola Council of Canada
- Clubroot in Europe – Dr. Elke Diederichsen, Freie Universität Berlin
- Current Incidence of Clubroot on Oilseed Rape in East Europe – Dr. Malgorzata Jędryczka, Polish Academy of Sciences
- Clubroot, a Permanent Threat to Swedish Oilseed Rape Production – Dr. Ann-Charlotte Wallenhammar, Swedish University of Agricultural Sciences
- Research Status of Clubroot Disease of Cruciferous Crops in China – Dr. Yueqiu He, Yunnan Agricultural University
- Hormone Signaling During the Development of the Clubroot Disease in Arabidopsis thaliana Roots – Dr. Jutta Ludwig-Müller, Technische Universität Dresden
- Breeding for Clubroot Resistance at the University of Alberta – Dr. Habibur Rahman, University of Alberta
- Identification of Clubroot Resistance Genes and Development of Clubroot Resistant Canola Germplasm – Dr. Fengqun Yu, Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More
- Mapping Clubroot Resistance Genes in Chinese Cabbage and ECD Accessions of Brassica rapaAlso referred to as Polish canola, it is the less commonly grown species of canola currently grown in Canada. More – Dr. Genyi Li, University of Manitoba
- Integrated Management of Clubroot on Canola with Crop Rotation, Variety Resistance, and Fungicides/Biofungicides – Dr. Gary Peng, Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More
- Environmental parameters that Affect Clubroot Risk on the Canadian Prairies – Dr. Bruce Gossen, Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More
- Evaluating Methods for Cleaning and Disinfesting Equipment Contaminated with Clubroot – Dr. Ron Howard, Alberta Agriculture and Rural Development
- Agricultural Industry Perspective – Paul Muyers, Solid Ground Solutions Ltd.
- Petroleum Industry Perspective – Andrea Bullinger, Encana
- Construction Industry Perspective – Shannon Leggo, ATCO Electric
- Custom Equipment Cleaning and Decontamination – Sean Hannigan, Swift Environmental
- Clubroot Management – Clinton Jurke and Dan Orchard, Canola Council of Canada
- Clubroot Regulation in Leduc County – Aaron van Beers, Leduc County
- Clubroot Regulation in Manitoba – Holly Derksen, Manitoba Agriculture
- Clubroot Regulation in Saskatchewan – Dr. Cecilia Peluola, Saskatchewan Ministry of Agriculture
- 2013 International Clubroot Workshop agenda
- Poster presentation abstracts
- Oral presentation abstracts
2012 Clubroot Summit
- Clubroot resistance development at AAFC Saskatoon
- Saskatchewan – present clubroot situation
- Alberta – present clubroot situation
- Mapping clubroot resistant genes in B Rapa
- Clubroot resistance development and mapping from B napus rutabaga
- Clubroot biocontrol and fungicides projects
- Clubroot management projects
- Development of new clubroot differentials – AAFC Saskatoon
- Clubroot pathology projects from AAFC Saskatoon
- Clubroot pathology projects from U of Alberta
- Development of new clubroot differentials – U of Alberta
- Clubroot sanitation
2010 Clubroot Summit
Summary notes from the 2010 Clubroot Summit
2009 Clubroot Summit
The first Clubroot Summit was held in 2009 and this served to launch the Clubroot Risk Mitigation Initiative.
- Summary notes from 2009 Clubroot Summit
- Alberta clubroot survey 2008. Murray Hartman, Alberta Agriculture and Rural Development.
- Cleaning and disinfesting machinery and equipment. Ron Howard, Alberta Research Council.
- Clubroot biocontrol. Gary Peng, AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More.
- Clubroot management research. Gary Peng, AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More.
- Development of clubroot resistant canola. Habibur Rahman, University of Alberta.
- Integrated management of clubroot of crucifers. Stephen Strelkov, University of Alberta.
- Pathology Model – clubroot vision. Stephen Strelkov, University of Alberta.
- Pioneer 45H29. Igor Falak, Pioneer Hi-Bred.
- Potential distribution and severity of clubroot in western Canada. Kelly Turkington and Randy Kutcher, AAFCAgriculture and Agri-Food Canada is a department of the Government of Canada. More.
- Proposed clubroot breeding research. Habibur Rahman, University of Alberta.
- Resistance rating scale for clubroot. Stephen Strelkov, University of Alberta.
- Seedborne transmission of clubroot. Stephen Strelkov, University of Alberta.
Clubroot Resources
Clubroot management factsheets
Clubroot management video
Video (YouTube): Clubroot Management | Teaser | Trailer 1: Resilient Resting Spores | Trailer 2: Potential Vectors for the Disease
Chapter 1 | Chapter 2 | Chapter 3 | Overview (video segment) | Summary (video segment)
Video (Vimeo): Clubroot Management | Teaser | Trailer 1: Resilient Resting Spores | Trailer 2: Potential Vectors for the Disease
Additional Content: Digital Ad | Cover image | Canola Watch article
The clubroot management video was funded by Alberta Canola, SaskCanola and the Manitoba Canola Growers and produced by the Canola Council of Canada.
Clubroot of Canola Disease Cycle video
Clubroot equipment management resources
Managing Clubroot: Equipment Sanitation Guide
This pamphlet provides the steps needed to be taken when sanitizing field equipment.
Recommended Guidelines for Disinfesting Farm Machinery and Equipment (PDF)
This document outlines detailed steps that can be utilized to minimize the risk of clubroot spread via contaminated soil on field equipment. Any inquiries regarding this document should be directed to duke@gov.ab.ca.
Recommended Guidelines for the Oil and Gas Industry
The Canadian Association of Petroleum Producers adopted a set of best management practices to limit the spread of clubroot.
Recommendations for Managing Clubroot Risks Associated with Field Research (MS Word)
This document highlights the prevention measures that the agricultural research industry should practice to minimize the risk of clubroot movement. These guidelines were developed by the Saskatchewan Clubroot initiative for the larger agricultural industry.
See more clubroot-related resources in the Contact and Resources section.
Clubroot links and resources
August 2017 Release: Scout canola fields for clubroot galls
June 2014 Release: Equipment sanitation is first line of defense to help prevent spread of different clubroot pathotype
April 2014 Release: Clubroot data reinforces need for vigilant field scouting
Guidelines for Canola Researchers
The canola research industry, as represented by the WCC/RRC (Western Canadian Canola/Rapeseed Recommending Committee), adopted these clubroot management guidelines to help ensure that canola research and cultivar trials would not contribute to the spread of clubroot on the Prairies. View the management guidelines here
Agriculture and Agri-Food CanadaAgriculture and Agri-Food Canada is a department of the Government of Canada. More
Clubroot of Crucifers, Control Strategies
Alberta Agriculture and Rural Development
Alberta Clubroot Management Plan
Clubroot Diseases of Canola and Mustard
Manitoba Agriculture
Clubroot of Brassica Crops
Saskatchewan Ministry of Agriculture
Saskatchewan’s Clubroot Management Plan
Is Clubroot Really Important?
- Alberta Agriculture and Forestry (Government of Alberta). 2015. Clubroot Disease of Canola and Mustard. Agri-Facts, Agdex 140/638-1 (Revised September 2015). [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Government of Saskatchewan, 2024 [↩]
- The Canadian Phytopathological Society. 2014. Canadian Plant Disease Survey – Disease Highlights. Retrieved from https://phytopath.ca/wp-content/uploads/2014/10/cpds-archive/vol94/cpds_vol_94_2014.pdf. [↩]
- The Canadian Phytopathological Society. 2019. Canadian Plant Disease Survey – Disease Highlights. Retrieved from https://www.tandfonline.com/doi/full/10.1080/07060661.2019.1619270. [↩]
- Manitoba Agriculture. 2022. Clubroot Distribution in Manitoba. Available online (January 2023). [↩]
- Government of Alberta, June 2019. Preventing clubroot: agricultural sanitization: which disinfectants kill resting spores? Retrieved from https://open.alberta.ca/publications/prev enting-clubroot-agricultural-sanitization-which-disinfectants-kill-resting-spores [↩] [↩]
- Saskatchewan Ministry of Agriculture (Crops and Irrigation Branch), Saskatchewan Government [↩]
- Strelkov, S.E., Hwang , S.F., Howard, R.J., Hartman, M., & Turkington, T.K. 2011. Progress towards the Sustainable Management of Clubroot [Plasmodiophora brassicae] of Canola on the Canadian Prairies. Prairie Crops and Soils Journal, 4, 114-121. [↩]